Clear Sky Science · en
Emergence of a novel reassorted high pathogenicity avian influenza A(H5N2) virus associated with severe pneumonia in a young adult
Why this new bird flu case matters
Most of the time, bird flu is a problem for farmers and wildlife experts, not everyday people. This article describes the first known human infection with a new kind of bird flu virus called H5N2 that can cause serious disease in birds and, in this case, severe pneumonia in a young woman in Mexico. Understanding how this virus appeared, how it affected the patient, and how it might spread helps doctors and public health officials spot future threats before they become wider outbreaks.
A rare and serious illness in one person
The story begins in late 2025, when a young adult woman arrived at a respiratory hospital in Mexico City after more than a week of fever, sore throat, cough, shortness of breath, and coughing up blood. She had obesity but no major chronic diseases and no confirmed close contact with poultry, pigeons, or other animals. On admission, her oxygen levels were dangerously low, and chest scans showed cloudy, patchy areas and signs of bleeding in both lungs, consistent with severe pneumonia that was damaging the tiny air sacs responsible for breathing.
Tracking down an unusual virus
Initial lab tests showed that the patient was infected with influenza A, the broad family that includes both seasonal and avian flu. However, standard tests could not match it to any known seasonal subtype, raising concern that this might be an unusual strain. More specialized testing on fluid taken from deep inside the lungs confirmed that it belonged to the H5 family of avian viruses. The patient was placed under infection-control precautions and treated with the antiviral drug oseltamivir and supplemental oxygen. Over the following days she steadily improved and was eventually discharged without complications, while health authorities began investigating the source of her infection.
How different bird flus fused into something new
To understand exactly what they were dealing with, scientists sequenced the complete genetic material of the virus from the patient. Influenza viruses carry their genes in eight separate pieces, which can be swapped when two different viruses infect the same bird or animal. The analysis showed that this patient’s virus was a genetic patchwork: some segments matched highly pathogenic H5N1 viruses that have been circulating widely in wild birds and causing large outbreaks in poultry across North America, while other segments matched a long‑standing, lower‑grade H5N2 virus that has been present in Mexican poultry for decades. This mixing process, called reassortment, had produced a new version of H5N2 with features of both parent viruses.
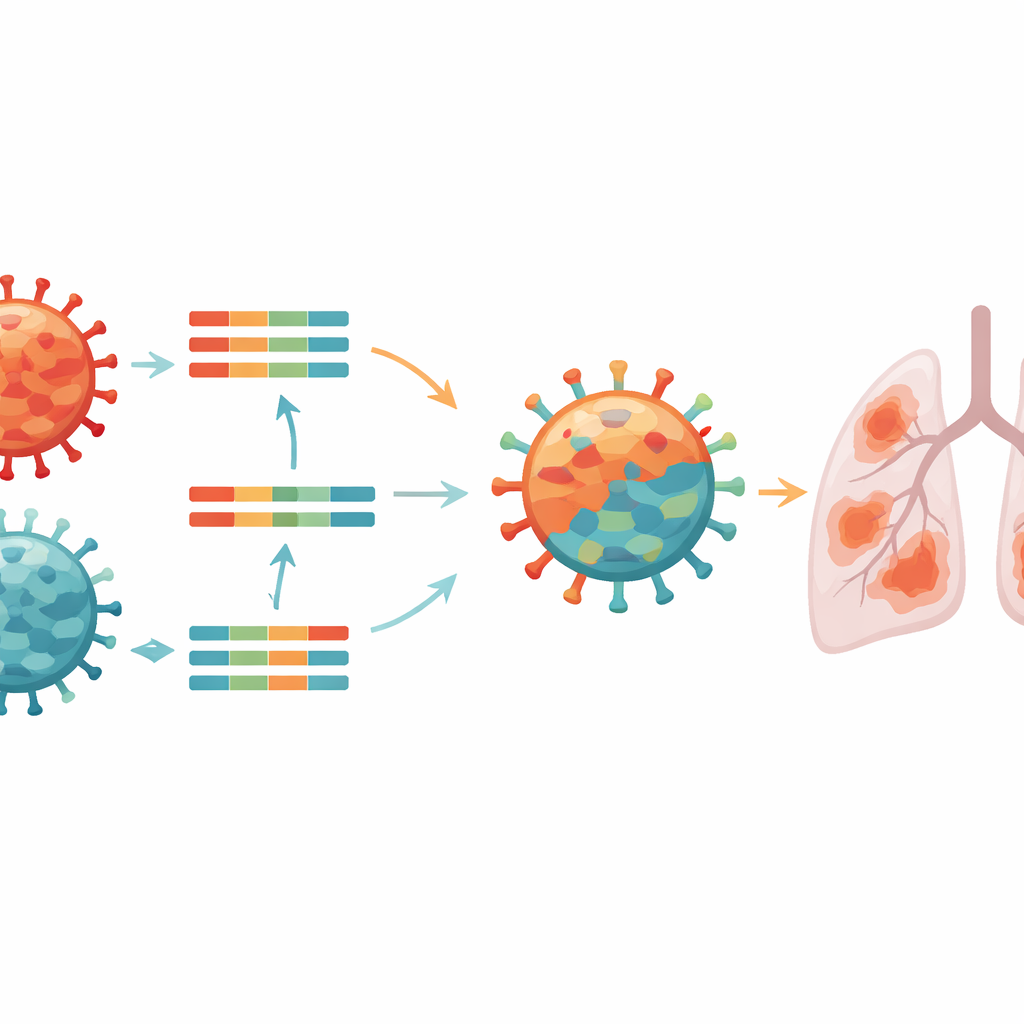
Clues from genes about risk to humans and animals
By comparing the virus’s genes to thousands of other bird flu samples from North America and Mexico, the team could place it on detailed family trees and assign it to a previously unseen genetic pattern they provisionally named B3.14. They also looked for specific changes that might make the virus better at infecting mammals or resisting drugs. The virus still carried a molecular signature typical of highly lethal bird strains and lacked changes known to block common antiviral medicines. However, it did harbor several alterations linked in other studies to better growth in mammalian cells and to shifts in how the virus latches onto receptors in the airways, raising concern that it may be somewhat better suited to infecting humans or other mammals than earlier relatives.
What this single case tells us about future threats
Despite the severity of the woman’s illness, follow‑up testing of more than 40 close human contacts found no additional infections, and there was no sign of person‑to‑person spread. Samples from a chicken, two pigeons, and a dog at the patient’s home did harbor related H5 viruses, underscoring that the likely source was ongoing circulation of mixed bird flu strains in local animals rather than spread between people. The authors conclude that this is the first documented human infection with a highly pathogenic H5N2 virus of this new genetic makeup and a warning sign of how quickly bird and animal viruses can shuffle their genes and jump species. They argue that continued genetic surveillance in wild birds, poultry, and mammals—along with rapid diagnosis and treatment in humans—is essential to catch such dangerous newcomers early and to guide vaccines and antiviral strategies before they have a chance to spread more widely.
Citation: Vázquez-Pérez, J.A., Becerril-Vargas, E., Ramírez‐González, J.E. et al. Emergence of a novel reassorted high pathogenicity avian influenza A(H5N2) virus associated with severe pneumonia in a young adult. Sci Rep 16, 8841 (2026). https://doi.org/10.1038/s41598-026-41122-0
Keywords: avian influenza, H5N2, reassortant virus, pneumonia, zoonotic infection