Clear Sky Science · en
In-situ growth of heterogeneous Au on MoS2 nanosheets for SERS detection of breast cancer-derived miR-210-3p and miR-9-3p
Turning Tiny Cancer Signals into Clear Warnings
Doctors have long known that tumor cells shed tiny fragments of genetic material into the bloodstream, but reliably detecting these faint whispers of disease has been difficult. This study introduces a new nanoscale sensor that uses a special combination of gold and a two-dimensional material called molybdenum disulfide (MoS₂) to amplify these weak signals. The goal is to make it easier to spot breast-cancer–related microRNAs—short pieces of RNA linked to how tumors grow and spread—using a gentle light-based technique that could one day support earlier and more precise diagnosis.
Why These Tiny Molecules Matter
MicroRNAs are extremely short strands of genetic code that help control how cells behave. In cancer, some microRNAs become unusually abundant or scarce, turning them into powerful biomarkers for diagnosis and prognosis. The challenge is that they are present at very low levels, often mixed into complex biological fluids such as blood or cell extracts. Conventional tools like PCR and sequencing can detect them, but they require specialized labs, trained staff, and time-consuming workflows. The researchers set out to build a more direct sensing platform that could, in principle, be simpler to run while still achieving very high sensitivity and the ability to distinguish multiple microRNA targets at once.
Building a Light-Boosting Nanosheet
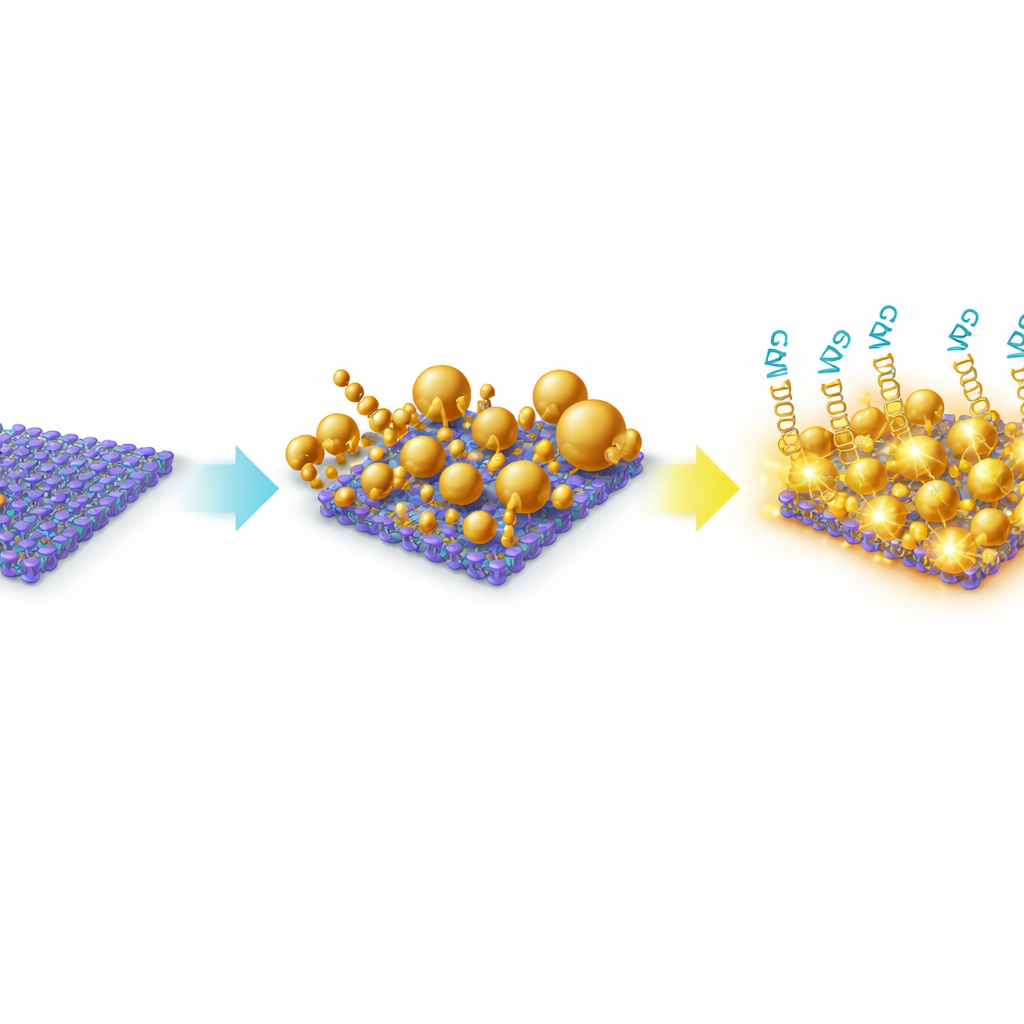
To achieve this, the team engineered a hybrid material made from thin MoS₂ nanosheets decorated with gold nanoparticles. MoS₂ is a sheet-like material only a few atoms thick, offering a large surface for molecules to stick to and a strong interaction with metals. Instead of adding pre-formed gold particles, they grew the gold directly on the MoS₂ surface in solution. This in-situ growth produced a deliberately heterogeneous landscape of gold particles—mostly spheres, but also triangles and irregular shapes—spread across and along the edges of the MoS₂ sheets. Microscopy and spectroscopy confirmed that the gold and MoS₂ formed a stable composite, with gold tightly anchored at defect-rich sites on the sheets. This irregular, “rough” architecture is crucial because it naturally creates many tiny gaps and sharp features where light can be strongly concentrated.
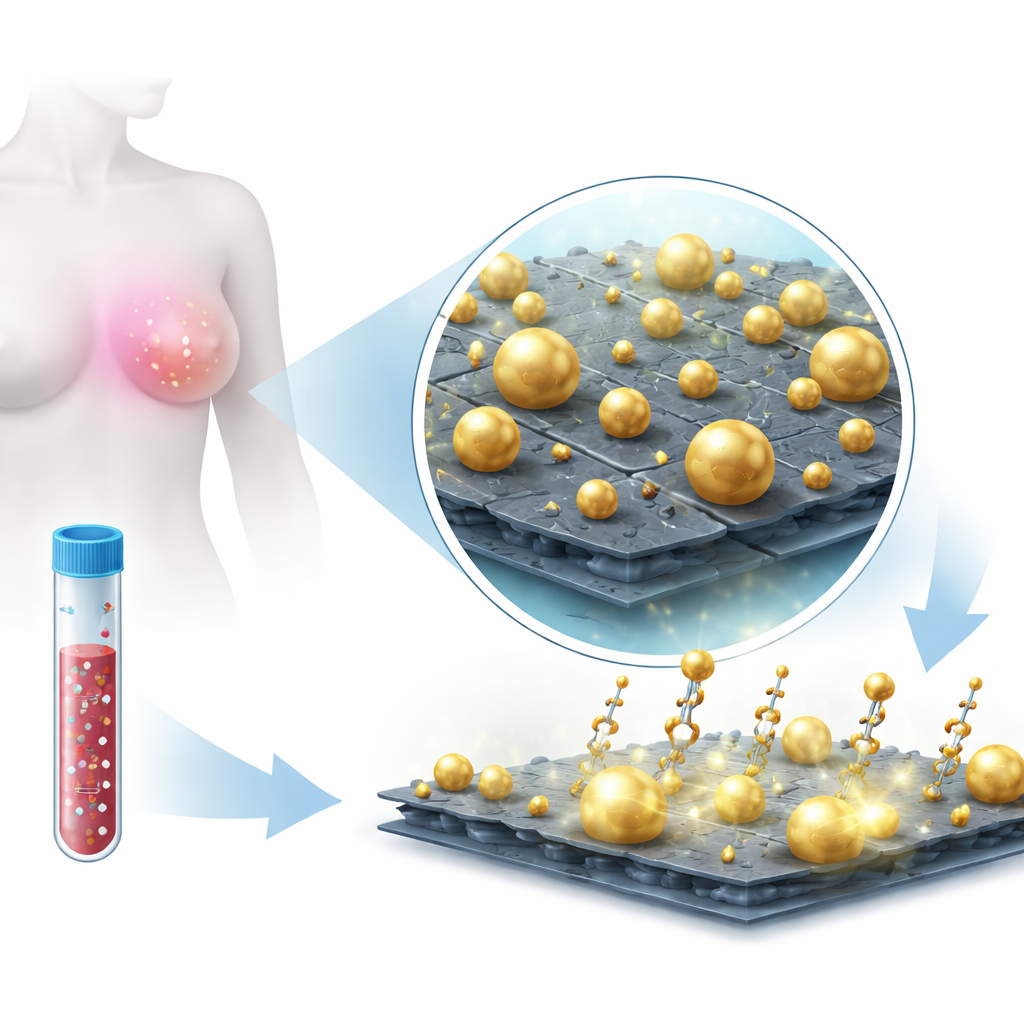
Harnessing Light to Read Molecular Fingerprints
The platform operates using surface-enhanced Raman scattering (SERS), a technique where laser light scatters off molecules and returns a spectral “fingerprint.” On its own, a microRNA is too small and quiet to be easily heard in this way. The researchers instead used short DNA-like probes called locked nucleic acids (LNAs) that latch onto specific microRNA sequences associated with breast cancer, notably miR-210-3p and miR-9-3p. These probes carried bright dye molecules (Cy3 and Cy5.5) that produce strong Raman fingerprints when held near the gold-decorated MoS₂ surface. When a target microRNA binds to its matching LNA probe, the dye is positioned inside the gold “hotspots,” where the local light field is greatly amplified by the combined action of the metal nanoparticles and the MoS₂ sheet. By shining a low-energy near-infrared laser, the team could record clear Raman spectra with distinct peaks that tracked how much microRNA was present.
From Synthetic Targets to Real Cancer Cells
After confirming the structure and stability of their nanocomposite, the researchers tested it with synthetic microRNA sequences to calibrate and validate performance. They found that different spectral peaks from each dye responded sensitively and linearly over wide concentration ranges, allowing them to calculate detection limits down to a few trillionths of a mole (picomolar levels). Importantly, they did not rely on a single peak but used a multi-peak analysis to increase reliability, especially in complex samples. The platform was then challenged with microRNAs extracted from an aggressive breast cancer cell line. Independent PCR measurements showed that one microRNA, miR-210-3p, was much more abundant than miR-9-3p in these cells. Using the same SERS setup, the MoS₂–gold sensor could detect cell-derived miR-210-3p and miR-9-3p down to about 0.1 nanomolar and 0.018 nanomolar, respectively, while preserving the fragile RNA with gentle illumination.
What This Could Mean for Future Cancer Tests
Although this work is still at the laboratory stage, it maps out a clear path for turning a carefully engineered nanosurface into a practical diagnostic tool. By deliberately growing gold of different shapes and sizes on MoS₂, the researchers created a dense network of light-amplifying hotspots that can read out the presence of specific microRNAs through their dye tags. The modest but well-controlled signal enhancement, combined with multi-peak analysis, enables quantitative detection over a broad concentration range in both clean and biologically complex samples. In the long term, this approach could support compact, multiplexed tests that monitor several cancer-linked microRNAs at once, giving clinicians a richer picture of tumor status from small samples and potentially guiding more personalized treatment decisions.
Citation: Zablon, F.M., Pathiraja, G., Dellinger, K. et al. In-situ growth of heterogeneous Au on MoS2 nanosheets for SERS detection of breast cancer-derived miR-210-3p and miR-9-3p. Sci Rep 16, 8902 (2026). https://doi.org/10.1038/s41598-026-41084-3
Keywords: breast cancer biomarkers, microRNA detection, SERS nanosensors, gold nanoparticle composites, MoS2-based biosensing