Clear Sky Science · en
Bronchial epithelial cell-derived extracellular vesicle analysis using conventional, imaging, and nanoscale flow cytometry technologies
Why Tiny Bubbles in the Lungs Matter
Our lungs are lined with cells that are constantly talking to the immune system, especially when we breathe in dust, pollution, or germs. Much of this “conversation” happens through microscopic bubbles called extracellular vesicles—packages of fats and proteins released by cells. These vesicles can worsen or calm lung disease, and they may one day serve as early warning signals for conditions like asthma, chronic obstructive pulmonary disease (COPD), and fibrosis. This study explores how best to measure these elusive particles using different types of advanced laser-based instruments, with an eye toward making such measurements practical for research and future clinical tests.
Invisible Messages from Airway Cells
Extracellular vesicles (EVs) are tiny, membrane-wrapped packets released by nearly all cells into fluids like blood, urine, and the thin liquid coating our airways. EVs from human bronchial epithelial cells—the cells that line the breathing tubes—have already been linked to several lung processes: they can influence how we develop allergies, help fight lung infections, shape airway structure in COPD, and even limit scarring in lung tissue. Because they carry bits of proteins, fats, and genetic material from their parent cells, EVs could act as fingerprints of lung health or disease, and might eventually be used as treatments themselves. The challenge is that they are extremely small, highly varied, and present in low amounts, making them hard to see and count accurately.
Moving Beyond Slow and Fragmented Testing
Traditionally, scientists have relied on several separate methods to study EVs. Ultracentrifugation or size-exclusion chromatography are used to isolate them; nanoparticle tracking analysis measures how they move to estimate size and number; and electron microscopes provide detailed images. While powerful, these approaches are slow, labor-intensive, and not ideal for routine or clinical use. Crucially, they are poor at telling which specific proteins each vesicle carries. Flow cytometry—a technology widely used to analyze blood and immune cells—offers a more streamlined alternative. It sends particles one by one past lasers, measuring their light scattering (related to size) and fluorescence (from dyes or antibodies). Recent instrument designs promise to push flow cytometry down to the scale of the smallest vesicles, raising the possibility that one machine could size, count, and characterize EVs in a single run.
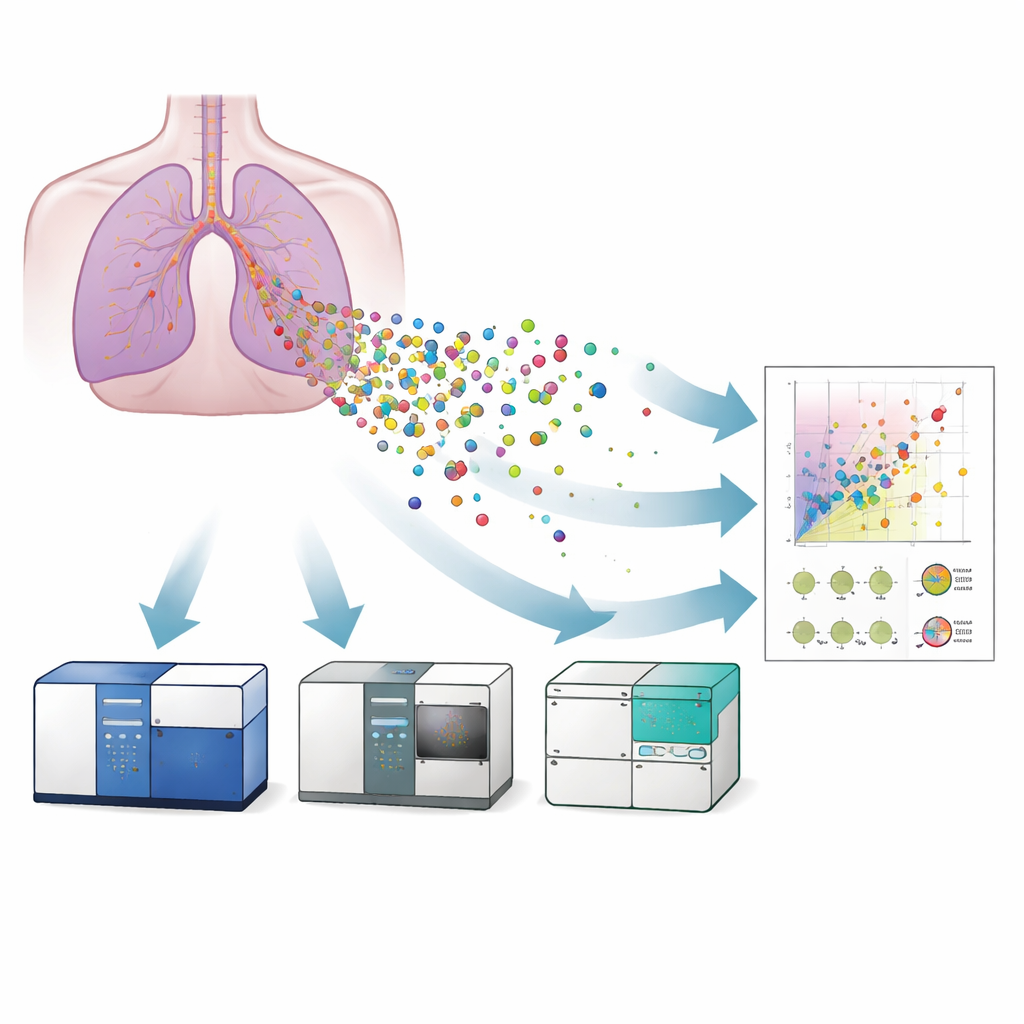
Three Machines, One Goal
The researchers compared three flavors of flow cytometry for studying EVs released from a lab-grown model of human bronchial epithelium: a conventional flow cytometer, an imaging flow cytometer, and a nanoscale flow cytometer. First, they confirmed the presence and typical appearance of vesicles using electron microscopy and super-resolution microscopy, which also showed that the vesicles carried surface proteins known as tetraspanins (CD9, CD63, CD81)—standard markers used to identify EVs. They then used carefully calibrated reference beads and liposome standards to translate each instrument’s light signals into approximate vesicle sizes. The nanoscale flow cytometer could detect vesicles as small as about 40 nanometers, the imaging instrument down to about 55 nanometers, while the conventional machine struggled below roughly 120 nanometers. This meant that only the more sensitive systems could fully capture the smallest, and potentially most medically relevant, vesicles.
Tagging and Counting the Vesicles
To make sure they were measuring intact, biologically active vesicles rather than debris, the team used a dye called calcein-AM. This dye only lights up once it crosses an intact membrane and is processed by enzymes inside the vesicle. They carefully tuned the dye concentration so that it marked vesicles strongly while minimizing background signal, settling on a concentration that produced clear, linear responses across dilutions of samples. Calcein was then combined with fluorescent antibodies that latch onto tetraspanins on the vesicle surface, allowing the instruments to separate vesicles that carried different combinations of these markers. The group also measured how sensitive each machine was to these fluorescent tags—how many antibody molecules had to be present on a vesicle before it could be reliably detected. Imaging flow cytometry generally offered lower detection thresholds than the conventional cytometer, making it better suited for particles with only a small number of marker proteins.
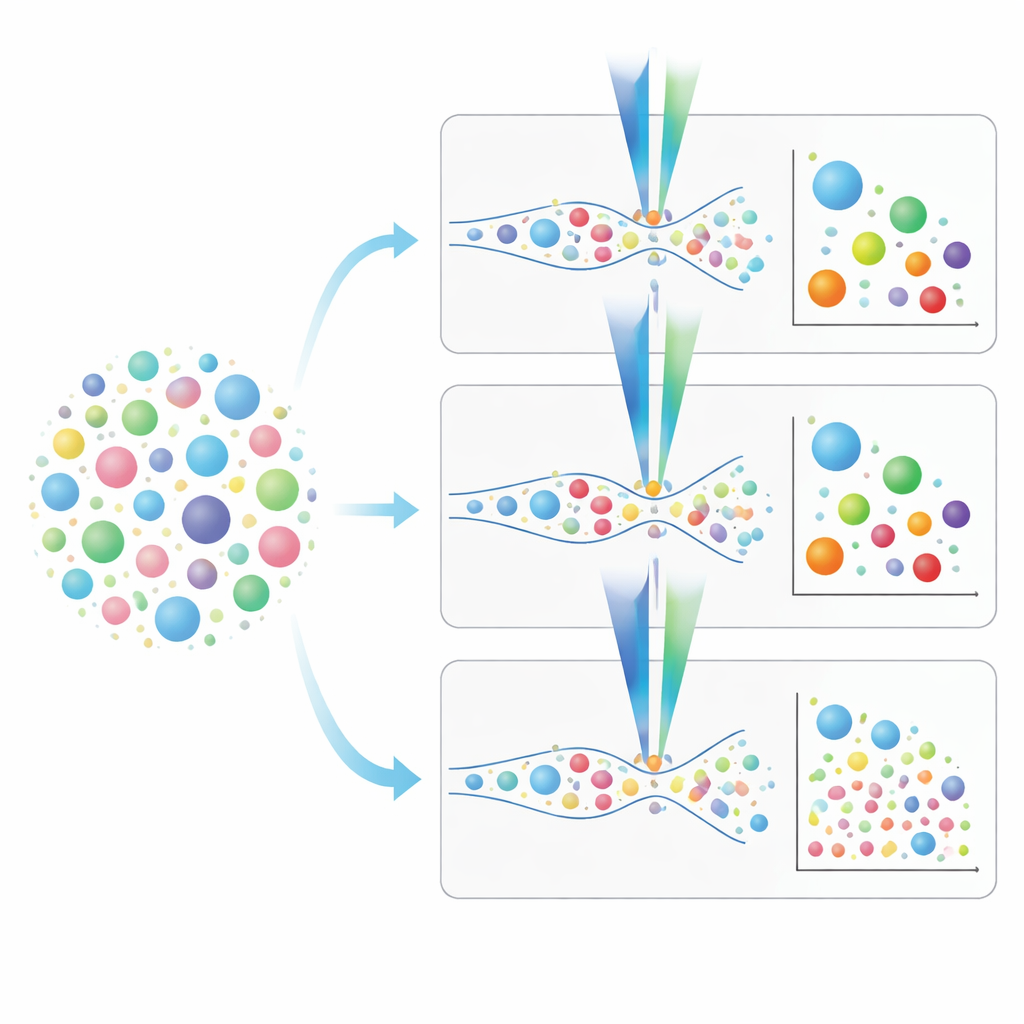
Choosing the Right Tool for Lung Vesicle Studies
From this head-to-head comparison, a practical picture emerges. Conventional flow cytometers are faster and can analyze samples without prior isolation, but they miss many of the smallest vesicles that may carry crucial disease signals. Imaging and nanoscale flow cytometers, while slower and in the nano instrument’s case more reliant on prior sample cleanup, can see down to much smaller sizes and support richer “fingerprinting” of vesicle surface markers. The authors outline step-by-step staining and calibration strategies, emphasize the importance of knowing each fluorescent tag’s detection limit, and show how to avoid misleading signals from dyes that also label fragments or non-vesicle particles. In plain terms, they provide a roadmap for researchers to decide which instrument and protocol best fit their lung EV questions, helping bring the field closer to using these microscopic messengers as practical tools in understanding, diagnosing, and eventually treating respiratory disease.
Citation: Hopkins, G., Browne, W., Tucis, D. et al. Bronchial epithelial cell-derived extracellular vesicle analysis using conventional, imaging, and nanoscale flow cytometry technologies. Sci Rep 16, 11162 (2026). https://doi.org/10.1038/s41598-026-41848-x
Keywords: extracellular vesicles, lung epithelium, flow cytometry, respiratory disease, biomarker analysis