Clear Sky Science · en
Hybrid tuned deep learning model for breast cancer diagnosis using genetic data
Why this matters for patients and families
Breast cancer is now the most commonly diagnosed cancer in women worldwide, and catching it early can mean the difference between life and death. Doctors increasingly have access to a person’s genetic information, but turning tens of thousands of gene measurements into clear answers is extraordinarily difficult. This paper describes a new computer model that reads these complex genetic patterns to spot breast cancer and predict outcomes with striking accuracy, potentially giving clinicians a powerful assistant for earlier and more reliable decisions.
From genes to warning signs
Every breast tumor carries a molecular fingerprint encoded in the activity of thousands of genes. The authors set out to build a system that could read this fingerprint directly, instead of relying only on images or a handful of well-known genes such as BRCA1 and BRCA2. They worked with two of the largest public resources in cancer genomics: the TCGA breast cancer cohort, which includes gene activity for 17,814 genes in 590 samples, and the METABRIC study, which contains genomic and clinical information for more than 1,400 patients. Their goal was ambitious: design a method that can handle this flood of information, find the most telling signals, and still work reliably in completely separate patient groups.
Boiling thousands of genes down to a useful set
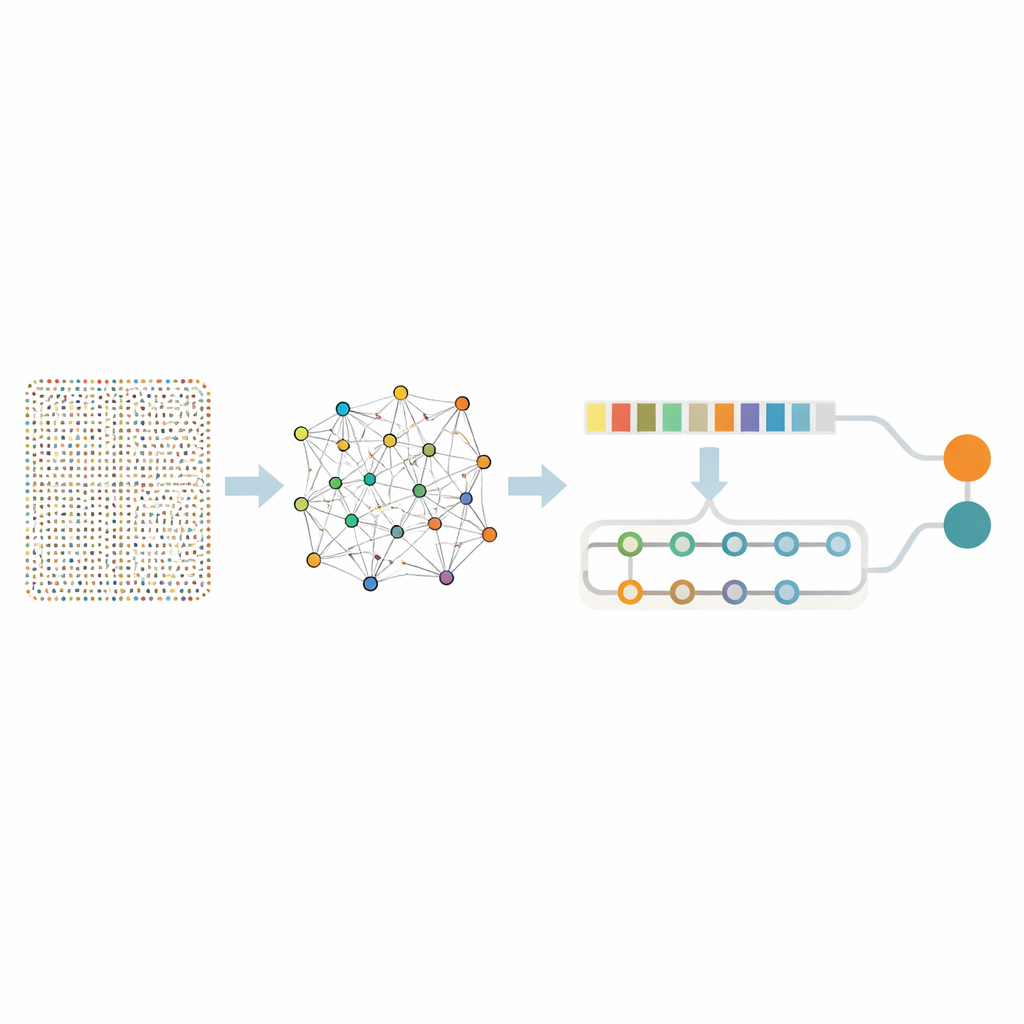
Looking at nearly eighteen thousand genes at once is overwhelming even for advanced algorithms, and it risks picking up meaningless noise. The researchers therefore used a two-step “sieve” to isolate a smaller set of truly informative genes. First, they applied a technique called Random Forest, which effectively asks many decision trees which genes matter most for telling cancerous tissue from healthy samples. This step trimmed the list down to 436 promising genes. Next, they examined how these genes behave together using association rule mining, a method that spots groups of genes that tend to be active at the same time in tumors. This extra layer of analysis identified gene pairs and networks tied to key cancer processes such as rapid cell division, DNA damage repair, and changes to the tissue surrounding the tumor. After this refinement, 332 genes remained—still rich in biological meaning but far more manageable for deeper analysis.
A two-part neural network that learns patterns and context
With this focused gene set in hand, the team built a hybrid deep learning model that combines two types of neural networks. One part, known as a convolutional network, scans along the gene list to pick up local patterns—clusters of genes that tend to rise or fall together. The second part, a bidirectional memory network, looks at the same information while keeping track of long-range relationships, capturing how distant genes influence each other over the whole profile. Before training, the authors balanced the data so that cancer and non-cancer samples were represented fairly and added small amounts of artificial noise, teaching the model not to be fooled by random fluctuations.
How well the system performs in real-world tests
When trained and tested on TCGA data, the hybrid network correctly distinguished tumor from normal samples with about 97% accuracy and an almost perfect ability to separate the two groups. Importantly, it outperformed simpler deep learning setups and standard machine learning tools such as logistic regression and support vector machines, even when those competing methods received the same carefully chosen genes. The strongest test, however, was whether the model would hold up on an entirely different dataset. Applied to METABRIC, which was collected in other hospitals using different laboratory methods, the system maintained high performance: in its best run it achieved 99.3% accuracy and correctly identified every patient who later died from breast cancer, a crucial property if the tool is to be used to flag high-risk cases.
What this could mean for future care
To a non-specialist, the bottom line is that this study delivers a smart filter and reader for genetic data that can spot breast cancer and related risk with remarkable consistency across large patient groups. By combining a thoughtful gene-selection strategy with a two-branch neural network, the authors show that computers can extract clinically meaningful signals from enormous genetic datasets, not just in one study but across independent cohorts. While more work is needed to test the approach in diverse populations and to explain its decisions in detail, the method points toward a future in which a simple blood or tissue sample could feed into such models and help doctors detect tumors earlier and tailor treatment more precisely.
Citation: Hesham, F., Abbassy, M.M. & Abdalla, M. Hybrid tuned deep learning model for breast cancer diagnosis using genetic data. Sci Rep 16, 9664 (2026). https://doi.org/10.1038/s41598-026-41643-8
Keywords: breast cancer genomics, deep learning diagnosis, gene expression biomarkers, early cancer detection, clinical decision support