Clear Sky Science · en
Circulating immune cell phenotypes are associated with inflammatory biomarkers in dementia-free participants from the Framingham Heart Study Offspring cohort
Why Your Immune System Matters for Brain Health
As we grow older, our bodies often carry a low, constant level of inflammation that can quietly raise the risk of heart disease, diabetes, and memory problems. Scientists know that immune cells in the blood help drive these inflammatory signals, but most studies have looked at only a few cell types or a handful of blood markers at a time. This paper uses detailed blood measurements from hundreds of dementia-free adults in the long-running Framingham Heart Study to map how different kinds of immune cells relate to dozens of inflammation-related proteins. The work helps reveal what an “inflamed” immune system looks like in everyday people and how this may tie into aging and future brain health. 
Taking a Close Look at Blood and the Immune System
The researchers drew on data from 873 men and women, with an average age of 61, who were part of the Framingham Offspring cohort and did not have dementia. They analyzed frozen blood cells and plasma that had been collected around the year 2000. Using a lab method called flow cytometry, they sorted blood immune cells into 77 specific types, including many flavors of T cells, B cells, and monocytes that play different roles in defense and repair. In parallel, they measured 68 proteins related to inflammation, such as signaling molecules and growth factors, using a high-throughput protein panel. This gave them a detailed snapshot of both the “cell side” and the “protein side” of the immune system in the same individuals.
Linking Cell Types to Inflammatory Signals
With this rich dataset, the team examined how each immune cell type related to each protein, while accounting for factors like age, sex, and infection with cytomegalovirus, a common lifelong virus that reshapes the immune system. They also used statistical models that considered family relationships within the cohort and corrected for the large number of comparisons. One of the clearest patterns involved a group of cells called CD8 naïve T cells, which are “fresh” killer T cells that have not yet been heavily engaged in fighting infections. People with fewer of these naïve cells tended to have higher levels of many inflammatory proteins, including several chemokines and classic inflammation drivers. In other words, a blood profile showing fewer naïve CD8 T cells went along with a more activated inflammatory environment.
Signals of Immune Activation and Aging
Another key finding centered on a protein called the soluble CD8 alpha chain, which reflects activity of CD8-positive killer T cells. Higher levels of this protein were linked to greater numbers of cytotoxic and effector CD8 T cells and to a subset of T cells that produce the immune messenger interferon-gamma, all pointing to an immune system that has shifted toward experienced, battle-tested cells. At the same time, people with more naïve-like B cells tended to have lower levels of this CD8-related protein, hinting at a trade-off between maintaining youthful cell reserves and having a more “on-call” response. The researchers also saw meaningful ties between certain B cell and monocyte subtypes and proteins involved in cell migration, tissue repair, and inflammatory damage. These association patterns were especially strong in participants older than 60 and in men, and many persisted even after adjusting for heart disease, kidney function, and traditional cardiovascular risk factors. 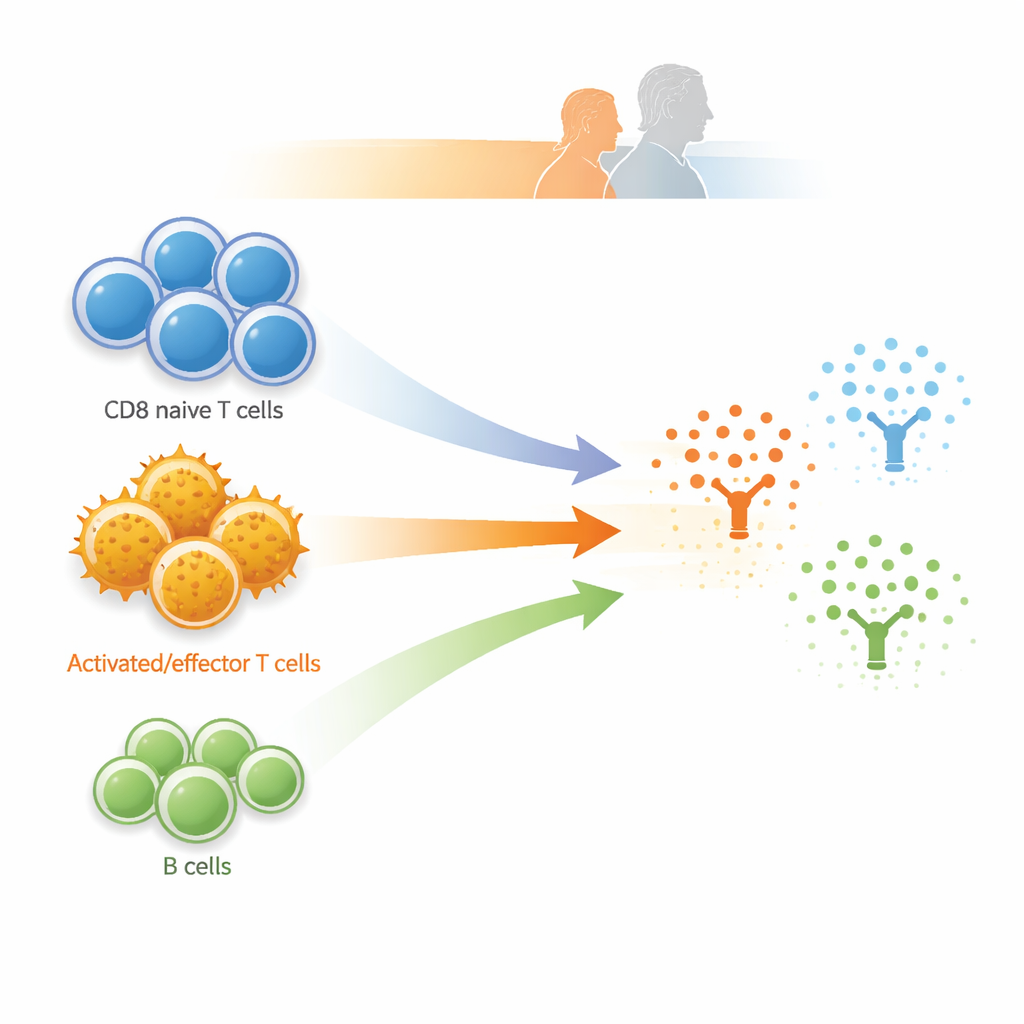
Seeing the Immune Network as a Whole
To move beyond one-pair-at-a-time comparisons, the team built a network map that highlighted which cells and proteins tended to move together. In this network, the CD8-related protein sat at the center of a hub of T cell features, while naïve CD8 T cells connected strongly to several inflammatory chemokines, and specific B cell groups were linked to proteins that help guide their movement or maturation. This integrated view showed that the pairwise results were not random: instead, they formed coherent clusters consistent with known biology, such as the gradual shift from naïve to memory and effector T cells, and the movement of memory B cells into inflamed tissues. The pattern as a whole fit with the idea of “inflammaging,” in which chronic, low-level activation of the immune system becomes more common with age and differs between men and women.
What This Means for Aging Brains
Although this study cannot prove cause and effect or reveal exactly what happens over time, it provides one of the most detailed population-level maps to date of how circulating immune cells and inflammatory proteins relate to each other in older, dementia-free adults. The authors note that several of the proteins most tightly linked to immune cell patterns in this work were also tied to thinking and dementia risk in their earlier research. Together, these findings suggest that shifts in the balance between naïve and more experienced immune cells, and the inflammatory signals they accompany, may be part of the chain linking aging, chronic disease, and later-life brain problems. Following these immune–inflammation patterns over time, and in more diverse populations, may help identify early warning signs and future targets for keeping both the body and brain healthier as we age.
Citation: Chen, J., Doyle, M.F., Cao, Y. et al. Circulating immune cell phenotypes are associated with inflammatory biomarkers in dementia-free participants from the Framingham Heart Study Offspring cohort. Sci Rep 16, 11313 (2026). https://doi.org/10.1038/s41598-026-41423-4
Keywords: immune aging, chronic inflammation, T cells and B cells, blood biomarkers, cognitive decline