Clear Sky Science · en
Genetic analysis of key players in PI3K signaling cascade of colorectal carcinoma
Why your genes matter for colon health
Colorectal cancer is one of the most common and deadly cancers worldwide, and its numbers are rising in many countries undergoing rapid lifestyle change. Yet not everyone with similar diets or habits faces the same risk. This study asks a simple but powerful question: do inherited differences in key growth‑control genes help explain who develops colorectal cancer, how aggressive it becomes, and how well standard drugs work—especially in a large group of patients from Pakistan, a population that has been under‑studied so far?
A busy control line inside our cells
Cells in the gut constantly receive signals telling them when to grow, divide, or self‑destruct. One of the busiest communication routes is the PI3K–AKT–mTOR pathway—a cascade of molecular switches that promotes cell growth, survival, blood‑vessel formation, and resistance to cell death. When this route is overactive, cells can start multiplying uncontrollably and form tumors. The genes PIK3CA, AKT1 and mTOR sit at crucial checkpoints along this pathway. Subtle inherited changes in these genes, called single nucleotide polymorphisms (SNPs), and stronger changes known as “hotspot” mutations may tilt the balance toward cancer or alter how tumors respond to treatment.
Scanning DNA in patients and healthy volunteers
To explore this, researchers carried out a case–control study that included 495 people with colorectal cancer and 495 cancer‑free volunteers matched for age and sex. Blood samples provided DNA, which was examined for seven SNPs across PIK3CA, AKT1 and mTOR, along with two well‑known hotspot mutations called E542K (in PIK3CA) and E17K (in AKT1). Using targeted PCR methods and DNA sequencing, the team compared how often each genetic variant appeared in patients versus controls and then linked these patterns to clinical features such as age, family history, bowel disease, tumor stage, and the drug regimens patients received. 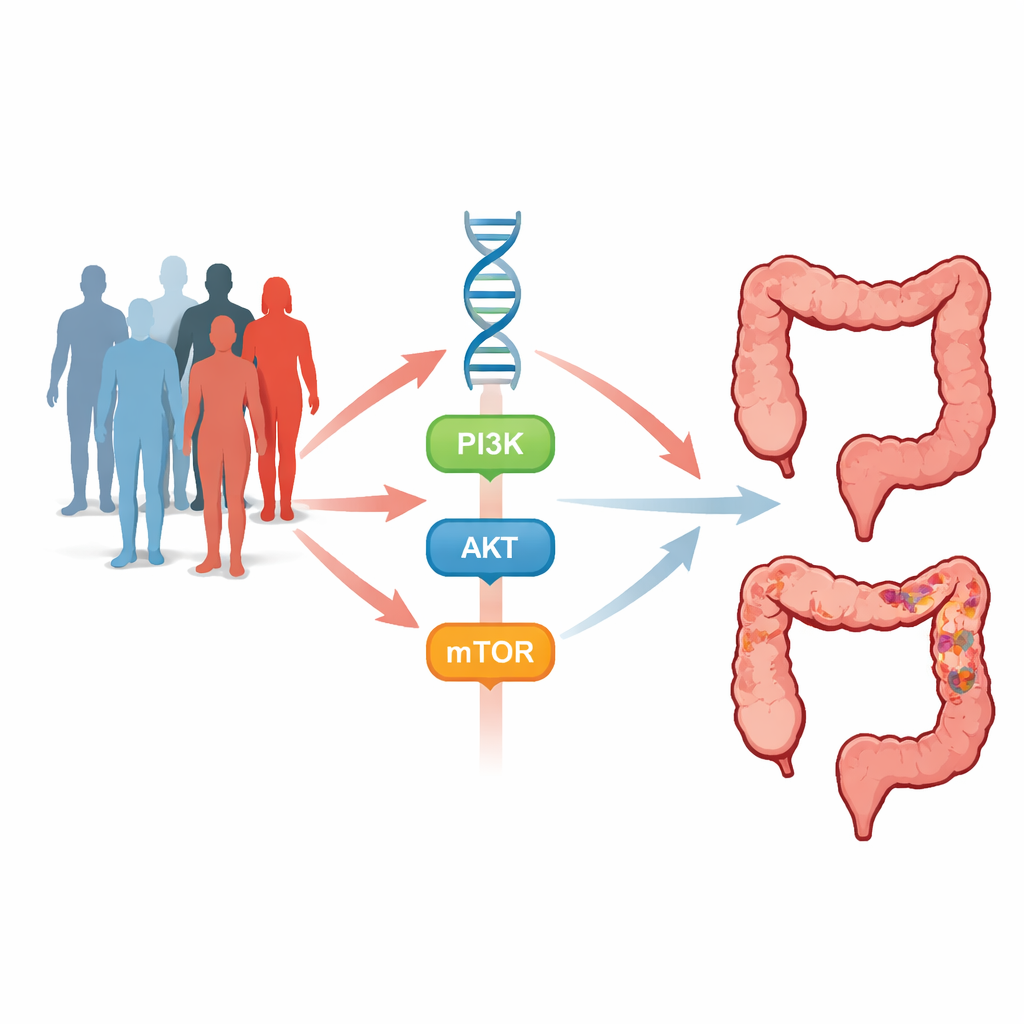
Risky variants and powerful mutations
The analysis revealed that most of the tested variants were far more common in people with colorectal cancer than in healthy controls, strongly suggesting they contribute to disease risk. Several AKT1 and mTOR variants, in particular, showed strikingly higher frequencies in patients, while certain “normal” versions of these genes were more common in controls, hinting at a protective role. The hotspot mutations told an even clearer story: the E542K change in PIK3CA appeared in about 15% of patients, and the E17K change in AKT1 in about 29%, both much more frequent than in controls. Computer‑based modeling indicated that these mutations do not dramatically distort the overall protein shape but instead seem to lock the pathway into a more “on” state, driving continuous growth signals that favor tumor development.
From genes to treatment and survival
Genetics alone does not act in isolation, so the researchers asked how these variants interact with real‑world risk factors and therapies. Many of the SNPs were linked to smoking, inflammatory bowel disease, family history, cancer type and stage, and whether patients had received chemotherapy or radiation. When the team followed patients over time, they found that certain gene versions were tied to better or worse overall survival. For example, people carrying normal versions of several AKT1 and mTOR variants tended to live longer than those with the high‑risk versions. The study also compared survival among patients receiving common drug combinations. Capecitabine, a widely used chemotherapy pill, was associated with the best survival estimates, and several of the pathway variants appeared to influence how well different regimens worked, hinting that inherited genetics could guide more personalized treatment choices. 
How changes travel together
Beyond individual variants, the authors examined how groups of changes tend to be inherited together—a pattern known as linkage disequilibrium. They discovered a block of neighboring variants spanning PIK3CA and AKT1 that traveled together more often in patients than in controls. This suggests that combinations of small DNA differences along the PI3K pathway may jointly shape a person’s baseline risk and how a tumor behaves, rather than any single change acting alone.
What this means for patients and precision medicine
For a lay reader, the bottom line is that this work strengthens the idea that “one size fits all” is a poor model for colorectal cancer care. In this Pakistani cohort, specific inherited variants and hotspot mutations in growth‑control genes clearly tracked with cancer risk, tumor features and survival, and they even appeared to modulate how patients fared on standard chemotherapy. While these findings need to be confirmed in larger and more diverse groups, they point toward a future in which a simple blood test for PI3K–AKT–mTOR variants could help identify those at higher risk, flag which patients might benefit from particular drugs, and spare others from ineffective or unnecessarily toxic treatments.
Citation: Pervaiz, H., Masood, N., Malik, P.A. et al. Genetic analysis of key players in PI3K signaling cascade of colorectal carcinoma. Sci Rep 16, 11317 (2026). https://doi.org/10.1038/s41598-026-42006-z
Keywords: colorectal cancer genetics, PI3K AKT mTOR pathway, single nucleotide polymorphisms, precision oncology, chemotherapy response