Clear Sky Science · en
The role of NLRP3 neuroinflammation in cognitive frailty diversity during aging and after LPS administration in mice
Why aging brains and bodies don’t all slow down the same way
As people grow older, some remain sharp and active while others become both physically weak and mentally slowed. Doctors call this combined decline “cognitive frailty,” and it greatly increases the risk of disability and dementia. This study uses mice to ask two key questions: why does cognitive frailty vary so much between individuals, and how much of it can be blamed on smoldering inflammation inside the brain? By teasing apart natural aging from inflammation triggered by an immune challenge, the work explores whether short bouts of illness really mirror what happens during long-term aging in the brain.
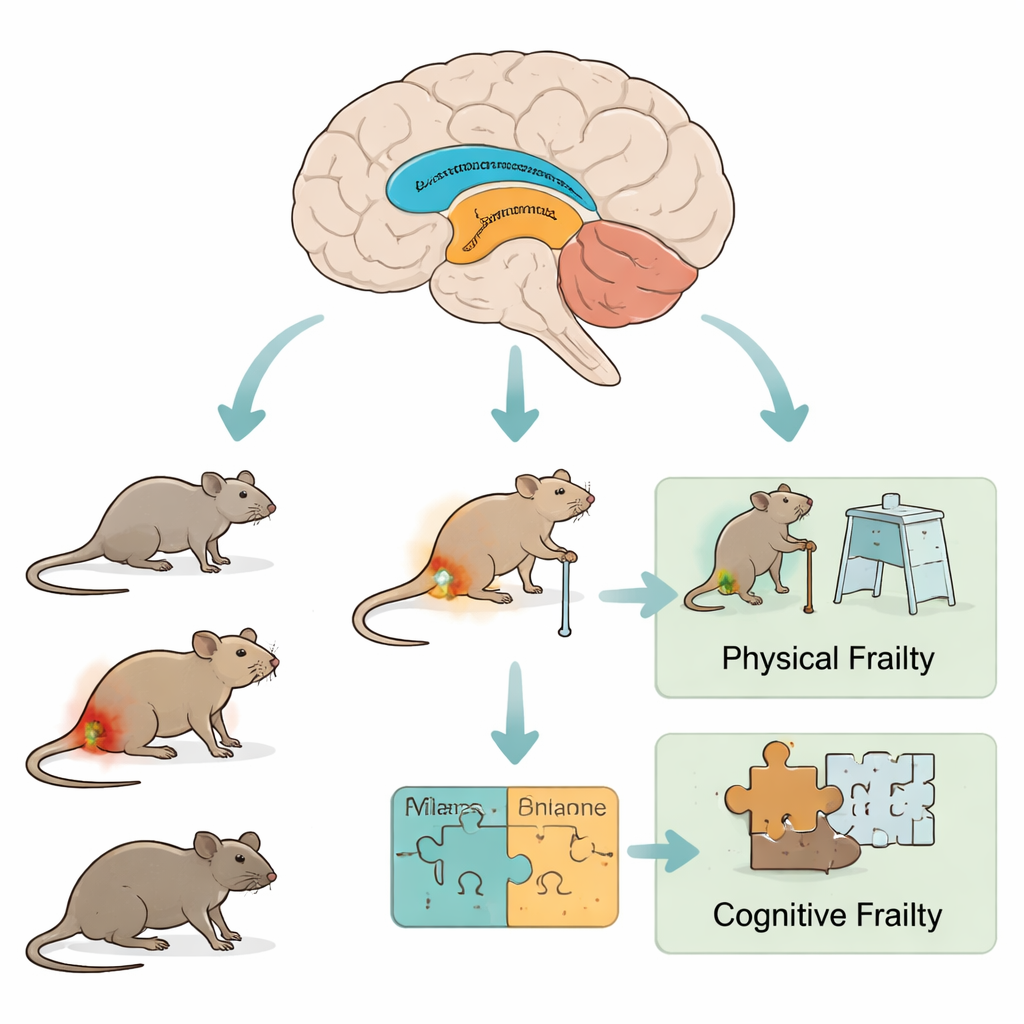
Different paths to growing old in the lab
The researchers compared three groups of male mice: healthy adults, naturally aged animals, and adults given a single injection of bacterial material (LPS) that briefly stirs up the immune system. Over five days they measured body weight, grip strength, movement, balance on a rotating rod, exploration in open spaces, and the ability to recognize a new object. These varied tests let the team score both physical robustness and mild thinking problems. At the end of testing, they examined the hippocampus and amygdala—two brain regions important for memory and emotion—for signs of inflammation and aging-related cell damage.
New ways to measure “body-and-mind” frailty
To move beyond simple yes-or-no labels, the authors combined many test results into several numerical scores. Standard frailty indices captured physical issues such as weight change, slowness, weak grip, and reduced activity. On top of this, they created a Cognitive Frailty Index (CogFI) that blends physical scores with measures of learning, memory, and exploratory drive. This new index was able to reveal how much animals differed from one another, especially among the older mice, where some performed almost like young adults while others showed clear combined decline.
What short-term illness can—and cannot—mimic
Adult mice exposed to LPS quickly lost weight, became weaker, moved less, and explored their surroundings reluctantly. In many of the standard frailty scores, they looked at least as impaired as the older animals. Yet these changes were transient: movement and motor learning began to recover within a few days. Importantly, LPS did not increase markers of cellular senescence in the hippocampus, nor did it cause lasting elevation of a key inflammatory sensor called NLRP3. Natural aging, in contrast, was linked to more senescent cells and a clear rise in NLRP3 levels in the hippocampus, even though some basic physical measures were still relatively preserved.
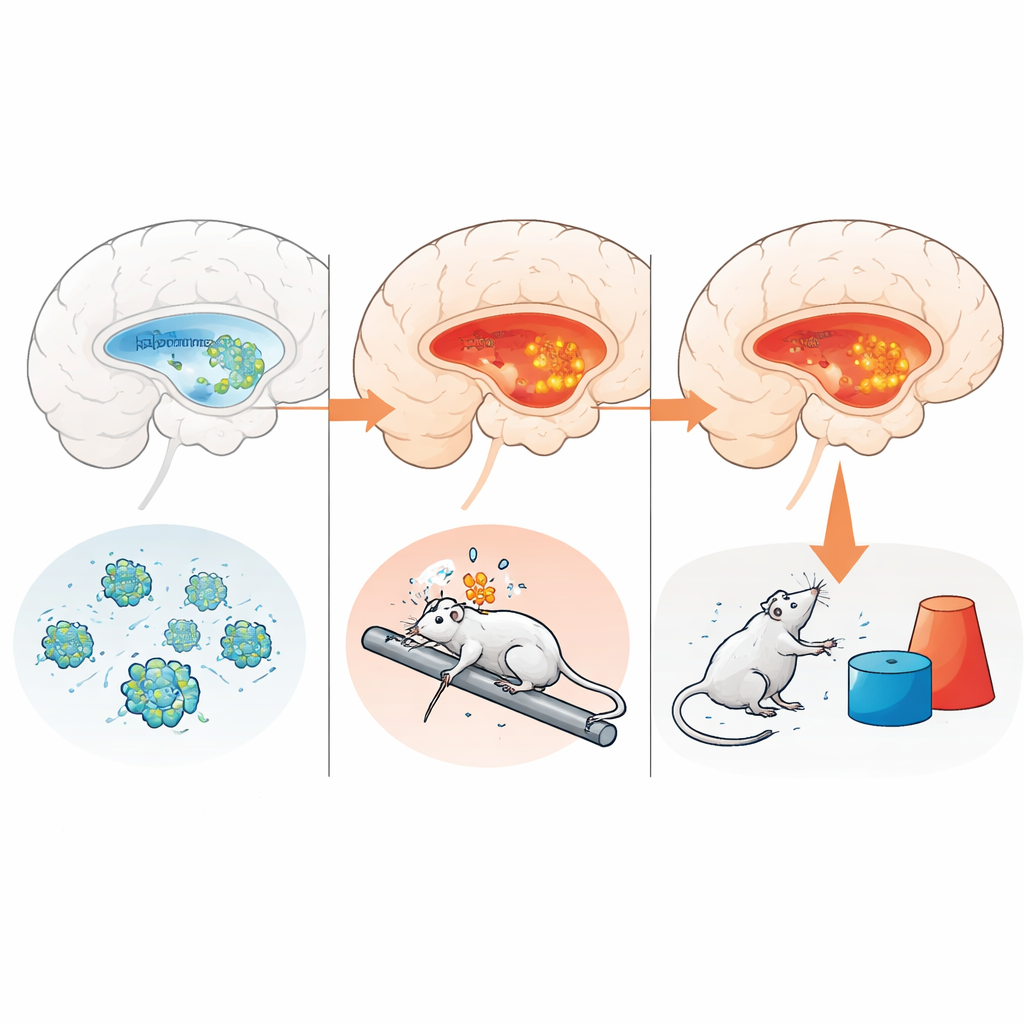
Inflamed memory circuits and hidden vulnerability
By matching brain chemistry to behavior, the team found that higher levels of NLRP3-related proteins—especially the enzyme Caspase-1—in the hippocampus went hand in hand with worse frailty scores. These links were strong for the combined indices that include both physical and cognitive features, but not for the cognitive measures alone or for proteins measured in the amygdala. In other words, chronic activation of inflammatory machinery in a memory-critical brain region appears tightly connected to overall vulnerability of both body and mind. The LPS-treated mice, measured after they had largely recovered, did not show this sustained brain signature, underscoring the difference between brief immune insults and long-term aging.
What this means for understanding healthy aging
This work suggests that cognitive frailty is not simply a matter of birthdays or of having been sick recently. Instead, it reflects a slow, uneven build-up of damage and inflammation in specific brain circuits, particularly in the hippocampus. The newly proposed Cognitive Frailty Index offers a sensitive way to track this combined decline in animal models, opening the door to testing treatments that target brain inflammation. While a single inflammatory hit can temporarily make an otherwise healthy organism look frail, only natural aging reproduced the deeper cellular changes and persistent brain inflammation linked to lasting vulnerability. For people, this points to the importance of tackling chronic low-grade inflammation over the lifespan to help preserve both physical independence and mental clarity.
Citation: Komleva, Y., Lukyanchuk, A., Blagova, A. et al. The role of NLRP3 neuroinflammation in cognitive frailty diversity during aging and after LPS administration in mice. Sci Rep 16, 9100 (2026). https://doi.org/10.1038/s41598-026-41104-2
Keywords: cognitive frailty, brain inflammation, aging, NLRP3 inflammasome, mouse behavior