Clear Sky Science · en
Prediction of colorectal cancer liver metastasis through an MRI radiomic model
Why this research matters
Colorectal cancer is one of the world’s leading killers, mainly because it often spreads to the liver before or after the first surgery. Catching this spread early can dramatically change a patient’s outlook, but today’s scans and blood tests are far from perfect. This study explores whether computers can “read between the pixels” of routine MRI scans of the original bowel tumor to forecast which patients are most likely to develop liver metastases, long before those metastases become visible.
Seeing more than meets the eye
Standard medical imaging is usually interpreted by the human eye, which is very good at spotting obvious masses but less able to perceive subtle patterns. Radiomics is a new approach that treats every scan as a mine of measurable data. By turning shades of gray, shapes and textures into hundreds of numerical features, radiomics allows computers to detect patterns linked to how aggressive a tumor is. In this work, the researchers focused on two common MRI sequences used in colorectal cancer: T2-weighted images, which show anatomy and fluid, and diffusion-weighted images, which capture how water moves within tissues and are sensitive to tumor cell density.
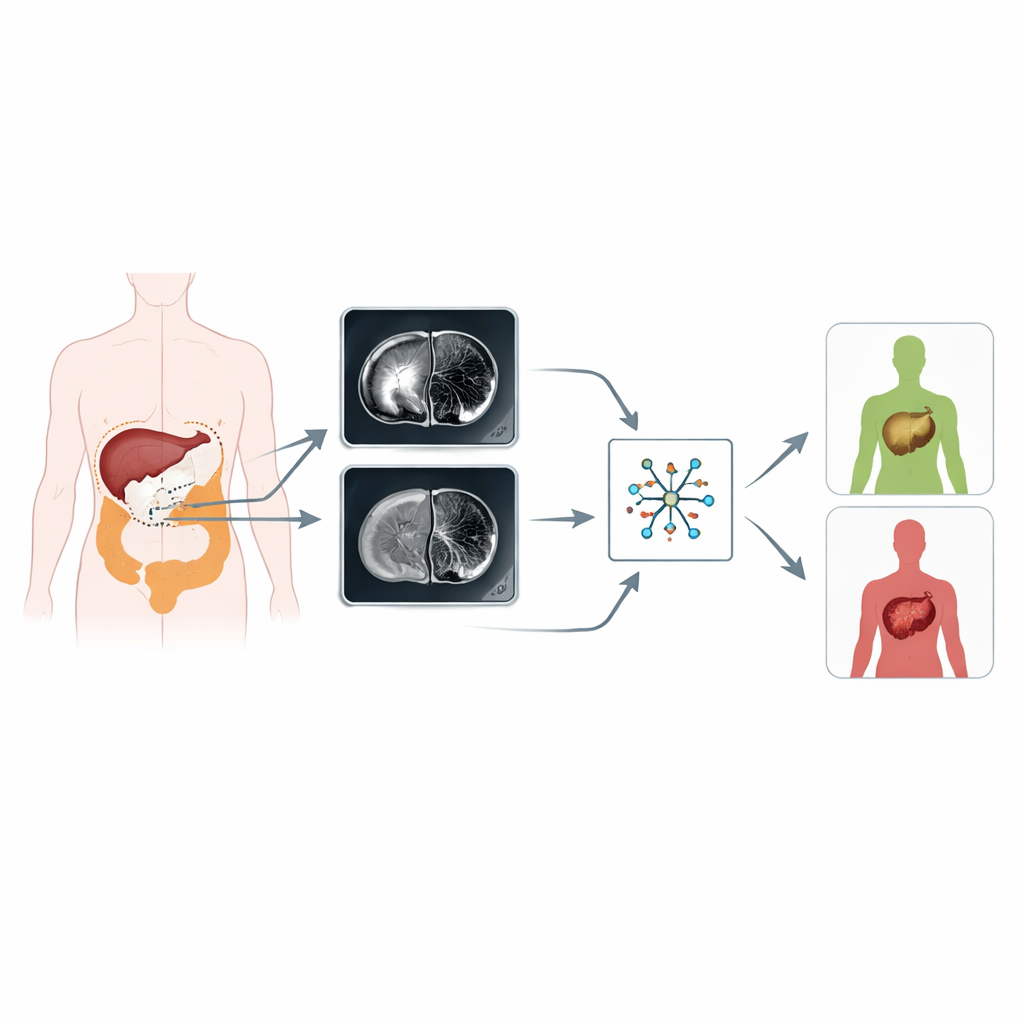
How the study was done
The team analyzed 194 patients with pathologically confirmed colorectal cancer from two hospitals. All patients had MRI scans before any treatment and were followed for at least a year to see who developed liver metastases. Using specialized software, radiologists carefully outlined each primary tumor on the T2-weighted and diffusion-weighted images, slice by slice, while avoiding gas, cysts and surrounding fat. From these regions, the computer extracted 352 radiomic features describing tumor shape and internal texture. Statistical techniques were then used to check that these measurements were reliable between different observers and scanners, filter out redundant information, and narrow the list down to the most informative features.
Building and testing prediction models
With the most useful features in hand, the researchers built several prediction models. One used only simple clinical information such as age and tumor markers in the blood; others relied separately on radiomics from T2-weighted images or diffusion-weighted images. A combined radiomic model merged features from both MRI sequences, and a final “union” model blended these imaging features with clinical risk factors. The team trained these models in the larger group of patients from one hospital and then tested them in an independent group from a second hospital, asking how accurately each model could distinguish patients who did and did not develop liver metastases.
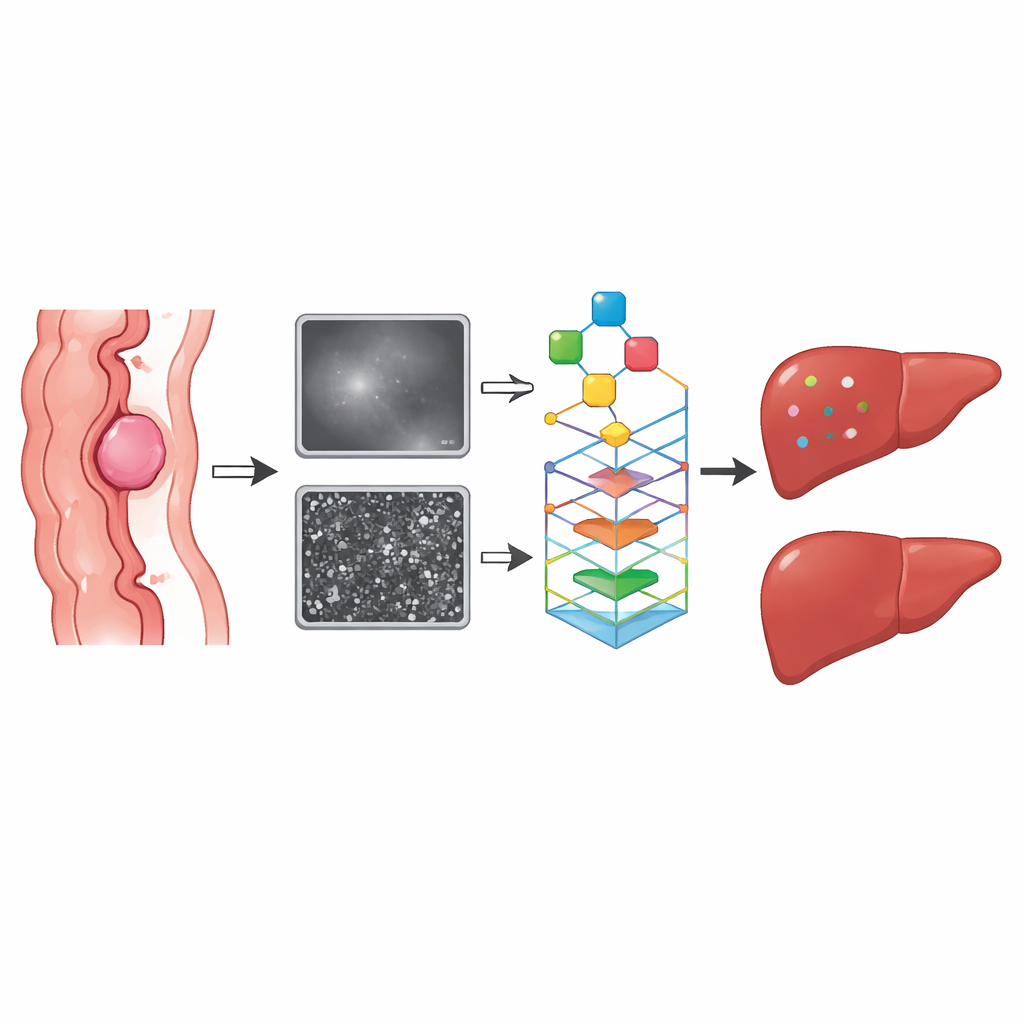
What the models revealed
The combined imaging–clinical model performed best. In both the training and validation groups, it outperformed models based on clinical data alone or on a single MRI sequence. Its ability to separate high- from low-risk patients, measured by the area under the receiver operating characteristic curve, was in the high “good” range. Features capturing tumor texture and shape were especially important: more irregular, heterogeneous tumors were more likely to be linked with later liver spread. A blood marker called carcinoembryonic antigen (CEA) emerged as an independent clinical risk factor and further boosted the model’s accuracy when integrated with the radiomic features.
Opening the black box
To help make this complex model more transparent, the investigators applied a method known as SHAP, which assigns each feature a contribution to each individual prediction. This analysis showed that a particular texture feature from the diffusion-weighted images had the strongest influence on whether the model labeled a patient as high risk. By highlighting which image patterns matter most, such interpretability tools can increase clinicians’ confidence in using artificial intelligence–driven predictions in real-world decision-making.
What this means for patients
The study suggests that routinely acquired MRI scans of the primary colorectal tumor contain hidden clues about whether cancer cells are likely to seed the liver. By combining these subtle imaging signatures with standard blood tests, a radiomic model could help doctors identify high-risk patients earlier, tailor follow-up schedules, and choose more intensive or targeted treatments when needed. Although larger, prospective studies are still required before this approach can be widely adopted, it points toward a future in which computers help unlock extra prognostic power from images that are already being taken, without exposing patients to additional procedures.
Citation: Wu, YK., Wang, X., Du, PZ. et al. Prediction of colorectal cancer liver metastasis through an MRI radiomic model. Sci Rep 16, 11148 (2026). https://doi.org/10.1038/s41598-026-40970-0
Keywords: colorectal cancer, liver metastasis, MRI radiomics, cancer prediction, medical imaging