Clear Sky Science · en
Visualizing malignant progression: in situ CD109-based spatial immunofluorescence assay delineates papillary to anaplastic thyroid carcinoma transformation within the tumor microenvironment
Why this thyroid cancer study matters
Anaplastic thyroid carcinoma is one of the most lethal human cancers, often turning a usually curable thyroid tumor into a rapidly fatal disease. Yet doctors still do not fully understand how a common, slow-growing papillary thyroid cancer can suddenly change into this aggressive form. This study follows that transformation directly inside a patient’s tumor, using a new imaging technique to watch how cancer cells and their surrounding neighborhood shift step by step. The work offers a more detailed picture of how the tumor’s local environment may push cells toward a deadlier state, hinting at new ways to slow or prevent this transition.
From a common tumor to a rare killer
Most thyroid cancers are papillary thyroid carcinomas (PTC), which generally grow slowly and have excellent outcomes. Anaplastic thyroid carcinoma (ATC), in contrast, is rare but often fatal within months. Genetic studies have shown that ATC and nearby PTC in the same patient frequently share many of the same DNA mutations, including changes in well-known cancer genes. That similarity suggests that genes alone may not explain why only some regions of a tumor become anaplastic. The authors therefore focused on what is happening in the tissue landscape around the cells—the tumor microenvironment—to see whether local conditions help drive this dangerous shift.
A new way to map cancer in place
The team developed a spatial immunofluorescence (SPI) assay, a type of multi-color microscopy that measures the brightness of specific proteins in tiny defined tiles across a tissue section. They centered this method on CD109, a surface protein they identified as a marker of malignant behavior in thyroid tissue, and CK8/18, a protein characteristic of ordinary papillary tumor cells. By computing the ratio of CD109 to CK8/18 in hundreds of small regions, they generated color-coded maps that separated clear PTC areas from clear ATC areas and, crucially, revealed intermediate zones where both markers overlapped. Instead of a sharp border, the transition from papillary to anaplastic cancer appeared as a gradual, patchy continuum.
Five zones along a path of change
To understand this continuum better, the researchers divided the tissue into five regions, from pure PTC to fully developed ATC. They then examined many additional proteins that report on cell identity, growth, movement, and the supporting tissue. Across these regions, classic epithelial features such as E-cadherin and CK8/18 steadily declined, while markers linked to cell motility and shape change, such as vimentin, went up. Proteins indicating rapid cell division rose in the more anaplastic zones. At the same time, the surrounding support cells—cancer-associated fibroblasts—and immune cells, especially certain macrophages, became more numerous and more activated, and the proteins they produced increased.
When the tumor scaffold turns hostile
A striking change occurred in the tumor’s scaffold, or extracellular matrix. In the anaplastic regions, fibroblast-like cells deposited more collagen types III and VI and a matrix protein called TGFBI, and these fibers were thicker, more tangled, and oriented in many directions rather than in smooth, parallel lines. This rearranged mesh likely makes the tissue mechanically stiffer. In these stiffer zones, a pressure-sensitive signaling protein called YAP was more active in both cancer and support cells. The same areas showed more signs of DNA damage and a form of inflammatory cell death, along with release of S100 proteins that can act as danger signals and further inflame the environment. The authors propose that this combination of compression, cell death, and chronic inflammation favors survival and spread of already mutated cancer cells that can withstand stress and undergo an epithelial–mesenchymal–like transition.
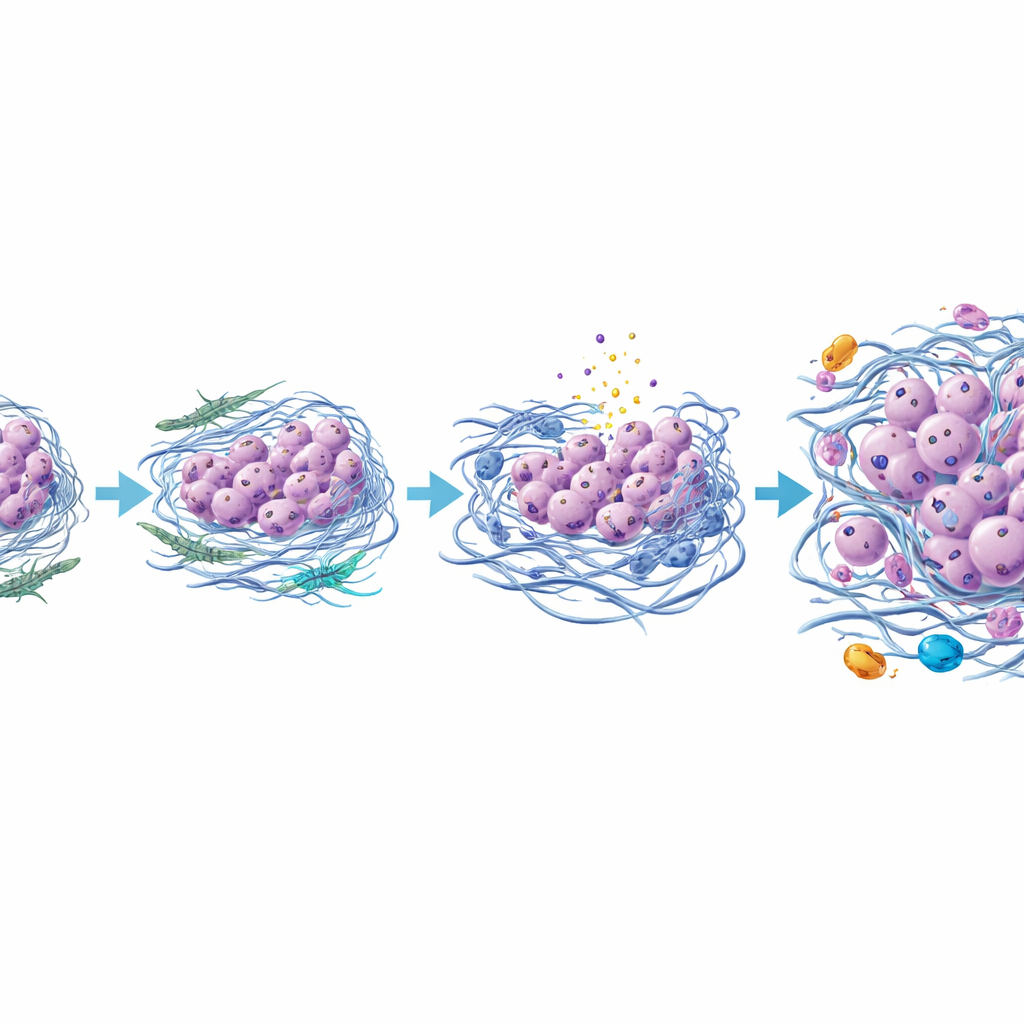
A new picture of how a thyroid tumor turns lethal
Putting these findings together, the study suggests that anaplastic thyroid cancer emerges not just from additional gene mutations but from a reshaped neighborhood around the tumor. As papillary tumor cells and nearby support cells interact, some fibroblasts evolve into a more aggressive type that thickens and stiffens the surrounding matrix, squeezes vulnerable cells to death, and calls in inflammatory immune cells. The surviving, more resilient cancer cells gradually lose their original thyroid features and adopt a more mobile, invasive identity, eventually forming an anaplastic tumor. While this work is based on a single well-studied case and needs confirmation in more patients, it introduces a powerful imaging tool and a detailed model in which targeting the tumor’s support cells, matrix, or inflammatory signals could help prevent or delay one of the most feared transformations in thyroid cancer.
Citation: Cohen, T., Suzuki, K., Matsuda, K. et al. Visualizing malignant progression: in situ CD109-based spatial immunofluorescence assay delineates papillary to anaplastic thyroid carcinoma transformation within the tumor microenvironment. Sci Rep 16, 8682 (2026). https://doi.org/10.1038/s41598-026-41927-z
Keywords: anaplastic thyroid carcinoma, papillary thyroid carcinoma, tumor microenvironment, extracellular matrix stiffness, spatial immunofluorescence