Clear Sky Science · en
DUSP29 does not regulate melanoma–myoblast interactions in a skeletal muscle co-culture model
Why muscle’s resistance to cancer spread matters
When cancer spreads through the body, it often lodges in places like the lungs, liver, or bones. Curiously, our skeletal muscles—together making up about half of our body weight—are rarely home to these dangerous deposits. This mystery has intrigued doctors and scientists for more than a century. The study summarized here asks a focused question within that bigger puzzle: do muscle cells themselves, and a muscle‑enriched protein called DUSP29, directly restrain cancer cells, or is muscle’s protection due to something else entirely?
A rare landing place for wandering cancer cells
Doctors see far fewer metastases in skeletal muscle than in many other organs, even in people whose cancer has spread widely. One idea is that muscle cells create an unfriendly neighborhood for incoming tumor cells, perhaps by sending out chemical signals or forcing cancer cells to change their behavior. Earlier work in mice hinted that when skin cancer cells (melanoma) grow next to immature muscle cells, they lose some of their pigment and even start to resemble muscle. Building on this, the authors turned to a specific protein, DUSP29, which is found at much higher levels in muscle than elsewhere and is part of a family of enzymes known to control growth and survival pathways that cancers frequently hijack.
Testing muscle–tumor encounters in a dish
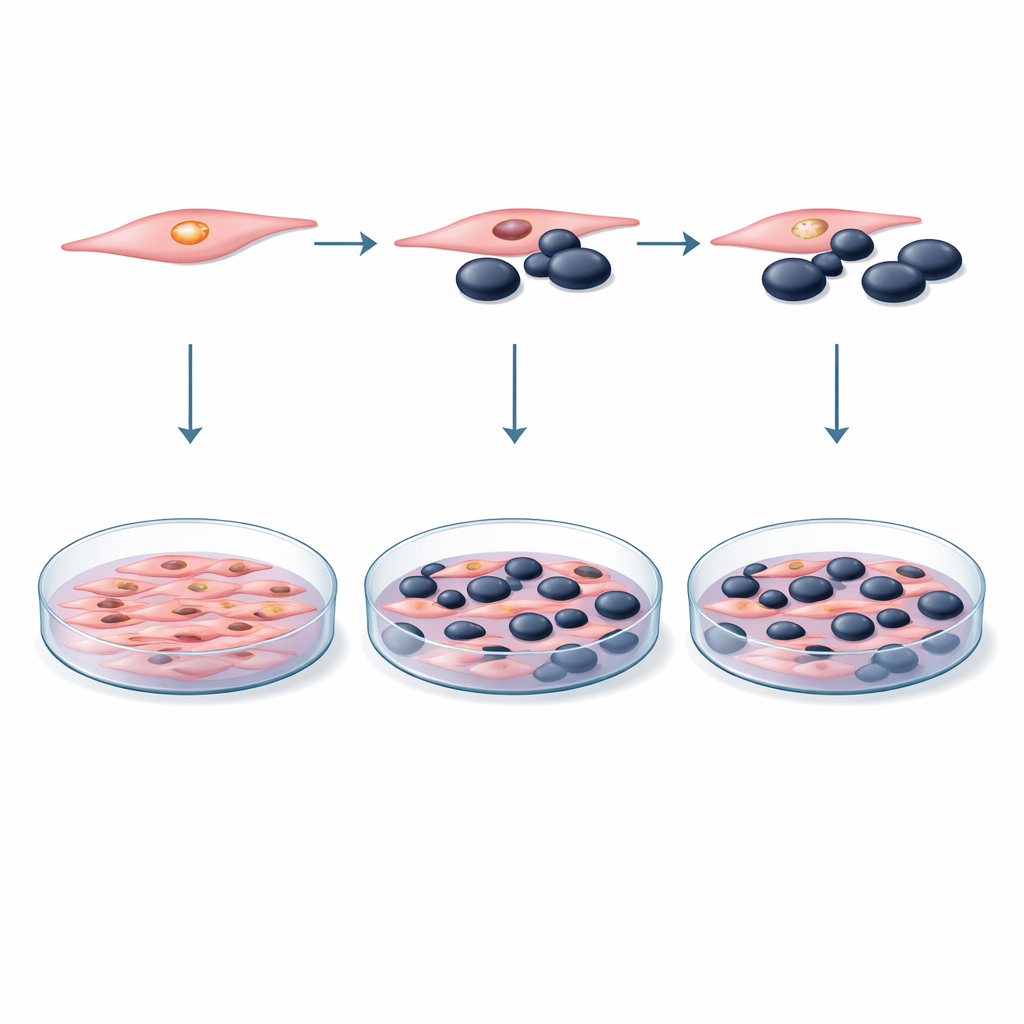
To probe these ideas, the researchers built a controlled laboratory model using two well‑studied mouse cell lines: melanoma cells (B16F10) and muscle precursor cells, or myoblasts (C2C12). They mixed these two cell types in shared dishes at different ratios, from equal numbers to situations where myoblasts vastly outnumbered tumor cells. In some experiments the cells touched directly; in others, melanoma cells were bathed only in the liquid growth medium taken from myoblast cultures, capturing any soluble factors the muscle cells released. Over several days, the team measured how many cells were alive and dividing using a standard color‑changing test, and they checked for signs of programmed cell death using flow cytometry, a technique that can distinguish living, dying, and dead cells.
Putting DUSP29 under the microscope
The second arm of the study focused on DUSP29 itself. First, the scientists confirmed that myoblasts produce this protein’s genetic message at appreciable levels. They then used small interfering RNA (siRNA)—short genetic snippets that act like targeted “off switches”—to sharply reduce DUSP29 levels in the muscle cells. Careful controls showed that the delivery chemicals and control siRNAs did not harm the cells on their own. With DUSP29 dialed down, the modified myoblasts were again grown together with melanoma cells, or their growth medium was applied to melanoma cultures. The same tests for cell survival and death were repeated across multiple time points to see whether silencing this muscle‑enriched protein would tip the balance in favor of or against the tumor cells.
When nothing happening is the important result
Across all of these experiments, the outcome was striking in its absence of drama. Whether melanoma cells were grown alone or alongside myoblasts, with or without direct contact, their survival remained essentially unchanged within normal experimental variation. Co‑culture did not trigger extra cell death in melanoma cells, nor did it boost their growth. Knocking down DUSP29 in the myoblasts likewise failed to alter melanoma cell viability, even though the gene’s activity in muscle cells was strongly reduced. In other words, under these short‑term, simplified conditions, neither myoblast neighbors nor the presence or absence of DUSP29 measurably steered melanoma cells toward survival or destruction.
What this means for the muscle–cancer mystery
The findings suggest that the relative protection of skeletal muscle from metastasis is unlikely to come from a simple direct conversation between melanoma cells and nearby myoblasts, or from DUSP29 acting alone in this early interaction. Instead, muscle’s resistance probably arises from a more complex blend of features—its dense structure, the mechanical forces of contraction, unique metabolic chemistry, and a host of other signaling molecules not captured in this dish‑based system. By clearly showing where a plausible mechanism does not operate, this work helps narrow the search for how muscle keeps most metastatic cancer at bay and points future studies toward more realistic tissue models, different tumor types, and the broader environment in which muscle and cancer cells meet.
Citation: Ön, S., İlhan, H.A., Günenç, D. et al. DUSP29 does not regulate melanoma–myoblast interactions in a skeletal muscle co-culture model. Sci Rep 16, 8372 (2026). https://doi.org/10.1038/s41598-026-41300-0
Keywords: skeletal muscle metastasis, melanoma, tumor–muscle interactions, DUSP29, tumor microenvironment