Clear Sky Science · en
Development and validation of green spectrophotometric methods for simultaneous determination of etoricoxib and tramadol
Why cleaner testing of pain medicines matters
Pain relief often relies on drug combinations that work together, such as etoricoxib and tramadol, to ease discomfort while limiting side effects. Before these medicines reach patients, manufacturers must constantly check that each dose actually contains the right amount of each ingredient. Many current testing methods are accurate but expensive, time‑consuming, and generate chemical waste. This study explores simpler, faster, and more environmentally friendly ways to measure both drugs at once using light, offering a greener path for routine quality control in the pharmaceutical industry.
Pain relief partners in a single dose
Etoricoxib and tramadol work in different but complementary ways to control pain. Etoricoxib targets inflammation with fewer stomach problems than many older painkillers, while tramadol acts on the nervous system to reduce the sensation of pain. New products like dispersible granules combine these two drugs to provide strong, multimodal relief, often allowing doctors to use lower doses of opioids. However, combining drugs in one packet creates a challenge for testing: analysts must be able to confirm both components in the same sample quickly and reliably, without driving up costs or harming the environment.
Using light instead of complex machines
Traditional high‑performance liquid chromatography, a standard laboratory tool, can measure both drugs accurately but requires costly instruments, specialized solvents, and more time for each run. The authors instead turned to UV spectrophotometry, which measures how strongly a solution absorbs ultraviolet light. This technique is simple, inexpensive, and widely available. The catch is that etoricoxib and tramadol absorb light in similar regions, so their signals overlap and blur together. The study set out to untangle these overlapping signals mathematically, allowing both drugs to be measured in the same solution without any physical separation step.
Two smart tricks for separating mixed signals
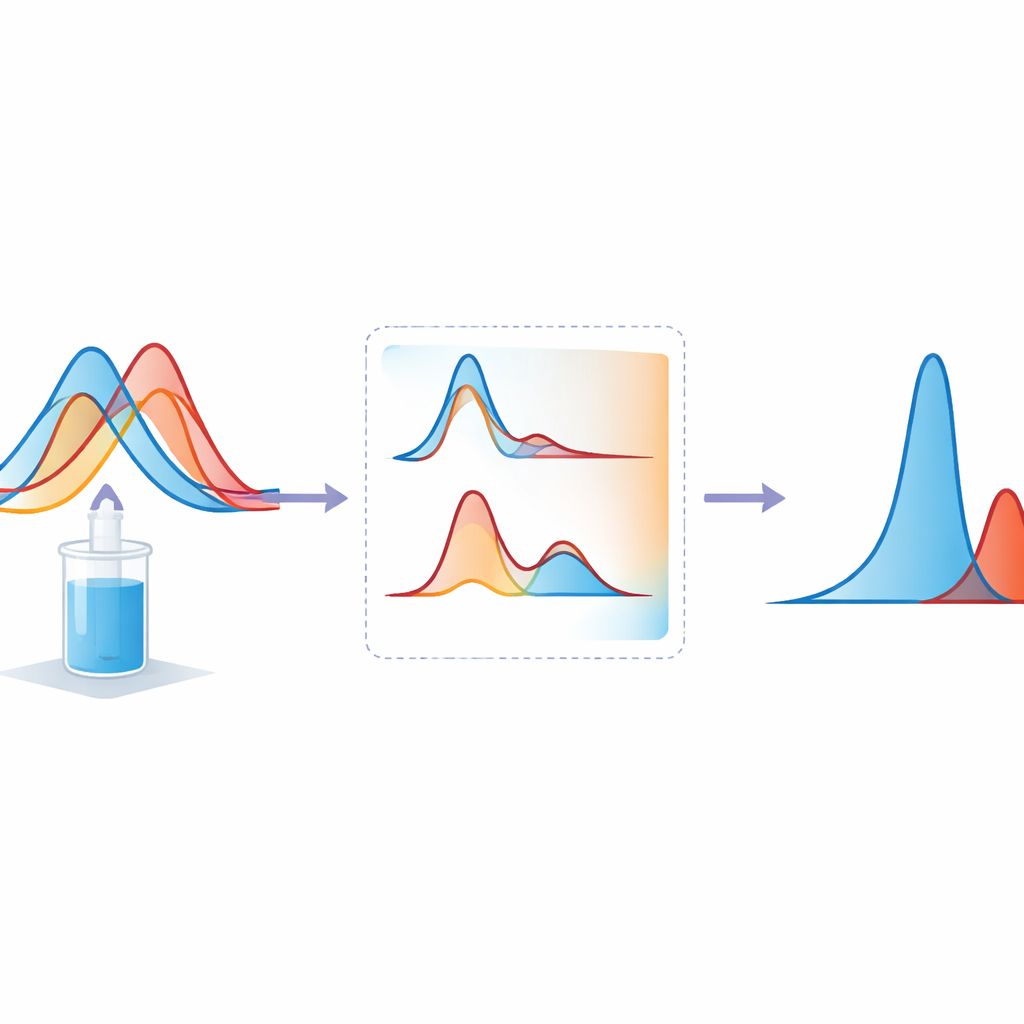
The team developed two related approaches. The first, called the simultaneous equation method, measures light absorption at two key wavelengths where each drug contributes a different proportion of the total signal. By using known response factors for each compound, simple equations can then be used to calculate the amount of etoricoxib and tramadol in the mixture. The second approach goes a step further by transforming the original light‑absorption curves into their first derivatives, which effectively highlights how quickly the signal changes with wavelength. In these derivative curves, the researchers identified points where one drug’s signal is zero while the other remains strong, allowing each component to be read almost as if it were alone.
Testing performance and real medicine samples
The researchers carefully checked how well both methods performed. Across practical concentration ranges, the measured signals rose smoothly with increasing drug levels, and the mathematical fits were almost perfect. Detection limits showed that very small amounts of each drug could still be picked up, with the derivative method offering slightly higher sensitivity. Accuracy was confirmed by adding known amounts of each drug to test mixtures and recovering values very close to 100 percent. Repeated measurements on the same day and on different days gave nearly identical results, confirming that the procedures are precise and robust. When applied to commercial etoricoxib–tramadol granules, the new methods matched the results from an established chromatography technique, but with less effort and lower cost.
Greener chemistry with fewer trade‑offs
Beyond performance, the study examined environmental impact. Because the UV methods use only small volumes of methanol and require no complex solvent mixtures, they generate much less waste than conventional chromatography. An established "green score" tool rated the new approaches favorably, reflecting low energy demands, minimal chemical hazards, and reduced disposal needs. While these light‑based methods are not as sensitive as high‑end chromatography and cannot reveal every impurity or breakdown product, they are more than adequate for routine checks of finished products. For everyday quality control of combined etoricoxib–tramadol medicines, these approaches offer a practical balance of accuracy, simplicity, and environmental responsibility, helping ensure that powerful pain relief reaches patients safely and sustainably.
Citation: Al-Mutairi, A.A., Felemban, R.A., Alqahtani, A.S. et al. Development and validation of green spectrophotometric methods for simultaneous determination of etoricoxib and tramadol. Sci Rep 16, 9531 (2026). https://doi.org/10.1038/s41598-026-40668-3
Keywords: pain medication testing, green analytical chemistry, UV spectrophotometry, etoricoxib and tramadol, pharmaceutical quality control