Clear Sky Science · en
Multi-epitope vaccine against nucleoprotein and envelopment polyprotein of Batai orthobunyavirus using molecular docking and molecular dynamics studies
Why a little-known virus matters
Batai orthobunyavirus is not a household name, but it is quietly spreading through mosquitoes across Europe, Africa, and Asia, infecting both people and farm animals. In livestock it can cause miscarriages and birth defects, and in humans it ranges from a flu-like fever to, in rare cases, brain inflammation and bleeding disorders. Yet there is no licensed vaccine and very limited testing. This study uses computer-based tools to design a first-generation vaccine blueprint that could, after future lab work, help protect both humans and animals from this overlooked threat.
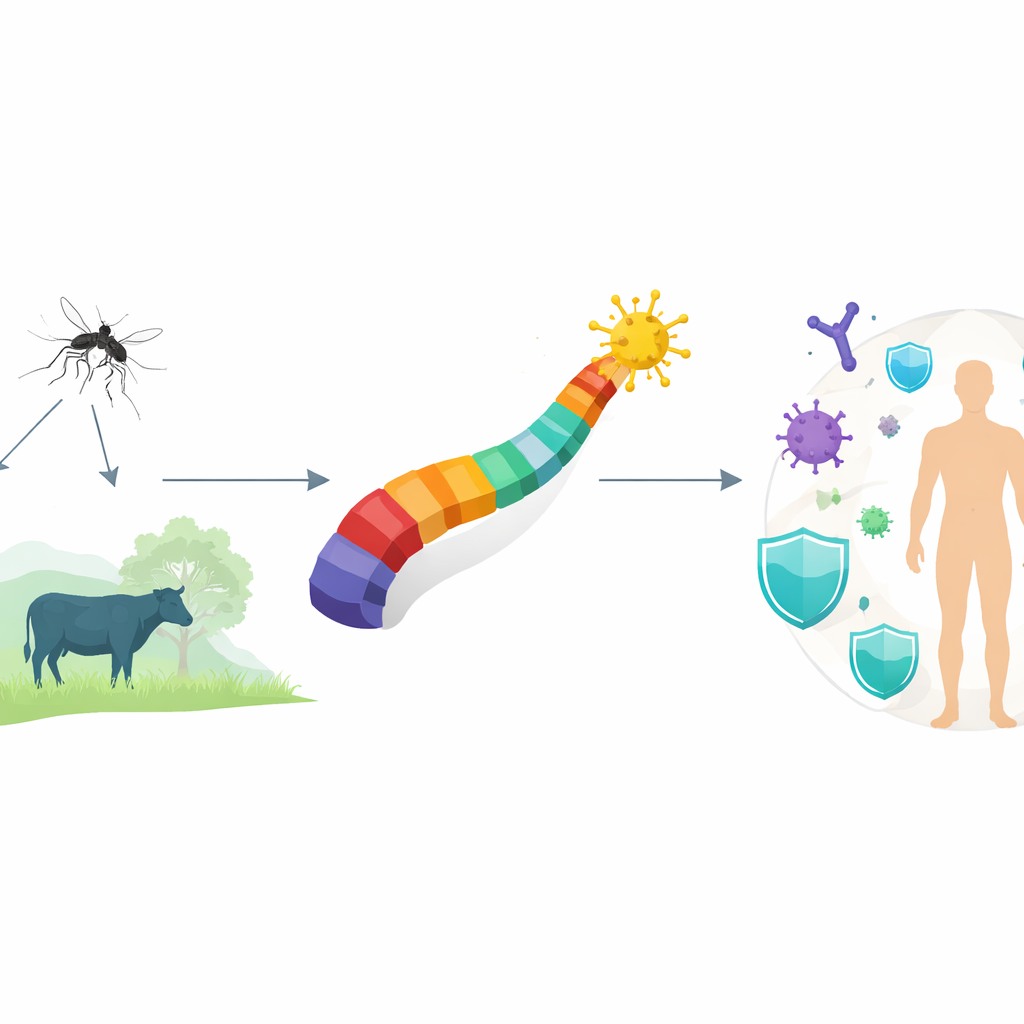
How the virus spreads and why it is missed
The virus travels mainly through common mosquitoes that also carry more familiar infections such as malaria and dengue. Because Batai infections often resemble routine fevers or flu, doctors rarely suspect it, and standard diagnostic panels do not usually test for it. Surveillance of mosquitoes and farm animals is patchy, so outbreaks can smolder undetected. The authors argue that this combination of broad geographic spread, effects on livestock reproduction, and underdiagnosis makes Batai orthobunyavirus a quiet but important public and veterinary health concern.
Building a vaccine from modular pieces
Instead of working with the whole virus, the researchers focused on two of its key proteins: the nucleoprotein, which helps package the viral genetic material, and the envelope polyprotein, which forms part of the virus’s outer coat. Using online databases and prediction servers, they scanned these proteins for short stretches—called epitopes—that immune cells are especially likely to recognize. They selected epitopes predicted to trigger strong immune reactions while avoiding those likely to cause allergies or toxicity. These pieces were then digitally stitched together, separated by flexible “linkers,” and combined with a small immune-boosting segment known as an adjuvant, forming a single chain of 247 amino acids that serves as their proposed vaccine construct.
Testing the design inside a computer
With the amino acid sequence in hand, the team examined whether this artificial protein should behave like a realistic vaccine candidate. Computational tools suggested that it would be stable, water-loving, and easy to produce in bacteria commonly used for vaccine manufacture. The predicted three-dimensional shape passed standard quality checks used for protein models. Importantly, when they simulated how the construct might interact with a human immune sensor called Toll-like receptor 3—a molecule that helps cells detect viral material—the modeled docking was tight and formed many stabilizing contacts. A 100-nanosecond molecular dynamics simulation showed that the complex stayed intact and compact, hinting that the interaction could be robust in real cells.
Will it trigger broad protection?
The authors next asked whether people around the world would be likely to respond to this design. They used a population coverage tool that matches the chosen epitopes to common versions of human immune genes. The analysis suggested that more than 97 percent of the global population carries at least one gene variant capable of recognizing parts of the construct, indicating broad theoretical reach. An immune system simulation predicted strong waves of antibodies, activation of helper and killer T cells, formation of immune memory, and engagement of innate defenders such as natural killer cells and macrophages. In virtual form, at least, the construct appears able to spark both arms of the immune response: antibody-based and cell-based.
What this work means going forward
This study does not produce a finished vaccine, but rather a carefully engineered starting point. All of the promising results—good stability, wide predicted population coverage, and strong simulated immune responses—come from computer models. The next steps are firmly experimental: making the protein in the lab, testing whether it is safe, and seeing if it actually protects animals, and eventually humans, from Batai infection. If future work confirms these predictions, the multi-epitope design outlined here could form the backbone of the first dedicated vaccine against this neglected mosquito-borne virus.
Citation: Naveed, M., Asim, M., Ali, A. et al. Multi-epitope vaccine against nucleoprotein and envelopment polyprotein of Batai orthobunyavirus using molecular docking and molecular dynamics studies. Sci Rep 16, 8973 (2026). https://doi.org/10.1038/s41598-026-41964-8
Keywords: Batai virus, mosquito-borne disease, epitope-based vaccine, computational vaccinology, multi-epitope design