Clear Sky Science · en
Multicenter clinicopathological study of odontogenic myxoma spectrum lesions using quantitative pathology
Why this matters for patients and dentists
Deep inside the jawbone, rare tumors can quietly grow for months or years before they are discovered. These growths, called odontogenic myxomas and myxofibromas, are not cancer, but they can expand, weaken bone, and complicate surgery. For decades, pathologists have judged them largely by eye, leading to differences in how hospitals describe and classify the same type of lesion. This study brings powerful image‑analysis tools to the microscope slide, showing how artificial intelligence (AI) can help doctors see these tumors more clearly and consistently.
Rare jaw tumors that look deceptively similar
Odontogenic myxoma and odontogenic myxofibroma arise from tissues involved in tooth formation. Both are soft, jelly‑like tumors that sit within the jawbone. Under the microscope, they contain a mix of loose, myxoid areas and denser fibrous bands. Traditionally, pathologists have tried to distinguish them by estimating how much of the tumor is fibrous versus myxoid. But these tumors are rare, and there are no universally agreed‑upon rules for how much fibrous tissue is enough to shift a diagnosis from one label to the other. As a result, different hospitals may give different names to very similar lesions, even when patients have comparable symptoms and scan findings.
Turning microscope slides into numbers
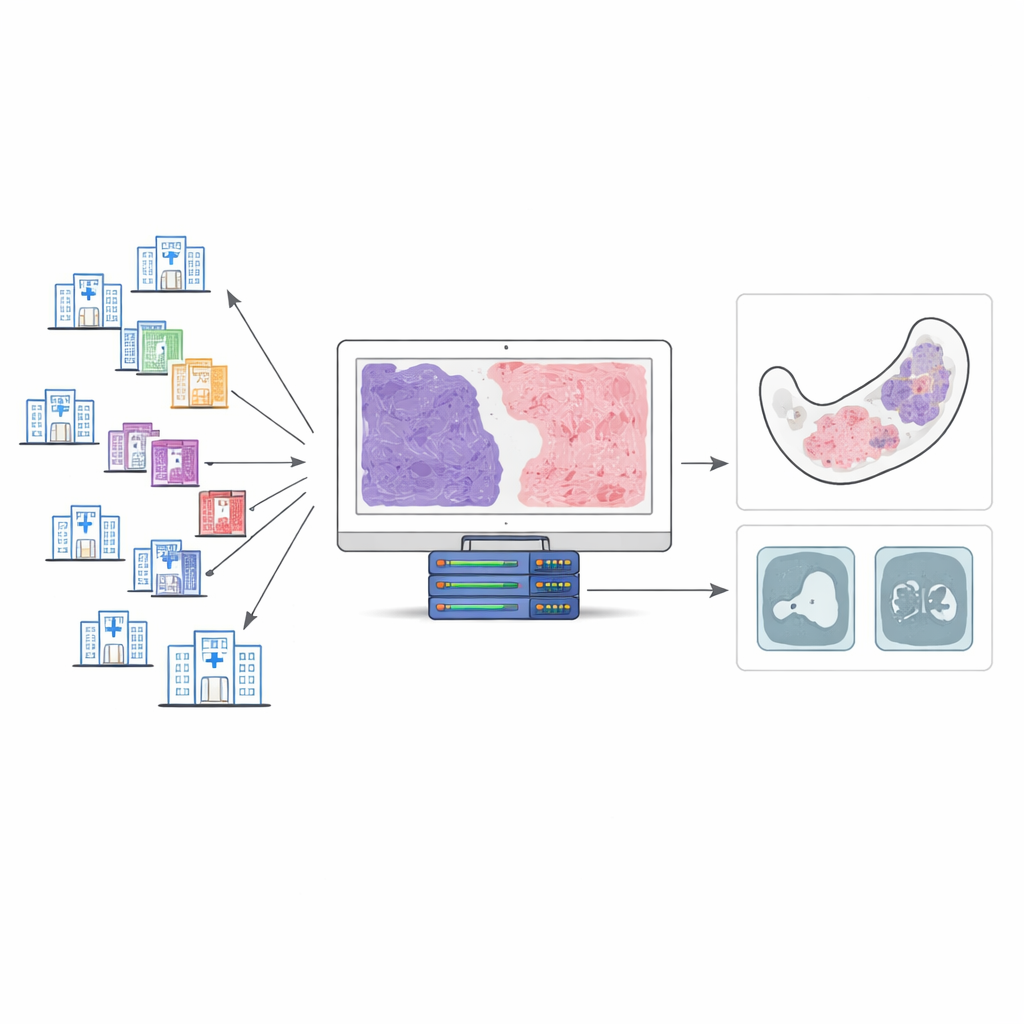
The research team collected 143 tissue samples from patients treated at 34 hospitals over two decades, ultimately focusing on 100 cases that met strict quality criteria. All samples were stained with a special dye (Masson’s trichrome) that colors fibrous tissue differently from the surrounding tumor. Two expert oral pathologists carefully reviewed each case and estimated the percentage of fibrous tissue in 10% steps. These expert estimates served as a reference for training an AI system to perform the same task automatically, but in a fully quantitative way.
How the AI "reads" a tumor slide
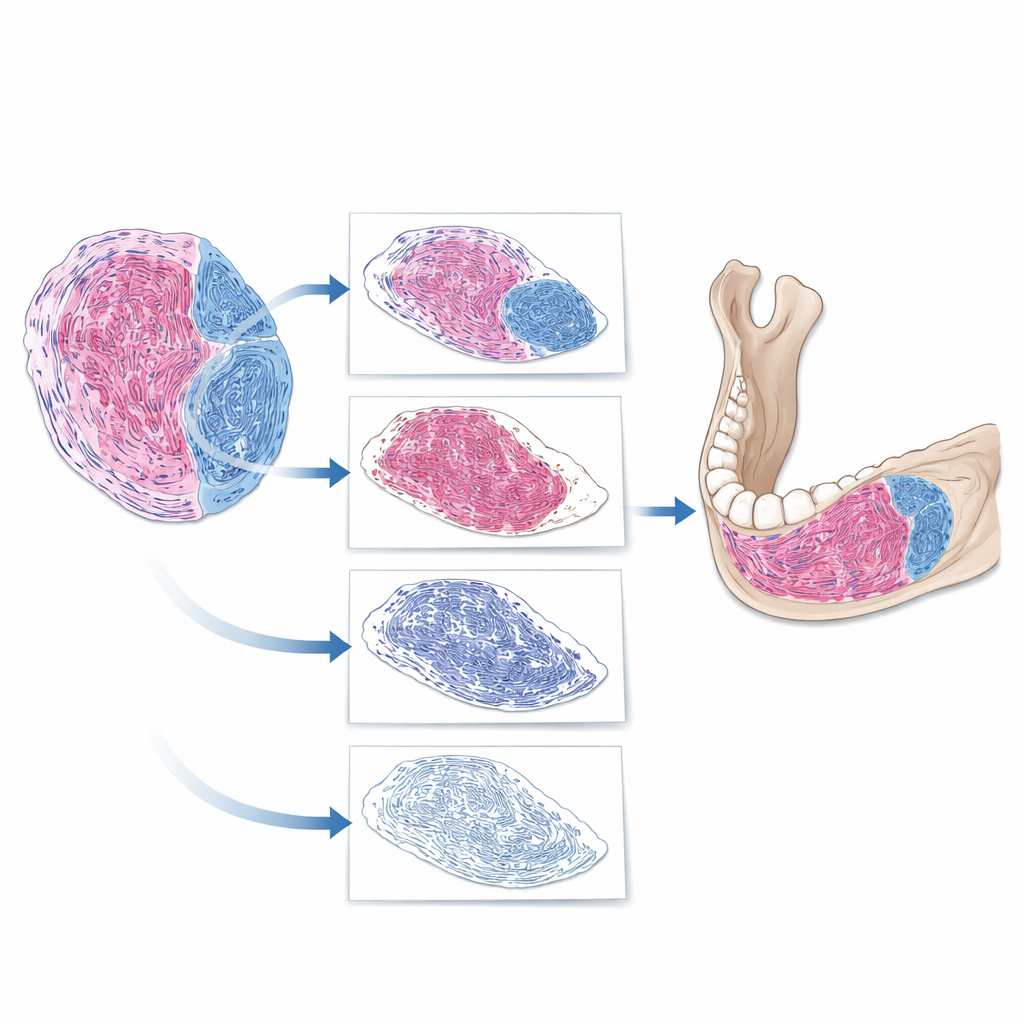
The team built a step‑by‑step digital pipeline to analyze whole‑slide images. First, the software separated true tissue from empty background on the slide. Next, it isolated the tumor from nearby structures such as bone and teeth. Finally, within the tumor, it removed cell nuclei and focused on the remaining material, identifying regions that took up the fibrous stain. From this, the AI computed a fibrous tissue proportion: the area of fibrous tissue divided by the total tumor area. When the researchers compared AI‑generated values with their own expert ratings, the agreement was strong, with only about a 10‑percentage‑point spread in most individual cases. This suggested that the AI could reliably mirror expert judgment, while expressing it as precise measurements instead of rough impressions.
What fibrous makeup reveals about tumor appearance
Armed with these measurements, the researchers examined how fibrous content related to clinical and radiological features. They found that the amount of fibrous tissue varied widely across institutions, reflecting differences in how pathologists had originally labeled the tumors. When they looked at imaging characteristics, one pattern stood out: tumors that appeared as a single cavity on scans (unilocular lesions) tended to have a higher proportion of fibrous tissue than those with multiple small compartments (multilocular lesions). This fits with the idea that more fibrous, dense tissue may form a more solid, single‑chambered mass, whereas looser, more fluid‑rich tissue may break up into several spaces.
What this means for future diagnosis and care
The study does not seek to rename diseases or replace pathologists with machines. Instead, it shows that AI‑based quantitative pathology can provide a shared measuring stick for rare jaw tumors across many hospitals. By turning the subtle textures of tumor tissue into reproducible numbers, this approach exposes how much everyday diagnoses depend on subjective judgment and offers a way to reduce that variability. For patients, more consistent interpretation of these lesions could support clearer treatment decisions and better comparison of outcomes between centers. As similar methods are extended to other rare tumors, AI‑assisted microscopy may quietly reshape how pathologists see disease—making their assessments more comparable, and ultimately more useful, wherever a patient happens to be treated.
Citation: Harazono, Y., Fukawa, Y., Iwasaki, T. et al. Multicenter clinicopathological study of odontogenic myxoma spectrum lesions using quantitative pathology. Sci Rep 16, 11221 (2026). https://doi.org/10.1038/s41598-026-42019-8
Keywords: odontogenic myxoma, jaw tumors, digital pathology, artificial intelligence, fibrous tissue proportion