Clear Sky Science · en
Pooled analyses of Clostridioides difficile vaccine trials identify baseline predictors for vaccine response
Why this vaccine story matters
Stomach bugs are usually a short-lived misery, but infections caused by the bacterium Clostridioides difficile can be severe, long-lasting, and sometimes deadly, especially in older or fragile patients. Scientists have been trying for years to develop a vaccine to prevent these dangerous infections, yet several major vaccine programs have failed in large trials. This study takes a different angle: instead of testing a new shot, the authors dig into data from two previous vaccine trials to ask a simple question with big implications—who actually mounts a strong immune response to a C. difficile vaccine, and what makes them different from those who do not?
The germ behind serious gut trouble
C. difficile lives in the environment and in many people’s intestines without causing symptoms. Problems start when the normal gut community is disturbed—often after antibiotic treatment—allowing the bacterium to flourish and release toxins that damage the gut lining. The result can be anything from persistent diarrhea to life‑threatening inflammation of the colon. Standard treatments rely on more antibiotics or antibody infusions and often fail to prevent the infection from coming back. Because of this, there has been strong interest in vaccines that teach the immune system to recognize and neutralize the two major toxins this bacterium produces, known as Toxin A and Toxin B.
Looking back at two large vaccine trials
The researchers combined detailed data from two clinical trials run by Sanofi, one mid‑stage (Phase II) and one large late‑stage (Phase III), together involving 1,096 adults who received three doses of an experimental vaccine or placebo. All participants had conditions that put them at risk of C. difficile infection, such as recent or upcoming hospital stays. Rather than asking whether the vaccine prevented disease—a question those trials had already struggled to answer—the team focused on how strongly each person’s blood antibody levels rose about a month after the final dose. They examined 16 different ways of defining a “good” antibody response and then used five complementary statistical approaches to search for patterns in who responded best.
Who responds best to the vaccine?
Across these many analyses, a consistent set of features emerged. People younger than 65 and those with fewer or milder existing illnesses were more likely to show strong increases in antibodies against both toxins. In other words, overall health seemed to matter more than age alone, with a higher burden of medical problems linked to a weaker vaccine response. Participants who were about to be hospitalized or enter a nursing or rehabilitation facility—"future risk"—also tended to respond better than those who had been in hospital or on antibiotics in the previous year, suggesting that timing of exposure and underlying frailty both influence how the immune system reacts to vaccination.
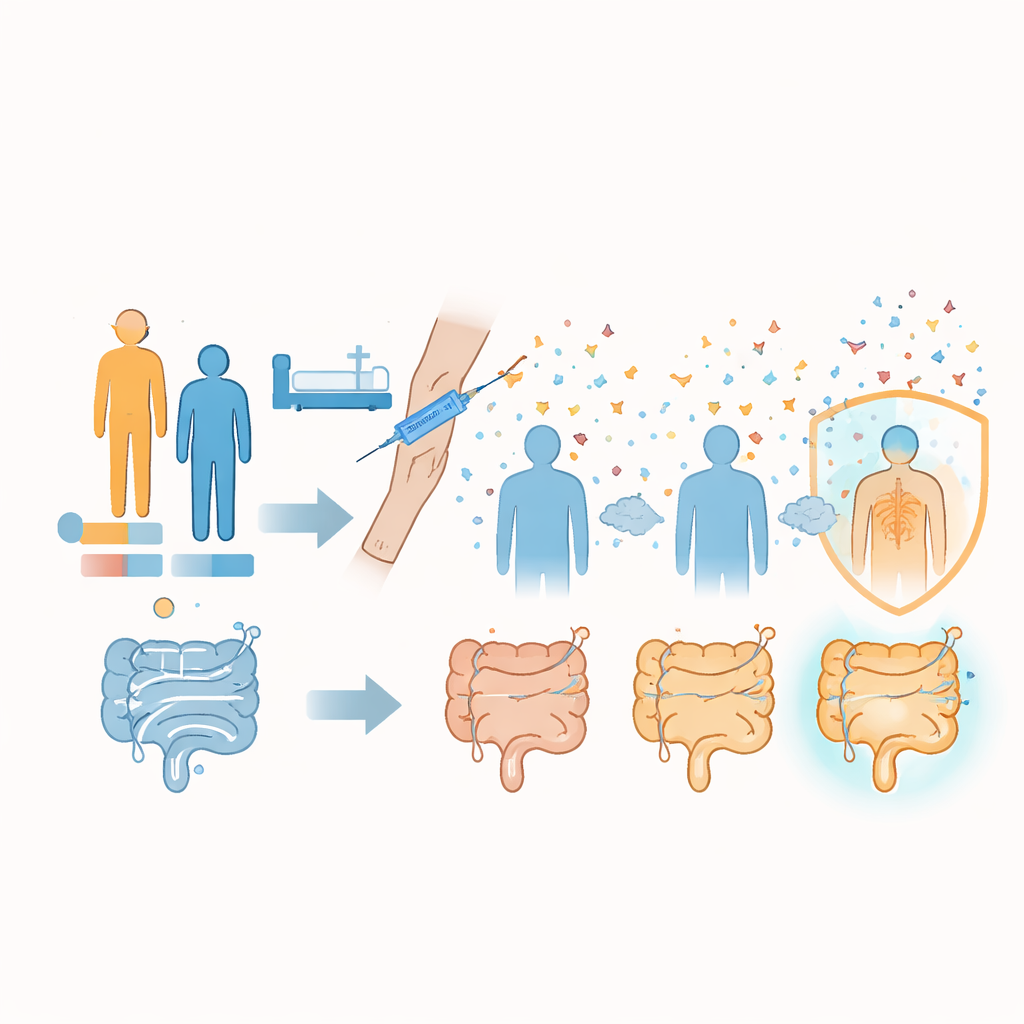
Clues from geography, sex, and prior immunity
Other differences were more specific to responses against Toxin B. Women, participants enrolled in North America, and people who already had higher baseline levels of antibodies before vaccination were more likely to develop especially strong responses to Toxin B after the shots. These patterns held up across multiple modeling methods, although the precise strength of each effect varied. The geographic signal may reflect differences in circulating C. difficile strains and their toxins, or differences in human genetics, healthcare settings, or unmeasured social factors. Pre‑existing antibodies likely point to earlier silent encounters with the bacterium that left behind memory immune cells, which a vaccine can then boost more effectively.
What this means for future vaccines
The study does not claim that any of these factors directly cause better or worse protection, and it cannot say whether strong antibody responses would have translated into fewer infections. Still, the patterns provide a roadmap for smarter vaccine development. They suggest that when testing C. difficile vaccines, researchers should pay close attention to participants’ overall health, upcoming hospital stays, existing antibody levels, sex, and where they live, and may want to design trials that deliberately balance or stratify for these features. For patients and clinicians, the broader message is that the success of a vaccine is not only about the shot itself, but also about the person receiving it. Understanding and using these baseline clues could eventually help tailor vaccination strategies so that those at highest risk of C. difficile infection also have the best chance of being protected.
Citation: Stojkov, I., Marchioro, L., Bekeredjian-Ding, I. et al. Pooled analyses of Clostridioides difficile vaccine trials identify baseline predictors for vaccine response. Sci Rep 16, 8981 (2026). https://doi.org/10.1038/s41598-026-42375-5
Keywords: Clostridioides difficile, vaccine response, gut infection, antibodies, personalized vaccination