Clear Sky Science · en
Effects of testosterone on gene expression in males and females across 40 human tissues
Why hormone differences matter for health
Men and women often face different risks for conditions like diabetes, heart disease, and Alzheimer’s disease, and doctors know that the sex hormone testosterone plays a role. But exactly how testosterone shapes these risks at the level of our genes has remained unclear. This study looks inside human tissues to ask a simple but far-reaching question: how does testosterone relate to gene activity in men and women, and could those differences help explain why the same hormone can have opposite health effects across the sexes?
Looking at hormone effects without drawing blood
Instead of directly measuring testosterone in thousands of new volunteers, the researchers combined two powerful public resources. Large genetic studies in the UK Biobank have already mapped which DNA changes are linked to higher or lower testosterone levels in men and in women. The Genotype–Tissue Expression (GTEx) project has collected detailed gene activity profiles from dozens of tissues donated after death. By using the DNA-based "polygenic scores" from UK Biobank, the team could estimate each GTEx donor’s likely testosterone level, separately for total testosterone and for the portion that is more loosely bound in the blood and thought to be more biologically active. They then asked how well patterns of gene activity in each tissue tracked these genetically predicted hormone levels within men and within women.
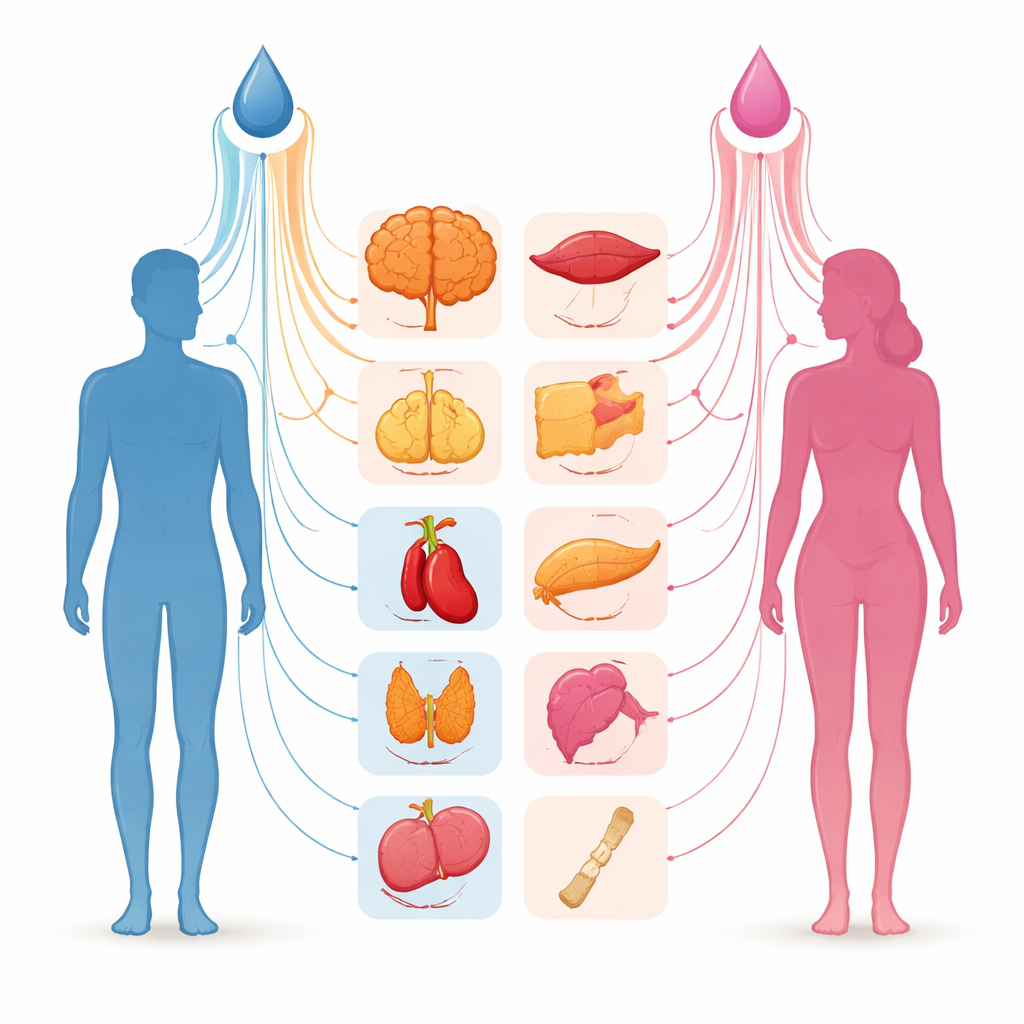
Checking hormone signals across the body
For each of 40 tissues present in both sexes—ranging from brain and muscle to fat, blood vessels, skin, and internal organs—the authors estimated how much of the variation in predicted testosterone could be "explained" by all genes acting together. They found that the strength of this link varied widely from one tissue to another and from men to women. In women, gene activity in the mammary gland, deep belly fat, parts of the esophagus, and skin captured a relatively large share of the variation in total testosterone, although these estimates did not reach strict statistical significance after adjusting for many comparisons. In men, brain regions such as the anterior cingulate cortex and putamen showed some of the higher values, but again with wide uncertainty. For the bioavailable fraction of testosterone, no tissue in either sex showed a clearly stronger connection than the others.
Zooming in on individual genes
Beyond the overall patterns, the researchers searched for single genes whose activity rose or fell with predicted testosterone levels. Using a statistical approach that reduces false positives by accounting for hidden confounding factors, they tested more than 20,000 gene transcripts per tissue. Only four transcripts reached the very strict threshold required to be considered strongly associated—and all of them appeared in women, not men. These included NUPR1L and PSPHP1, which were most clearly linked to testosterone in the wall of the tibial artery and in skeletal muscle, as well as RP11-208G20.3 in arteries and pancreas, and PTPRD in the pancreas. In each case, higher predicted total testosterone in women went along with lower activity of these genes. No single gene showed a comparable signal for the bioavailable hormone measure or in any male tissue.
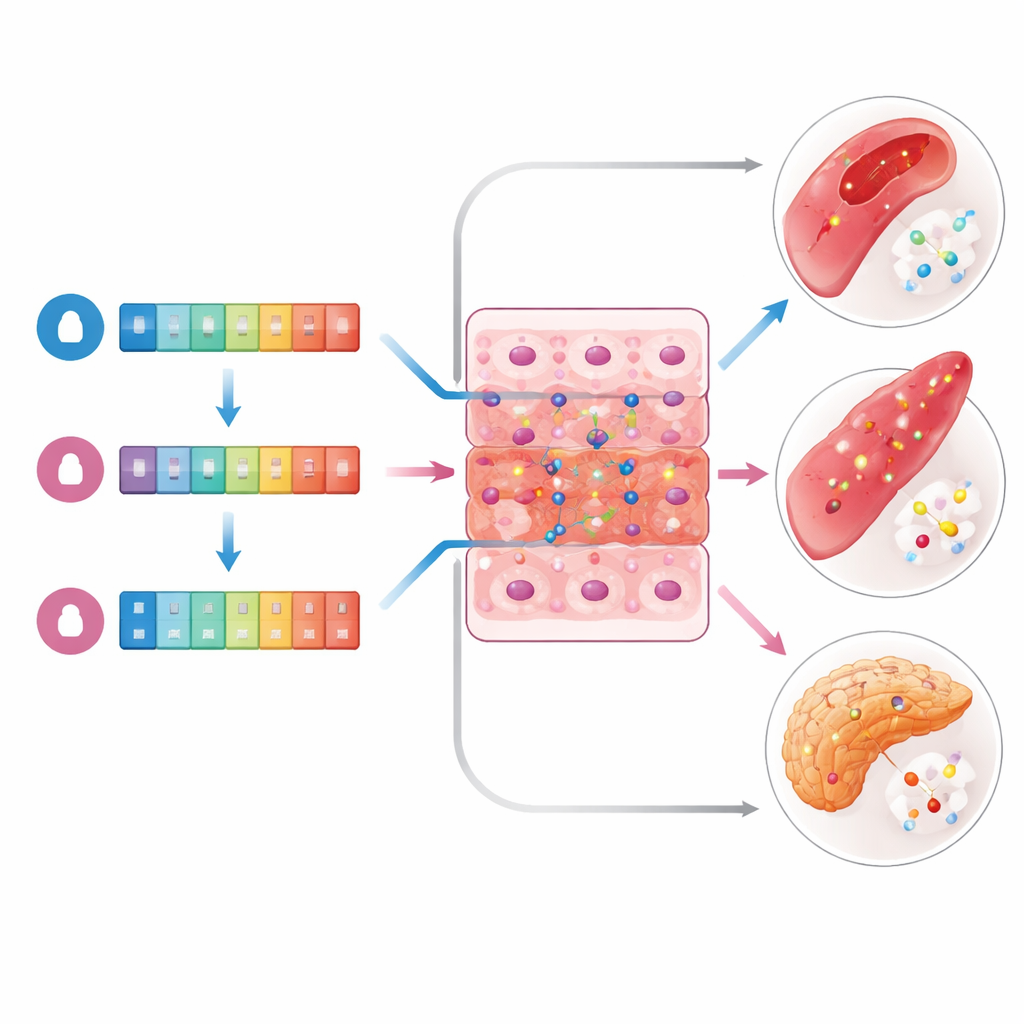
Clues from biological pathways
Although only a handful of genes met the strictest cutoff, many more showed weaker but still suggestive ties to testosterone in specific tissues. When these were grouped and tested for shared functions, some themes emerged. In women, genes linked to predicted testosterone were enriched for immune-related processes and for pathways involved in nerve cell signaling. In men, the genes pointed instead toward cellular activities such as energy-related enzyme functions and stress responses, as well as certain immune signaling routes. These hints suggest that testosterone’s influence on health may run through different biological "wiring" in male and female bodies, even when the same hormone is involved.
What this means for understanding sex differences
For a lay reader, the main takeaway is that testosterone does not act in a uniform way across the body—or across the sexes. In this large-scale analysis, gene activity in several female tissues, particularly breast and fat, showed stronger links to genetically predicted testosterone than was seen in male tissues. A small set of genes in women’s artery, muscle, and pancreas tissues stood out as especially sensitive to total testosterone, while no equally strong signals appeared in men. At the same time, most connections were modest, and many did not survive the most conservative statistical tests, reminding us that current datasets are still relatively small for questions this complex. Overall, the study offers early molecular clues to how the same hormone can contribute to different disease patterns in men and women and highlights the need for larger, more diverse studies with direct hormone measurements to fully map testosterone’s sex-specific genetic footprint.
Citation: Cheruiyot, E.K., Zhihong, Z. & McRae, A.F. Effects of testosterone on gene expression in males and females across 40 human tissues. Sci Rep 16, 10223 (2026). https://doi.org/10.1038/s41598-026-40863-2
Keywords: testosterone, sex differences, gene expression, human tissues, polygenic scores