Clear Sky Science · en
Antagonistic effects of amino acids support abiotic nano-environments in clay
How Tiny Pockets in Clay Could Nurture Life’s First Chemistry
Life may have started not in open seas, but inside unimaginably tiny spaces between layers of common clay minerals. This study explores how simple molecules related to today’s amino acids can reshape clay at the nanometer scale, carving out snug compartments that trap and organize chemistry. By showing how different amino acids either strengthen or disrupt clay layers, the work suggests a natural way that early Earth — or other rocky worlds — could have created miniature "laboratories" where the building blocks of life might assemble.
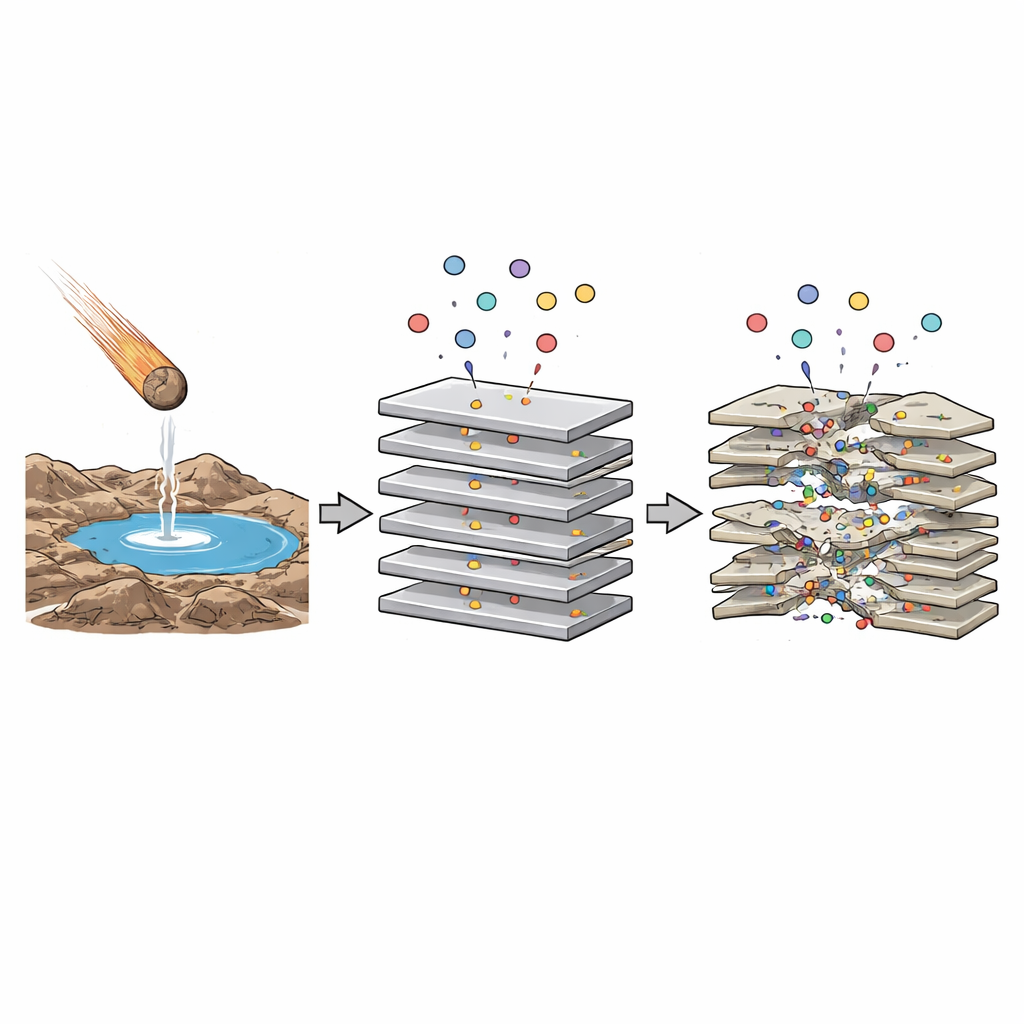
The Promise of Hidden Spaces
Scientists have long suspected that minerals did more than just sit beneath early oceans and ponds: they may have helped assemble the first chains of molecules. In particular, clays like montmorillonite can swell and shrink as water and small molecules slip between their stacked sheets. Inside these narrow gaps, water behaves differently, electric forces are stronger, and reactions that are hard in ordinary water can become easier. Such confined spaces might help link simple building blocks — such as amino acids and nucleotides — into longer chains, a key step toward life. But real prebiotic environments were chemical stews with many different organic molecules present at once. This raises a question: when several kinds of amino acids interact with clay together, do they cancel each other out, or can their competing effects actually create richer environments for chemistry?
Friendly Builders and Mischievous Disrupters
The researchers focused on a calcium-rich form of montmorillonite clay and three amino acids. Two, lysine and arginine, are standard protein-building amino acids familiar from biology textbooks. The third, gamma-aminobutyric acid (GABA), is not used in modern proteins but is common in meteorites and thus likely to have been delivered to early Earth. Previous work showed that lysine and arginine nestle into the spaces between clay layers and bind tightly, helping keep the layers ordered and relatively flat. GABA, by contrast, barely sticks at all—yet it has an outsized effect: it bends and partially peels apart the clay sheets, creating nanometer-scale cavities. This study asked what happens when a small amount of protein-type amino acid is mixed with a large excess of meteorite-like GABA, mimicking a pond that has been heavily seeded with extraterrestrial organics.
Watching Clay Layers Transform
To track these changes, the team combined several techniques that each reveal a different aspect of the clay. Infrared spectroscopy probed how the silicon–oxygen parts of the clay framework vibrate, which shifts when layers are distorted, separated, or filled with different species. X-ray diffraction measured the spacing and ordering of the stacked sheets, both in dry samples and after rehydration with water vapor. Thermogravimetric analysis monitored how strongly organic molecules and water were held in the clay. Finally, high-resolution electron microscopy provided direct images of the layer structures and any tiny cavities between them. Together, these tools allowed the authors to separate the stabilizing actions of lysine and arginine from the disruptive influence of GABA, even when all were present in the same mixture.
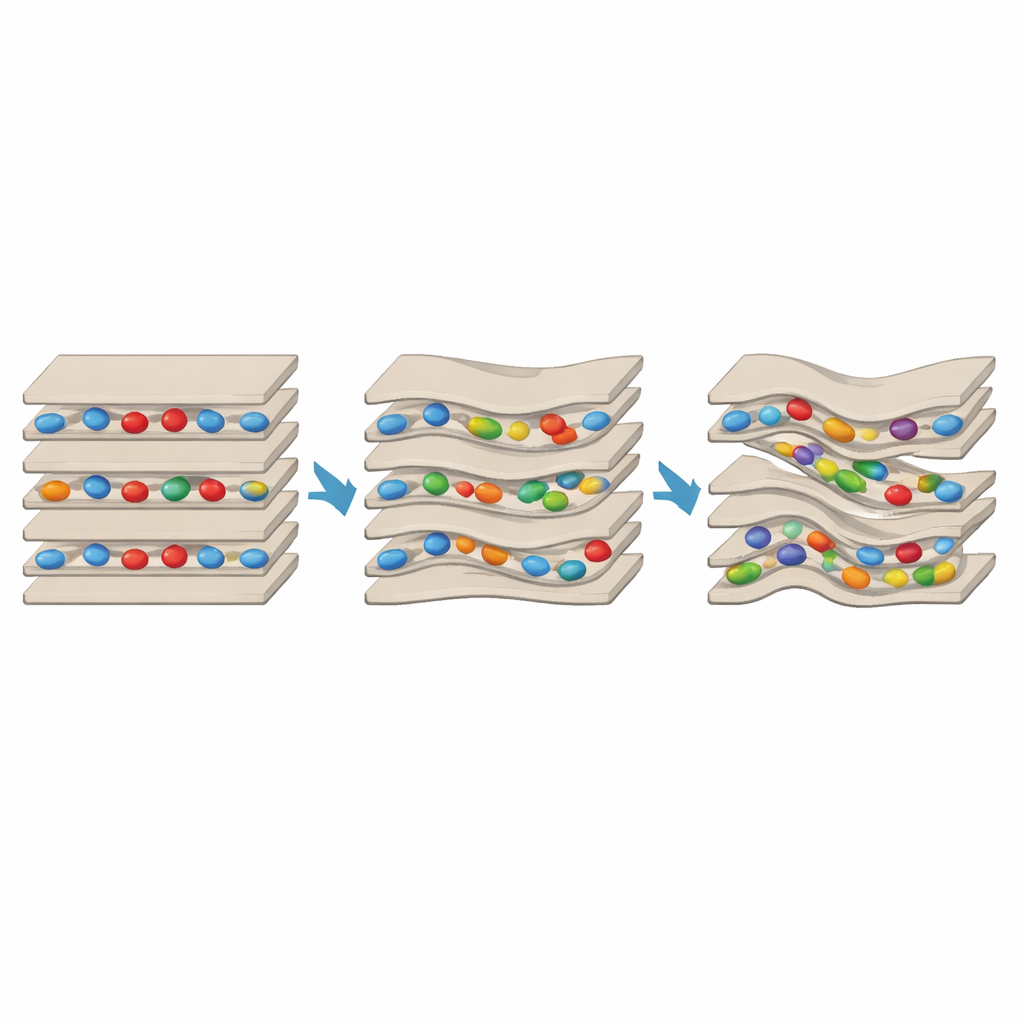
A Nano-Scale Yin–Yang in Clay
The results reveal a striking push-and-pull. When only lysine or arginine were present, they wedged into the interlayer spaces, acting like crossbars that tie one sheet to the next. This reduced the clay’s ability to swell with water and made the structure more ordered and resistant to peeling apart. But when GABA was added in large excess, it overcame this stabilizing effect in many regions. Spectroscopic signatures showed the presence of partially exfoliated domains, and X-ray patterns indicated more disorder and reduced layer alignment. Electron microscopy went further, revealing highly distorted stacks with clear nanocavities embedded mid-layer — small pockets a couple of nanometers across that did not appear in the control or in protein-only samples. Importantly, regions with bridged, tightly held layers coexisted alongside regions with distorted, cavity-rich layers in the same clay grain, demonstrating that both effects can operate side by side.
Natural Nano-Compartments for Early Chemistry
For a non-specialist, the key message is that simple mixtures of amino acids can sculpt clay into a patchwork of tiny compartments with different properties. Protein-building amino acids help hold layers together, while meteorite-derived GABA subtly pries them apart and carves out nanocavities. These confined pockets likely host water and dissolved molecules under conditions distinct from the surrounding environment, potentially favoring reactions such as polymer formation that are central to life’s origin. Because meteorites deliver abundant non-protein amino acids to worlds rich in clay, this antagonistic “yin–yang” between stabilizing and disrupting molecules may be a common way for rocky planets and asteroids to generate and sustain diverse nano-environments where primitive biochemistry could begin.
Citation: Bezaly, O.R., King, H.E. & Petrignani, A. Antagonistic effects of amino acids support abiotic nano-environments in clay. Sci Rep 16, 8959 (2026). https://doi.org/10.1038/s41598-026-41502-6
Keywords: origins of life, prebiotic chemistry, clay minerals, amino acids, nanoconfinement