Clear Sky Science · en
Effective dean vortex separation at reduced flow rates towards rare cell sorting
Why sorting rare cells matters
Cancer cells that drift into the bloodstream or other body fluids are like early warning beacons for disease. They are, however, vastly outnumbered by ordinary blood cells, making them extremely hard to find and study. This paper describes a new micro‑scale device that uses gentle swirling flows inside a tiny spiral channel to pull larger, cancer‑like cells away from smaller white blood cells at much lower liquid speeds than usual. This low‑stress, easy‑to‑integrate approach could help laboratories enrich rare cells for diagnosis and treatment monitoring without damaging them.
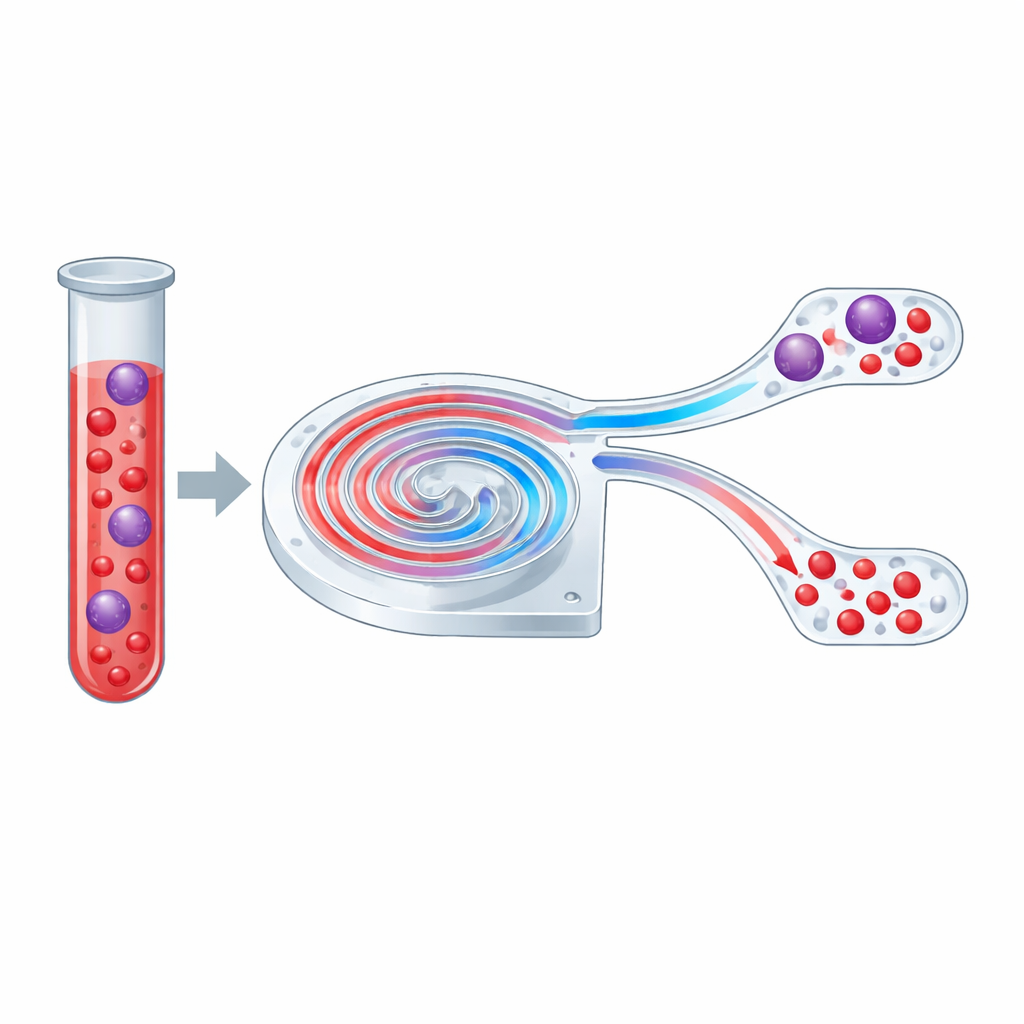
A tiny spiral road for cells
The heart of the study is a transparent microchip containing a spiral‑shaped channel thinner than a human hair. When fluid moves through a straight channel, cells mainly feel a push in the direction of flow and a subtle sideways nudging that depends on their size. In a curved, spiral path, a second effect appears: the liquid rolls into paired whirlpools across the channel cross‑section, known as Dean vortices. These whirlpools sweep smaller objects across the stream while larger ones are held closer to one side. By tuning this balance, the authors designed a spiral that can split a mixture of particles or cells into two streams based primarily on size.
Making spirals work at gentle speeds
Most existing spiral devices sort efficiently only when pushed very hard, at flow rates hundreds to thousands of milliliters per hour. Such conditions shorten processing time but raise shear stress on cells and make it difficult to connect the spiral to other microfluidic steps that prefer slower, more controlled flows. The team set a different goal: maintain strong size‑based sorting around 50 milliliters per hour, an order of magnitude gentler. To achieve this, they systematically varied the geometry of nine spiral designs—changing channel width, height, and the slope of the outer wall—then combined experiments with computer simulations of the circulating flow.
Following particles through the whirlpools
To first understand the physics, the researchers flowed fluorescent plastic beads of two sizes, 10 and 15 micrometers, through each spiral. At low speeds, both bead types gathered near the inner wall. As the flow increased, the focused stream migrated across the channel to the outer wall, but at different threshold speeds for each size. For one representative design, smaller beads shifted outward around 30 milliliters per hour, while the larger ones did so around 60 milliliters per hour. This created an intermediate range where big and small beads exited on opposite sides. Simulations of the fluid motion revealed that, at higher speeds, the beads settle very close to the centers of the Dean vortices, confirming a long‑standing but previously unproven idea about how these devices work.
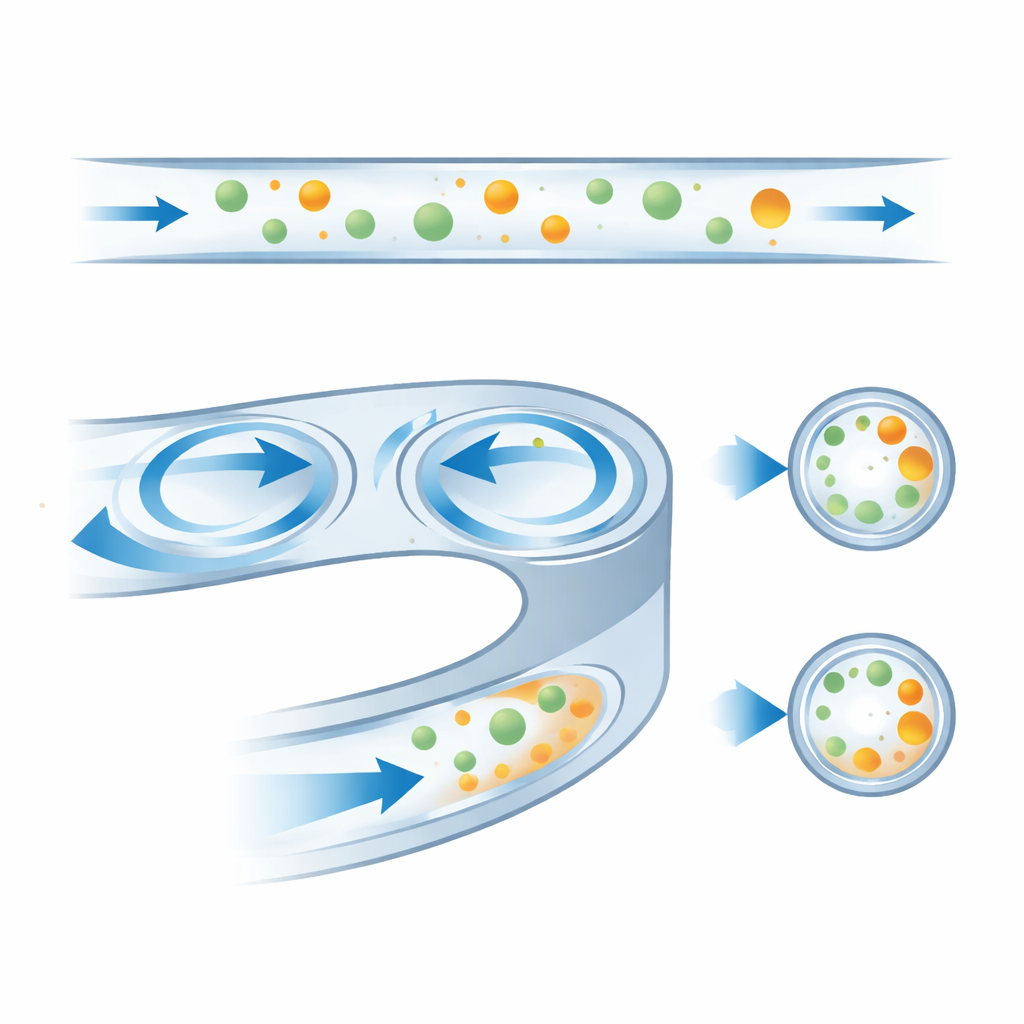
Shaping the channel for the best split
By comparing many designs, the authors identified how simple geometric choices control performance. A steeper slope on the outer wall pushes the internal whirlpools further outward, shifting the stable positions of beads and changing the flow rate at which they swap sides. Narrow channels bring the vortices too close to the inner wall, causing particles to be drawn into them too early, while very flat channels demand higher speeds before any shift occurs. The best compromise used a 250‑micrometer‑wide channel with a moderate wall slope and aspect ratio, which provided a clear gap between the two bead sizes at 40–60 milliliters per hour and remained robust to small flow variations.
From plastic beads to living cancer cells
With the optimal spiral in hand, the team turned to biological samples. They tested several cancer cell lines that mimic circulating tumor cells and compared their behavior to that of white blood cells from prepared blood. Despite being softer and more varied in size than beads, all cell types focused into narrow streams and shifted from the inner to the outer side as the flow increased, in a strongly size‑dependent way. Operating at 50 milliliters per hour, the device routed about 89% of white blood cells to the “waste” side while keeping 75–86% of the larger cancer‑like cells in the “enriched” side. Importantly, cell survival remained around 98%, even after passing through the spiral at different flow rates.
What this means for future cancer tests
In plain terms, the authors have built and decoded a tiny spiral filter that can gently skim off most of the background blood cells while retaining the rarer, larger cancer‑like cells, all at relatively slow, cell‑friendly flow rates. By clarifying how the inner and outer streams form and how channel shape controls them, the work turns a largely trial‑and‑error technology into a more predictable tool. On its own, the device will not capture every last cancer cell, but it provides a powerful pre‑enrichment step that can be directly linked to more selective biological methods. This combination could ultimately make it easier to detect, analyze, and monitor cancer using small fluid samples from patients.
Citation: Dupont, E., Artinyan, L., Brunin, C. et al. Effective dean vortex separation at reduced flow rates towards rare cell sorting. Sci Rep 16, 10422 (2026). https://doi.org/10.1038/s41598-026-40845-4
Keywords: microfluidic cell sorting, spiral microchannel, circulating tumor cells, Dean vortices, liquid biopsy