Clear Sky Science · en
Targeted activation of Nrf2 via sulforaphane-loaded exosomes attenuated azoospermic condition in the rat model
Why this matters for men’s health
Azoospermia, the complete absence of sperm in semen, is one of the most difficult forms of male infertility to treat. Many men develop this condition after chemotherapy drugs damage the testes. Current treatments often cannot restart sperm production, leaving assisted reproduction as the only option. This study explores a more natural repair strategy: using tiny biological bubbles, called exosomes, to carry a broccoli-derived compound directly into damaged testes and coax them back into making sperm.
Damage from cancer treatment
Chemotherapy drugs such as busulfan are powerful cancer fighters, but they can also wipe out the germ cells in the testes that produce sperm. In the rat model used here, a single injection of busulfan into the testes led to a sharp drop in sperm count and motility, a rise in misshapen sperm, and a loss of testis weight. Microscopic examination showed that the normal layered structure of the sperm‑producing tubules had collapsed, with many cells dead or missing and scar‑like tissue forming between them. Levels of testosterone fell as well, showing that hormone production was also affected.
Turning exosomes into tiny delivery capsules
To tackle this damage, the researchers turned to exosomes, natural nano‑sized packets that cells use to send molecules to one another. They isolated exosomes from donated human blood serum and confirmed their size and shape with several techniques. They then loaded these vesicles with sulforaphane, a plant compound best known for being abundant in broccoli and other cruciferous vegetables. Sulforaphane is a strong activator of Nrf2, a protein that helps cells turn on antioxidant defenses and cope with stress. By packaging sulforaphane inside exosomes and injecting them directly into rat testes, the team hoped to raise its local effect while avoiding the instability and low absorption that limit sulforaphane in free form.
Recovering sperm and rebuilding tissue
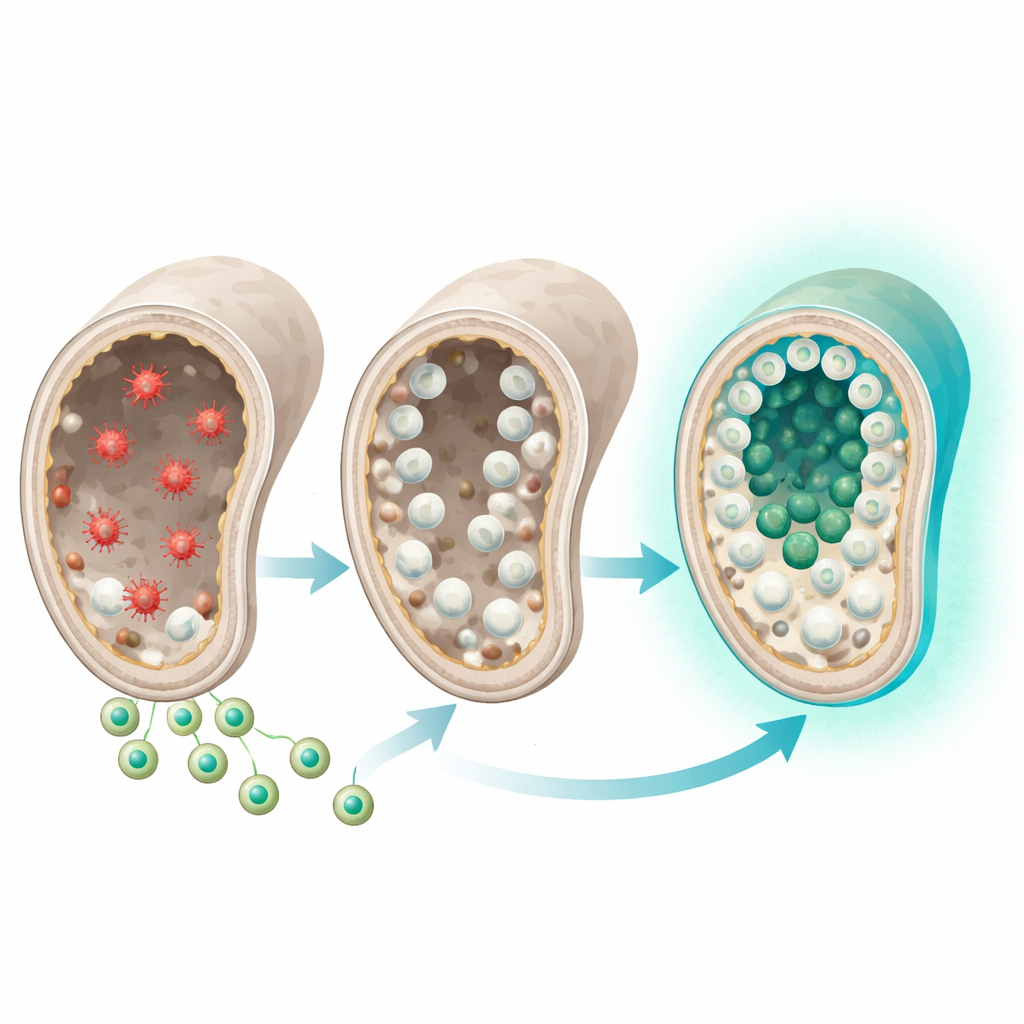
Rats were divided into five groups: healthy controls, untreated azoospermic animals, and three treatment groups receiving sulforaphane alone, empty exosomes alone, or sulforaphane‑loaded exosomes. Two months after a single injection, the combination treatment clearly stood out. Rats that received sulforaphane‑loaded exosomes showed much higher sperm counts and motility, fewer malformed sperm, and a greater testis weight index than untreated animals or those given either component alone. Under the microscope, their testes displayed a near‑normal arrangement of germ cells at all stages of development, from stem‑like precursors to mature sperm, and far less fibrous scarring. The expression of key germ cell genes, DAZL and VASA, which had been strongly reduced by busulfan, returned to levels similar to healthy controls only in the combination group.
Resetting cell stress and cleanup systems
The study also probed how the treatment affected internal stress‑response pathways. After busulfan injury, the testes showed signs of oxidative imbalance: the Nrf2 gene, which coordinates antioxidant defenses, was abnormally high, and genes involved in cellular “cleanup” through autophagy were also overactive. Such persistent activation can signal unresolved damage rather than protection. Sulforaphane‑loaded exosomes brought Nrf2 and the autophagy‑related genes LC3, Beclin1 and p62 back toward normal levels, while boosting the activity of antioxidant defenses such as glutathione peroxidase and overall antioxidant capacity. Sulforaphane or exosomes by themselves shifted these markers in the right direction but did not normalize them as completely. Testosterone levels also improved most strongly in the combination group, suggesting better recovery of hormone‑producing cells.
A new path toward restoring fertility
Taken together, the findings suggest that sulforaphane‑loaded exosomes can steer damaged testes away from a chronic stress state and toward balanced self‑repair. By quietly re‑tuning antioxidant and cleanup pathways, this targeted therapy allowed germ‑cell stem populations to recover and resume building mature sperm in a rat model of chemotherapy‑induced azoospermia. While much work remains—such as testing long‑term safety, optimizing doses and confirming that offspring are healthy—this approach points to a future in which some forms of male infertility might be treated not just by retrieving rare remaining sperm, but by helping the testes regenerate their natural sperm‑producing machinery.
Citation: Ahmadian, S., Fajri, M., Roelen, B.A. et al. Targeted activation of Nrf2 via sulforaphane-loaded exosomes attenuated azoospermic condition in the rat model. Sci Rep 16, 9752 (2026). https://doi.org/10.1038/s41598-026-40709-x
Keywords: azoospermia, male infertility, exosome therapy, sulforaphane, oxidative stress