Clear Sky Science · en
Quantitative ex vivo assessment of target temperature and ablation duration for protocol optimization of microwave ablation procedures with mr thermometry
Watching Tumor Treatments in Real Time
When doctors burn away liver tumors with heat, they want to see exactly which parts of the organ are being destroyed while the procedure is happening. Magnetic resonance imaging (MRI) offers a way to “see temperature” inside the body as heat spreads. This study explores how hot, and for how long, doctors can safely heat liver tissue during microwave ablation while still getting clear, reliable temperature maps from MRI—an important step toward more precise, less invasive cancer treatments.
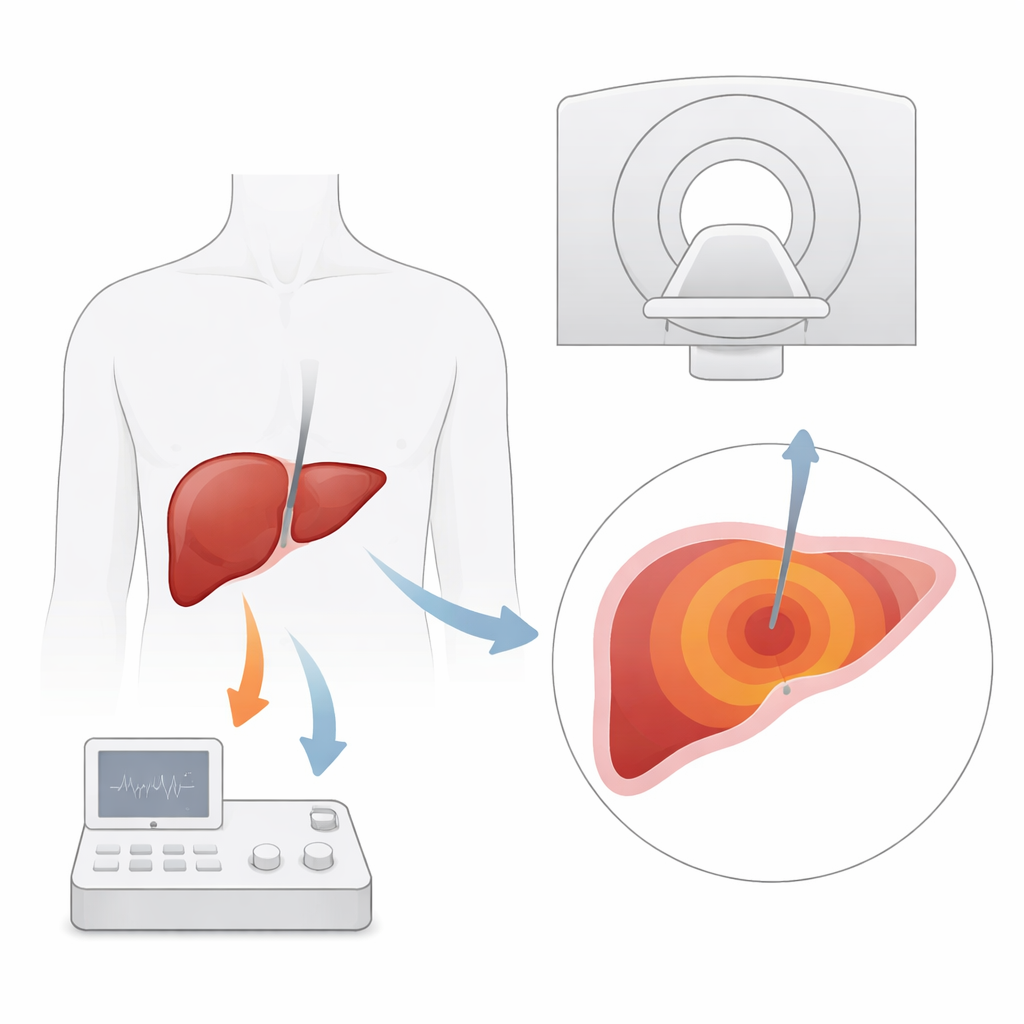
Why Heat-Based Liver Treatments Need Better Eyes
Microwave ablation uses a thin needle to deliver energy that cooks and kills tumor tissue inside the liver. It is especially important for patients with liver cancer or metastases who cannot undergo surgery. Traditional imaging tools such as ultrasound and CT scans guide where to place the needle, but they struggle to show small tumors clearly or to track how far the heat has spread during treatment. MRI stands out because it can not only show soft tissues in high detail, but also measure temperature changes using a property of water molecules. In theory, this allows doctors to watch a heat map of the liver in real time and ensure that the entire tumor, plus a safe margin, is destroyed.
When Too Much Heat Blinds the View
In practice, MRI temperature maps can become unreliable when tissue gets very hot. At high temperatures, water in the tissue boils, forming tiny gas pockets that distort the magnetic field. These distortions show up as false temperature readings and strange shapes on the map, making it hard to know where the true ablation zone ends. The researchers suspected that using slightly lower target temperatures might give cleaner, more trustworthy MRI maps, even if that meant smaller treated areas. Their goal was to find combinations of target temperature and heating time that best balance image clarity with sufficient tissue destruction.
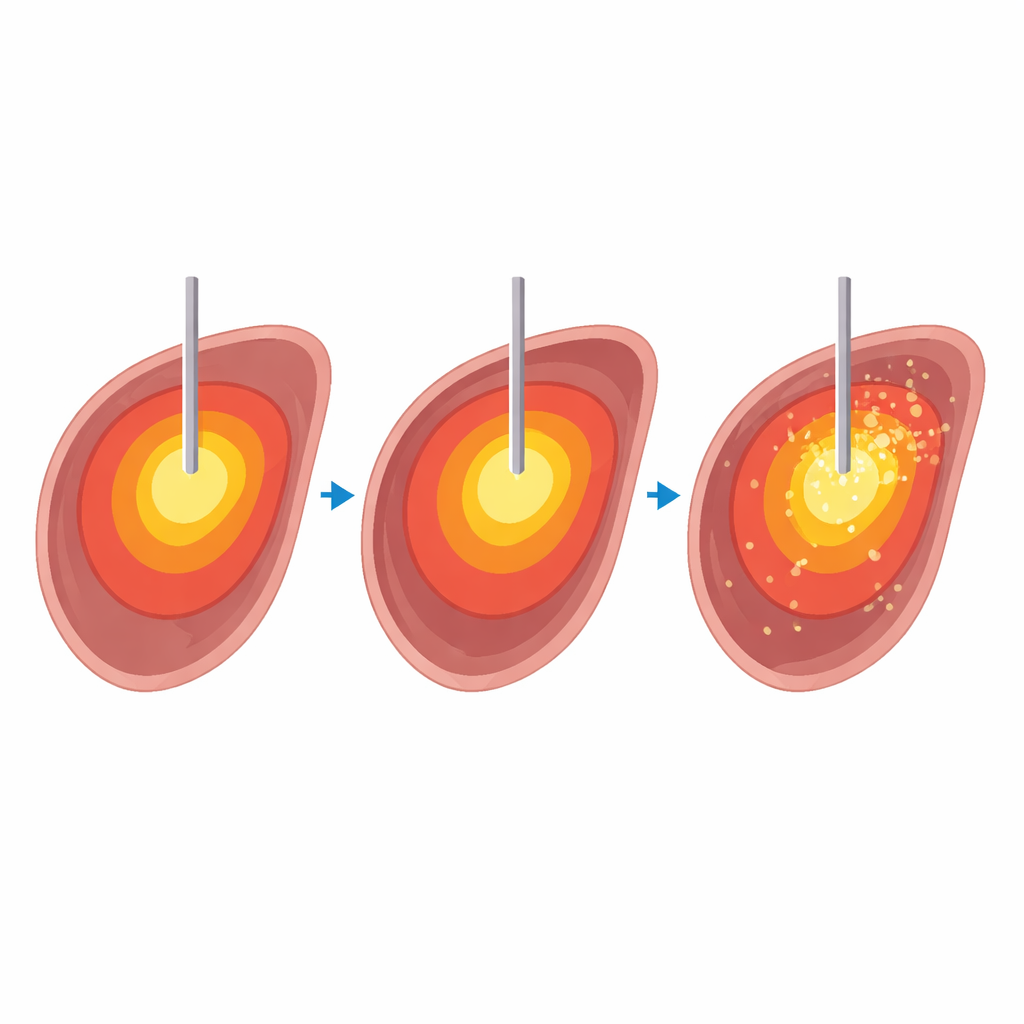
Testing Heat Settings in Donor Animal Livers
To investigate this safely and precisely, the team performed 32 microwave ablations in ten bovine (cow) livers that had been removed shortly after slaughter. Inside a 1.5‑tesla MRI scanner, they heated liver tissue to four target temperatures—60, 80, 100, and 120 degrees Celsius—for four different time periods between 5 and 15 minutes. During each ablation, a rapid MRI sequence produced three‑dimensional temperature and “thermal dose” maps every few seconds. After heating, the livers were sliced along the needle track, and the visible dead tissue areas were measured and compared with the areas predicted by the MRI maps. Two radiologists rated the MRI map quality on a five‑point scale, considering how round and regular the heated zone appeared and how strongly artifacts from gas and the needle interfered.
Cooler Burns, Clearer Maps
The experiments showed a clear trade‑off. Higher temperatures produced larger zones of dead tissue, as one would expect, but the MRI temperature maps deteriorated markedly above 100 degrees. At these hotter settings, the ablation shapes became more irregular, and gas‑related distortions caused apparently falling or even negative temperature values near the needle. In contrast, at 60 and 80 degrees, the MRI maps were smoother, more circular, and much better aligned with the true lesion sizes seen in the tissue slices. Statistical analyses confirmed strong agreement between MRI‑derived areas and actual necrosis at lower temperatures, with correlations weakening or disappearing at higher temperatures. Extending heating time partly compensated for lower temperatures in terms of total damage, but could not fully match the largest lesions seen at 120 degrees.
A Two-Step Strategy for Safer, Smarter Ablation
Based on these findings, the authors propose a practical compromise for future MRI‑guided treatments. A first heating phase at around 80 degrees Celsius for 15 minutes provided a good balance: image quality was high, the ablation zone was reasonably large, and tissue studies showed effective cell death. For cases requiring even bigger treatment zones, they suggest a two‑step approach: start with a lower‑temperature phase to draw a clean thermal map and verify coverage, then, once placement and margins look satisfactory, increase temperature in a second phase to expand the killed area, accepting that the temperature map will be less reliable during this final boost.
What This Means for Future Patient Care
To a non‑specialist, the key message is that “turning the heat down a bit” can actually make heat‑based cancer treatments safer and more precise—at least in controlled lab settings. Lower target temperatures gave much clearer MRI temperature maps, making it easier to see where tissue had truly been destroyed. The downside is that cooler burns produce smaller lesions, so doctors may need longer or multi‑step treatments to cover large tumors. Because this research was done in non‑living animal livers without blood flow or breathing motion, further work in patients is needed. Still, the study points toward treatment protocols that use MRI not just to aim the needle, but to watch and fine‑tune the burn in real time, potentially improving outcomes for people with liver tumors.
Citation: Nardone, L., Tan, A.S.M., Bour, P. et al. Quantitative ex vivo assessment of target temperature and ablation duration for protocol optimization of microwave ablation procedures with mr thermometry. Sci Rep 16, 8153 (2026). https://doi.org/10.1038/s41598-026-41656-3
Keywords: microwave ablation, MRI thermometry, liver cancer, thermal ablation, image-guided therapy