Clear Sky Science · en
Protocol of the randomized double blind sham controlled AddVNS study of transcutaneous vagus nerve stimulation mechanisms in depression
Why this study matters
Many people with depression do not get enough relief from standard treatments like medication and talking therapy. Doctors and scientists are searching for safe add‑on options that can boost recovery without requiring major surgery. This study describes a carefully designed clinical trial testing a gentle form of electrical stimulation delivered to the ear, aimed at a major nerve that links the body and brain. By deeply tracking brain activity, body signals, blood chemistry, and gut microbes, the researchers hope to uncover how this treatment works and who might benefit most.
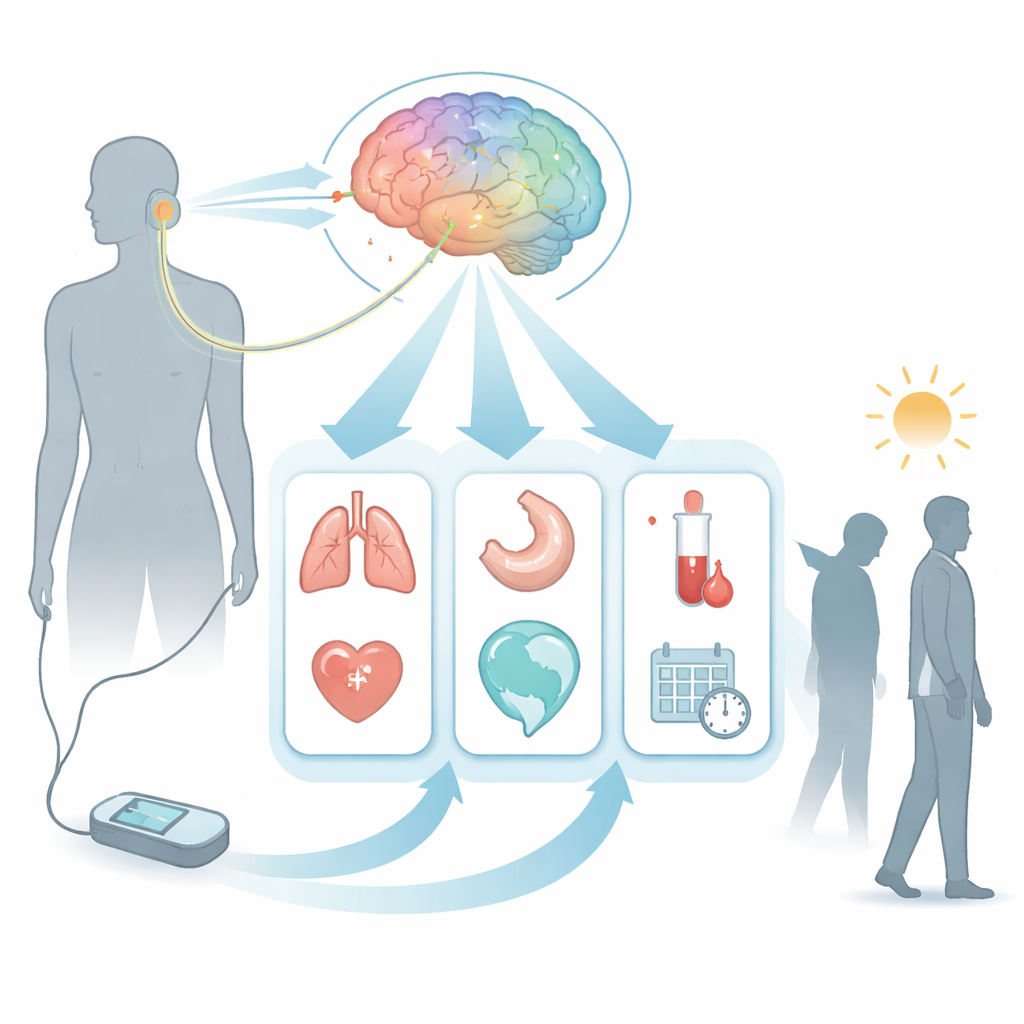
A gentler way to reach the brain
The vagus nerve is sometimes called a “superhighway” between the body and the brain. For years, doctors have used implanted devices on this nerve to help people with severe, treatment‑resistant depression, but surgery is costly and not without risk. The AddVNS study focuses on a noninvasive alternative: transcutaneous auricular vagus nerve stimulation, or tVNS. Instead of surgery, a small device sends mild electrical pulses through the skin of the outer ear, where a branch of the vagus nerve runs close to the surface. Early studies suggest this approach may ease depressive symptoms and is generally well tolerated, but its true effectiveness, ideal settings, and underlying biology are still unclear.
How the trial is set up
AddVNS is a single‑center, randomized, double‑blind, sham‑controlled trial conducted at a research hospital in Munich. Adult patients with a current depressive episode, either in major depressive disorder or bipolar disorder, receive their usual treatments plus either active tVNS or a sham version for six weeks. Both groups use the device three times a day on weekdays, with each session lasting 30 to 60 minutes depending on comfort. The active electrode stimulates a specific region of the left ear thought to be richly supplied by the vagus nerve; the sham electrode looks identical but delivers no current. Neither patients nor most of the study team know who is in which group, which helps separate true biological effects from expectations or placebo responses.
Looking at the whole person
What sets AddVNS apart is its “deep phenotyping” approach—an unusually broad and repeated measurement of mind and body. Participants undergo detailed psychophysiological testing, including heart and breathing signals, pupil responses, and stomach activity, as well as continuous activity tracking with a wrist device. Brain scans are performed before and after the six‑week stimulation period, using advanced magnetic resonance imaging to visualize key brainstem nuclei, reward circuits, and stress‑sensitive networks. At the same time, blood and stool samples are collected to probe genes, immune signals, hormones, tiny cell‑derived particles, and the composition of the gut microbiome. Standard depression scales, anxiety ratings, cognitive tests, and personality questionnaires round out the picture, repeated at several time points and followed for up to three months after stimulation ends.
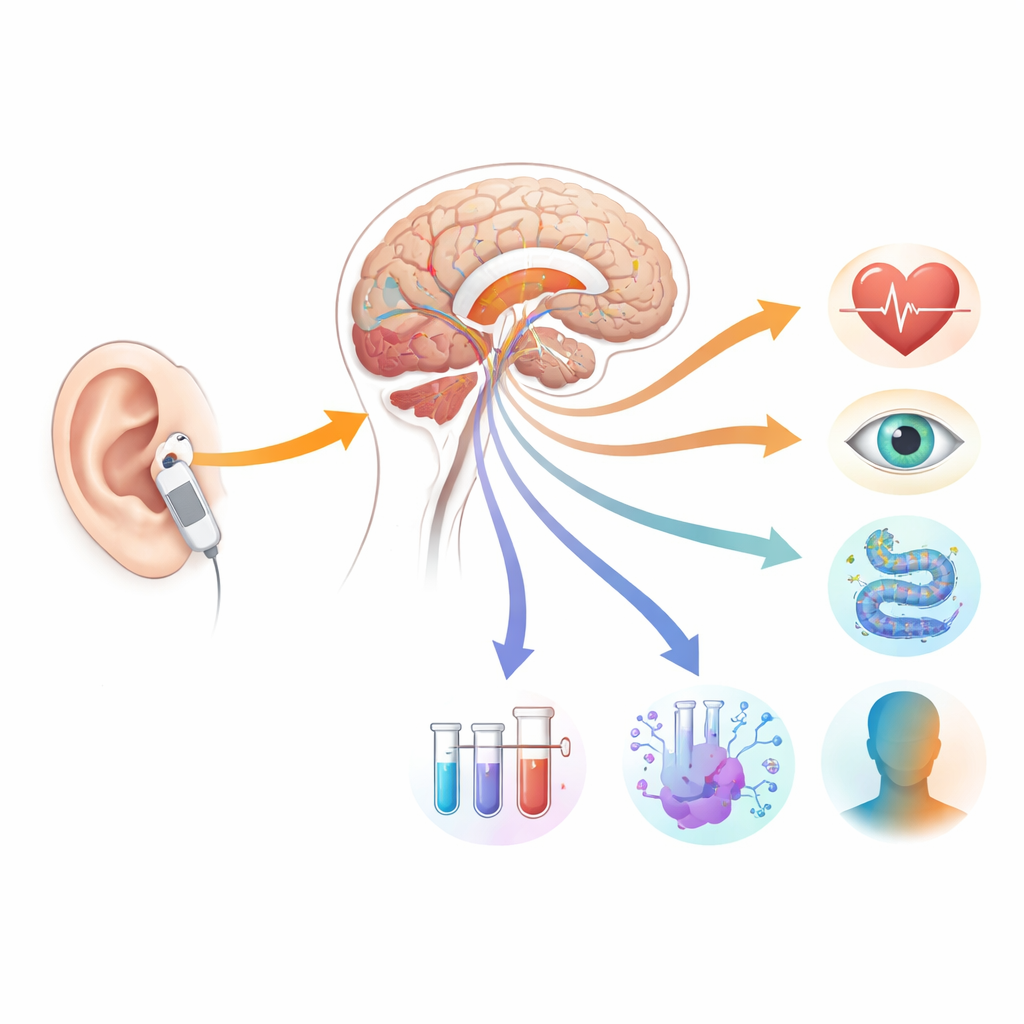
What the researchers hope to discover
The main aim is not just to see whether symptoms improve, but to learn how and why. The team expects tVNS to produce measurable changes in brain function, body rhythms, and molecular markers, and to test whether the size of these changes tracks with shifts in mood and daily functioning. They will compare active and sham groups over time, looking for patterns such as strengthened reward‑related brain activity, more balanced stress responses, altered inflammatory signals, and shifts in gut bacteria. By linking these readouts to stimulation dose and individual traits such as heart rate variability or personality dimensions, the researchers hope to identify biological “fingerprints” of people who respond well.
What this could mean for people with depression
If AddVNS can map reliable connections between ear‑based stimulation, biological changes, and symptom relief, it could move this approach from an experimental idea toward a more personalized treatment option. A clearer picture of how the vagus nerve influences mood—through the brain, immune system, and gut—may also point to new drug targets or behavioral strategies. While the study itself is exploratory and not yet designed to change clinical guidelines, its comprehensive design makes it one of the most ambitious efforts so far to understand this promising, low‑risk intervention for depression.
Citation: Kokolakis, E., von Mücke-Heim, IA., Pape, J.C. et al. Protocol of the randomized double blind sham controlled AddVNS study of transcutaneous vagus nerve stimulation mechanisms in depression. Sci Rep 16, 8149 (2026). https://doi.org/10.1038/s41598-026-42459-2
Keywords: depression, vagus nerve stimulation, brain-body connection, biomarkers, gut microbiome