Clear Sky Science · en
Corrosion mitigation of carbon steel in acidic solution using ionic liquids based on chemical, electrochemical, and characterization studies
Why protecting everyday steel really matters
From bridges and buildings to cars, pipelines, and oil rigs, much of our modern world rests on carbon steel. Yet this workhorse metal has a hidden weakness: in acidic conditions, such as those used to clean and maintain industrial systems, steel can dissolve rapidly. The resulting corrosion costs industries billions of dollars each year and can threaten safety. This study explores a new, more environmentally friendly way to shield carbon steel from harsh acid using a special class of salts called ionic liquids.
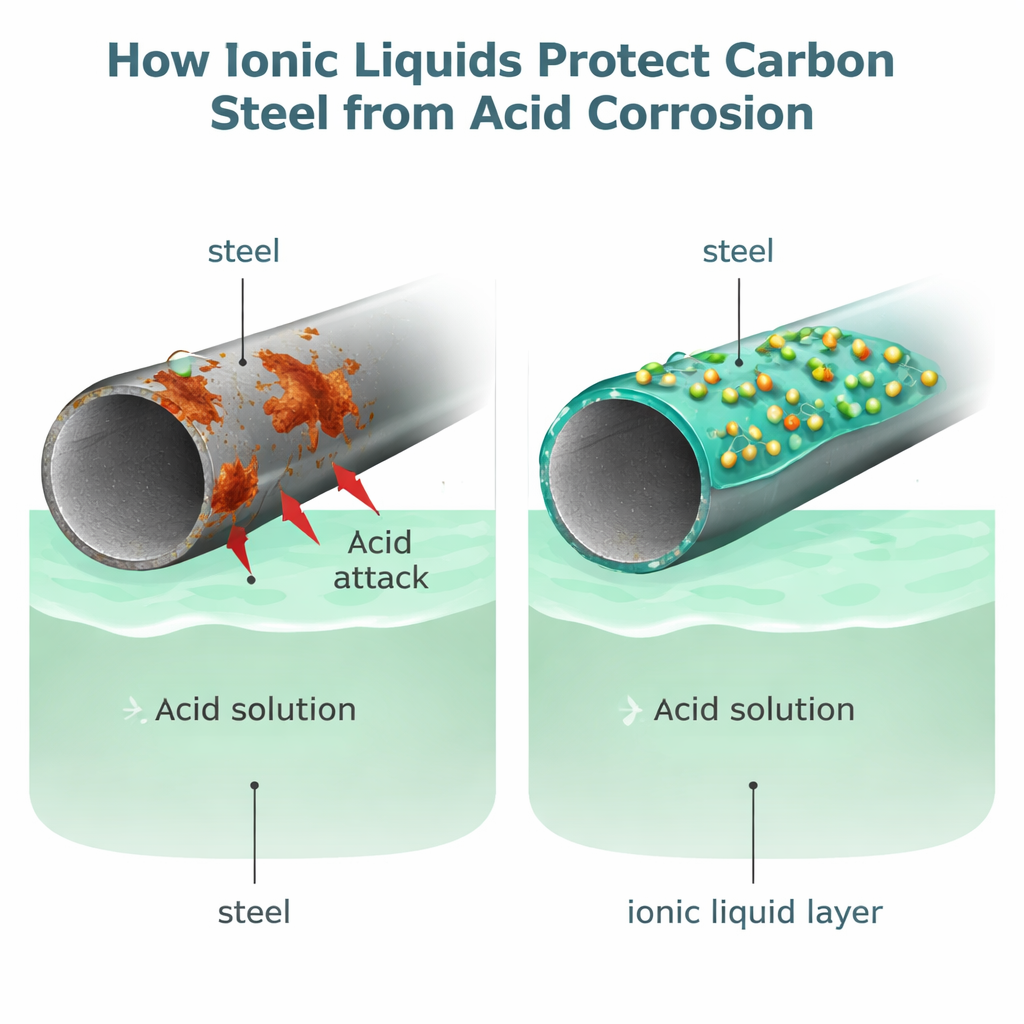
New liquids that act like smart shields
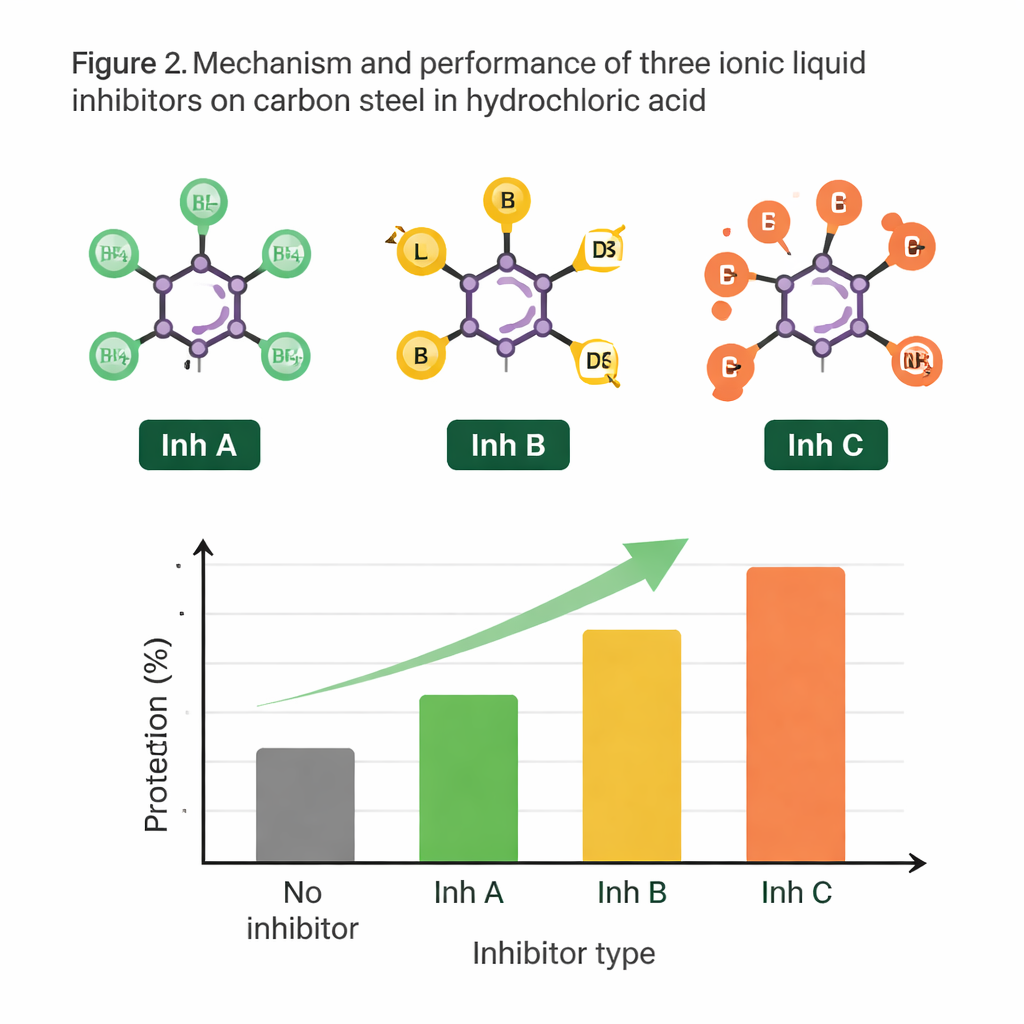
Ionic liquids are salts that are liquid at relatively low temperatures. They do not evaporate easily, can tolerate heat, and their structures can be tuned like Lego pieces. The authors focused on three closely related ionic liquids that all share the same positively charged "head"—an imidazolium group attached to butyl and methyl chains—but differ in their negatively charged partners: acetate (Inh A), hexafluorophosphate (Inh B), and tetrafluoroborate (Inh C). Because only the negative ion changes, any performance differences can be tied directly to how that part of the molecule behaves on steel in acid.
Putting steel in acid on purpose
To test these liquids, the researchers immersed small carbon steel samples in strong hydrochloric acid, similar to solutions used to clean industrial equipment. They measured how much metal was lost over time and used electrochemical methods to track how fast corrosion reactions occurred on the surface. At the same time, they used nuclear magnetic resonance spectroscopy and elemental analysis to verify that each ionic liquid had the intended composition and high purity. By testing a range of inhibitor concentrations and temperatures between 40 and 60 °C, they mimicked real operating conditions where both acid strength and heat can speed up damage.
How a thin molecular film holds back the acid
The central idea is that these ionic liquids form a protective film on the steel, blocking acid from reaching the metal. As the concentration of each ionic liquid increased, the corrosion rate dropped, and the calculated "surface coverage"—how much of the steel was blanketed by inhibitor molecules—went up. The data fit a well-known adsorption model, indicating that the liquids mainly cling to the surface through relatively weak physical forces rather than forming permanent chemical bonds. Even so, this physisorbed layer proved surprisingly effective. Microscopy images revealed that untreated steel developed rough, pitted surfaces packed with rust and salt deposits, while steel exposed to acid plus ionic liquids remained much smoother and cleaner.
Which liquid works best when things heat up
Across all three ionic liquids, protection improved at higher temperatures, a notable advantage because many industrial systems run hot. Among the inhibitors, Inh A—the acetate-based liquid—consistently provided the strongest defense. At 60 °C, it cut corrosion by about 97 percent compared with bare steel. Inh B and Inh C also worked well, but slightly less so. Electrochemical measurements showed that all three slowed both sides of the corrosion process, reducing the overall rate at which metal atoms dissolved and hydrogen gas formed. The authors conclude that the acetate ion and its interaction with the shared imidazolium head group promote a particularly robust and uniform protective film.
What this means for safer, greener infrastructure
For non-specialists, the takeaway is straightforward: by carefully designing the ingredients of these ionic liquids, scientists can create ultra-thin, invisible shields that dramatically extend the life of steel in harsh acidic environments. Such inhibitors could help reduce maintenance costs, limit unexpected failures in critical infrastructure, and replace more toxic traditional chemicals. While further work is needed to test long-term stability and performance in full-scale systems, this study shows that ionic liquids—especially the acetate-based Inh A—are promising tools for cleaner and more reliable corrosion control.
Citation: Deyab, M.A., El Rabiei, M.M., Mohamed, H.H. et al. Corrosion mitigation of carbon steel in acidic solution using ionic liquids based on chemical, electrochemical, and characterization studies. Sci Rep 16, 7944 (2026). https://doi.org/10.1038/s41598-026-42153-3
Keywords: corrosion, ionic liquids, carbon steel, acid protection, green inhibitors