Clear Sky Science · en
Extraction method shapes soil water-soluble organic matter composition as revealed by absorbance, fluorescence, and parallel factor analysis (PARAFAC)
Why the hidden life in soil water matters
Every time it rains, tiny carbon-rich molecules are washed out of soils and carried toward streams, rivers, and eventually the ocean. These invisible ingredients—forms of dissolved organic matter—help feed microbes, bind pollutants, and control how much carbon stays locked in the ground versus returning to the atmosphere. This study asks a deceptively simple question with big implications: when scientists pull that material out of soil in the lab, does the way they do it change what they see? The answer turns out to be yes—and that choice shapes how we understand the carbon cycle linking land and water.
Two ways to rinse the soil
The researchers focused on the portion of soil organic matter that can dissolve in water, known as water-soluble organic matter. This fraction is small in quantity but plays an outsized role in moving carbon and nutrients through landscapes. To compare methods, they took 217 soil samples from 83 cores spread across four contrasting regions in Germany and Austria, from alpine slopes to forested hills. In the lab, each sample was split in two and rinsed either with pure distilled water or with a salty solution of potassium sulfate. Because both portions came from the same soil, any differences in the extracted material could be traced back to the chemistry of the rinse, not to natural variation in the field.
Reading the light from dissolved carbon
Instead of trying to identify every molecule, the team used how the extracts interact with light as a sensitive fingerprint. They measured how strongly the solutions absorbed ultraviolet light and how they glowed when excited with different colors—techniques called absorbance and fluorescence spectroscopy. With a statistical approach known as parallel factor analysis, they separated the complex glow into nine recurring components. Some of these behaved like “humic-like” substances—dark, older, more processed material that tends to be less available to microbes. Others looked “protein-like,” a sign of fresher, microbially derived organic matter. Simple indices derived from the light signals helped indicate how aromatic, humified, or biologically fresh each extract was.
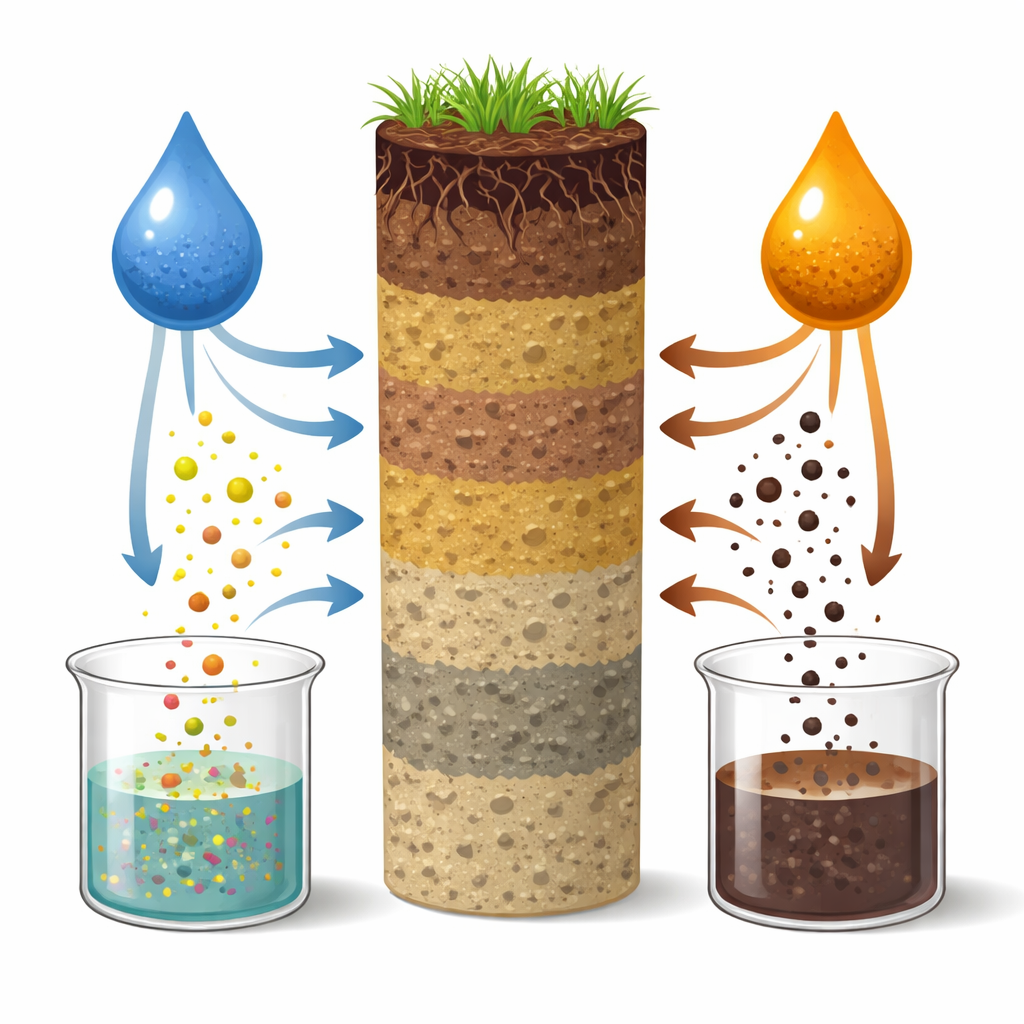
What water sees versus what salt sees
The way the soil was rinsed made a clear difference. The salt solution consistently pulled out more total dissolved carbon, but that carbon was dominated by the humic-like signal that points to older, mineral-bound material. The water extracts, in contrast, contained less carbon overall but showed much stronger protein-like and microbially linked signals, higher indicators of recent biological activity, and more light-absorbing compounds. They also varied much more from sample to sample. This suggests that pure water is especially sensitive to the freshest, most reactive soil carbon—the part that responds quickly to changes in plants, microbes, and weather—while the salty solution emphasizes more stable stores that change slowly over time.
Shifting patterns with soil depth
Because samples came from multiple depths down to two meters, the team could also ask how these light-based fingerprints change with depth. Both extraction methods showed that dissolved carbon generally declined as soils became deeper and older, though alpine sites with thin soils were an exception. Water-based extracts revealed the clearest transitions: surface layers were richer in humic-like material linked to plant litter, while deeper layers gradually shifted toward more protein-like, microbially derived compounds, reflecting the growing importance of microbial processing with depth. The salt-based method showed similar tendencies but with weaker contrasts and less variability, consistent with its focus on a more uniform, mineral-associated pool.
Why method choice changes the story
For scientists and environmental managers, these findings highlight that how you rinse a soil sample is not a minor technical detail but a lens that emphasizes different parts of the carbon story. Using distilled water gives a sharper view of the short-lived, microbe-friendly carbon that is most likely to be mobilized during rain and to fuel life in streams and rivers. Using a potassium sulfate solution recovers more carbon overall and highlights longer-lived, less reactive stores that underpin long-term soil carbon reservoirs. To draw reliable conclusions about how soils feed carbon to aquatic systems, or how they respond to climate and land-use change, future studies will need to match their extraction method to their scientific question—and be cautious when comparing results obtained with different approaches.
Citation: Fasching, C., Boodoo, K.S., Feld-Golinski, A. et al. Extraction method shapes soil water-soluble organic matter composition as revealed by absorbance, fluorescence, and parallel factor analysis (PARAFAC). Sci Rep 16, 8488 (2026). https://doi.org/10.1038/s41598-026-41455-w
Keywords: soil organic matter, dissolved organic carbon, water-soluble organic matter, fluorescence spectroscopy, carbon cycling