Clear Sky Science · en
Synthesis, characterization of Mg doped CuFe2O4 nanoparticles for potential anticancer applications
Why tiny magnets could matter for cancer
Cancer treatments like chemotherapy and radiation can damage healthy tissue as well as tumors. Scientists are searching for smarter therapies that hit cancer cells harder than normal cells. This study explores ultra-small magnetic particles made from iron, copper, and magnesium, designed to home in on cancer cells and trigger their self‑destruct programs from the inside. The work suggests that carefully tuning the recipe and size of these particles can make them especially lethal to tumor cells while sparing healthy ones.
Building smart metal particles
The researchers created a family of “nanoferrites” – tiny crystals containing iron – by combining copper and magnesium in different ratios with iron and oxygen. These particles, only about 17–30 billionths of a meter across, are small enough to slip into cells. The team used several advanced microscopes and X‑ray techniques to confirm that the particles had the right crystal structure, were chemically pure, and formed roughly spherical clusters. They found that the mixed metal version with equal amounts of copper and magnesium produced the smallest particles, which increases surface area and tends to boost their chemical reactivity.
Putting the particles to the test in cancer cells
The team then tested how toxic each particle type was to human cancer cells grown in the lab, including prostate (PC‑3), colon (Caco‑2), breast (MCF‑7), and liver (HepG‑2) cancer cells, alongside normal intestinal cells as a safety check. All three formulations harmed cancer cells in a dose‑dependent way, but not equally. The mixed copper‑magnesium particles were the most potent overall, particularly against prostate and colon cancer cells, where they killed half the cells at relatively low doses. Importantly, normal cells tolerated higher doses, suggesting a degree of selectivity that is crucial for any future therapy.
Forcing cancer cells into controlled self‑destruction
To understand how the nanoparticles kill, the researchers looked at apoptosis, the tidy form of cell death in which damaged cells dismantle themselves rather than bursting. Using flow cytometry, they showed that treated prostate and colon cancer cells shifted strongly from a healthy state into early and late stages of apoptosis. Again, the mixed copper‑magnesium particles had the strongest effect, driving total apoptosis to many times that of untreated cells. There was also a modest rise in necrosis, a more chaotic form of cell death, hinting that multiple damage pathways may be engaged.
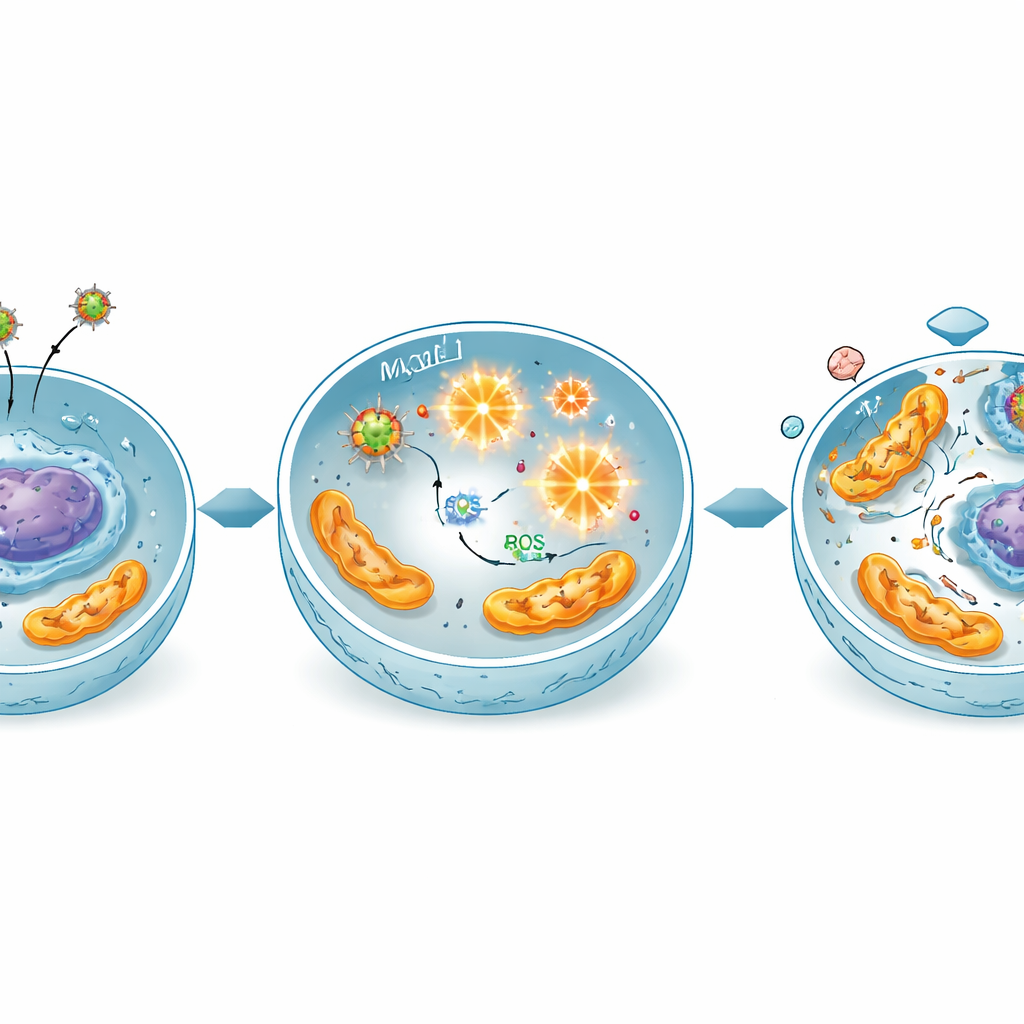
Overloading cancer cells with harmful oxygen chemistry
The study points to a chemical chain reaction at the heart of this effect. These iron‑based particles can act like tiny catalysts, converting natural hydrogen peroxide inside cells into highly reactive oxygen forms. Measurements showed that treated cancer cells produced much more of these reactive oxygen species, especially when exposed to the mixed copper‑magnesium particles. This oxidative surge damages cell components, particularly the energy‑producing mitochondria and DNA. Gene‑expression tests confirmed that key “guardian” and “executioner” genes linked to cell suicide were switched on, while survival and cell‑cycle genes were turned down, matching a picture of stress‑driven, mitochondria‑dependent apoptosis.
What this could mean for future cancer care
Overall, the work shows that by carefully choosing the metals and tuning the size and structure of nanoferrites, it is possible to create particles that strongly push cancer cells toward self‑destruction while being gentler on normal cells. The equal‑parts copper‑magnesium version stood out as the most effective, likely because its small size and mixed metal chemistry enhance its ability to enter cells and generate reactive oxygen. While these results are still limited to cell cultures and far from clinical use, they highlight a promising route toward more precise, nanotechnology‑based cancer treatments that rely on triggering a tumor’s own kill switches rather than flooding the body with broadly toxic drugs.
Citation: Ali, M., Zein, N., Abdo, M.A. et al. Synthesis, characterization of Mg doped CuFe2O4 nanoparticles for potential anticancer applications. Sci Rep 16, 8276 (2026). https://doi.org/10.1038/s41598-026-41540-0
Keywords: nanoparticles, cancer therapy, reactive oxygen species, apoptosis, ferrites