Clear Sky Science · en
Pancreatitis-associated chymotrypsin C (CTRC) variant p.R240Q selectively impairs trypsinogen degradation through disruption of long-range electrostatic interactions
Why this matters for pancreatic health
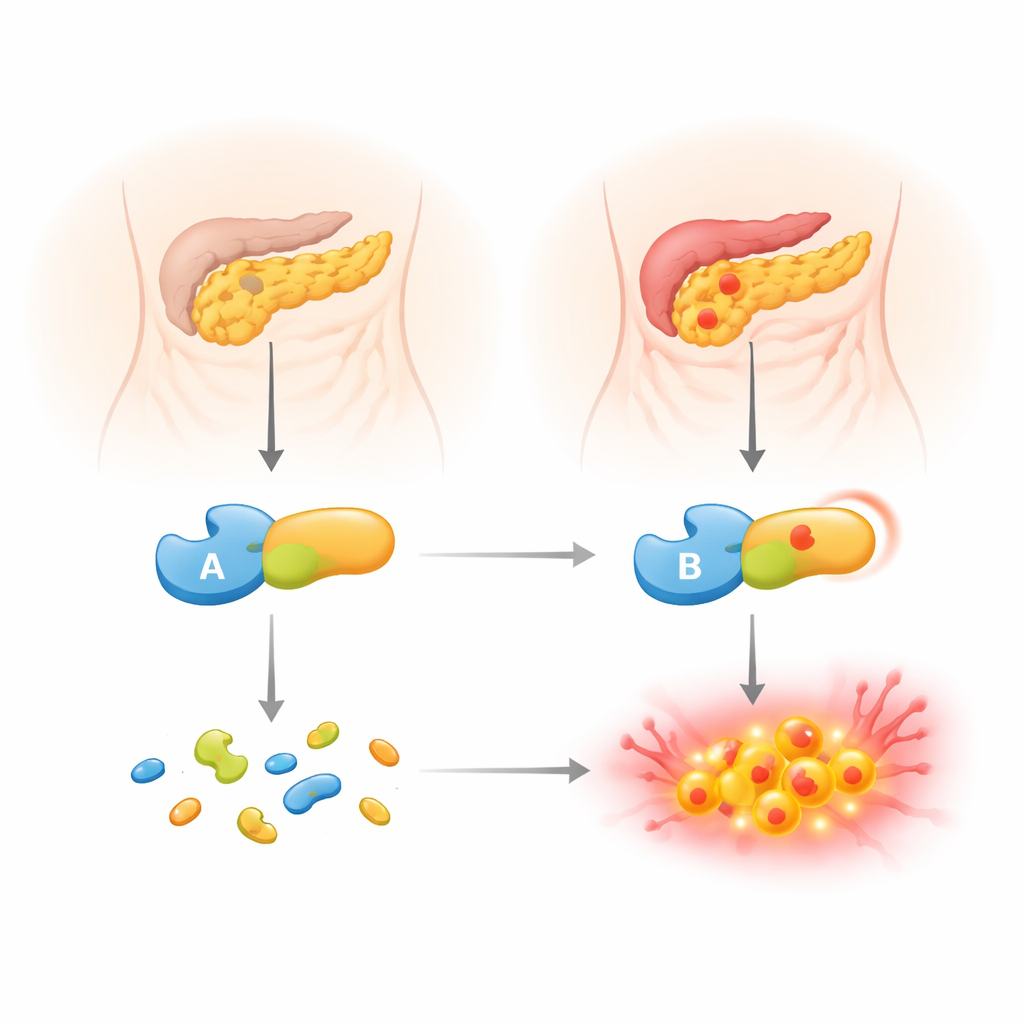
Chronic pancreatitis is a painful, long-lasting inflammation of the pancreas that can lead to diabetes, digestive problems, and a serious drop in quality of life. This study looks at why some people are more vulnerable because of small inherited changes in their digestive enzymes. By zooming in on a single genetic variant in one protective enzyme, the researchers show how a subtle molecular change can tilt the balance from protection toward self-damage inside the pancreas.
A built-in safety system in digestion
Our pancreas produces powerful protein-digesting enzymes that are normally released into the gut in an inactive form. One of these precursors, trypsinogen, can sometimes switch on too early, inside the pancreas itself. When that happens, it can start digesting the organ from within and trigger inflammation. To keep this in check, the body uses safeguards, including an inhibitor protein (SPINK1) and another digestive enzyme, chymotrypsin C (CTRC). CTRC has a dual role: it helps activate digestive enzymes when and where they are needed, but it also cuts up extra trypsinogen before it can turn into active trypsin and cause harm. People who inherit weakened forms of CTRC are known to have a higher risk of chronic pancreatitis.
A rare genetic change under the microscope
The team focused on an uncommon CTRC variant called p.R240Q, found in a boy from Slovakia with childhood-onset chronic pancreatitis and in his affected father. This variant changes a single building block on the enzyme surface, swapping a positively charged unit for a neutral one. Large genetic databases classify it as likely harmless, yet it has appeared in several young patients with the disease. To find out what it really does, the authors combined genetic information from the family with detailed laboratory experiments. They compared the variant enzyme side-by-side with normal CTRC, looking at how well it is produced by cells, how active it is on standard test substrates, and how it interacts with trypsinogen, the key substance it is supposed to control.
Normal strength, but misplaced aim
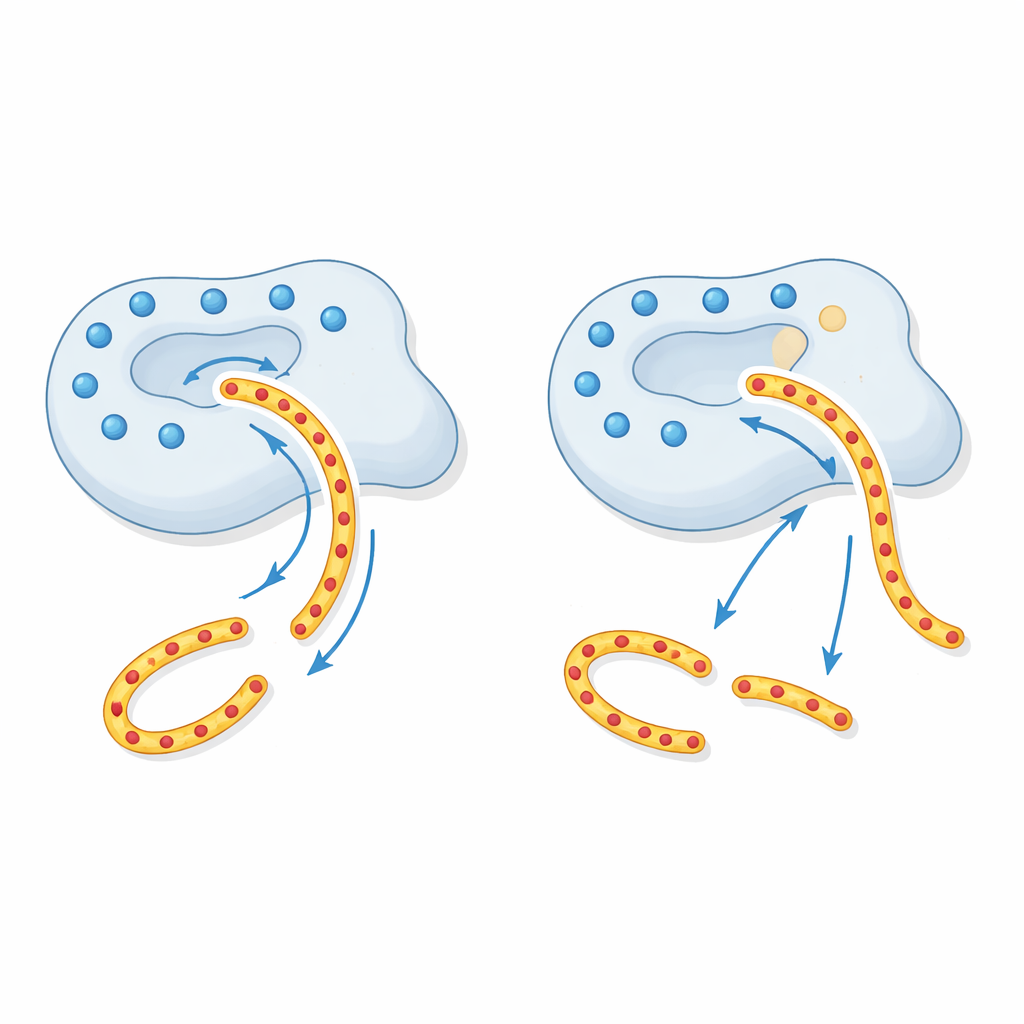
First, the researchers showed that cells make and secrete the p.R240Q enzyme just as well as the normal version, so the variant does not cause misfolding or a production failure. In test-tube assays using a small synthetic peptide and a milk protein (beta-casein), the variant worked as well as or even slightly better than normal CTRC. This means its cutting power is intact. The surprise came when they examined the one reaction that matters most for pancreatitis: breaking down human cationic trypsinogen. Here, the variant enzyme was much less effective—about four to five times slower at chopping up trypsinogen and far poorer at preventing trypsinogen from autoactivating into trypsin. In autoactivation experiments, much more variant CTRC was needed to keep trypsin levels in check compared with the normal enzyme.
A disrupted electrostatic “tractor beam”
To understand why this happens, the authors drew on structural models of CTRC. Around the groove where CTRC grips its substrates, there is a ring of positive electrical charge formed by several charged amino acids, including the site altered in p.R240Q. Trypsinogen regions that CTRC must cut are strongly negative, so this positively charged ring acts like a long-range “tractor beam,” guiding trypsinogen into the right position. By neutralizing one of these positive spots, the p.R240Q variant weakens this distant attraction. The enzyme is still sharp, but the damaged guidance system means trypsinogen does not bind as efficiently, so less of it gets safely degraded. Other substrates that do not depend as strongly on this charge pattern are processed normally, explaining the highly selective defect seen in the experiments.
What this means for patients and testing
Clinically, the affected boy and his father also carried a second damaging CTRC variant, and the father had additional lifestyle risks such as heavy alcohol use. This fits with the idea that chronic pancreatitis often results from several genetic and environmental hits added together. The work shows that a variant that looks harmless by common prediction tools can still be dangerous in a very specific way. For genetic testing, the message is clear: when judging whether a CTRC variant raises pancreatitis risk, scientists must test it on the real-life target, trypsinogen, not just on generic lab substrates. In everyday terms, this study reveals how a tiny change in the enzyme’s “guiding surface” can quietly undermine one of the pancreas’s key safety locks, nudging susceptible individuals toward chronic inflammation.
Citation: Nagy, Z.A., Sándor, M., Hegyi, E. et al. Pancreatitis-associated chymotrypsin C (CTRC) variant p.R240Q selectively impairs trypsinogen degradation through disruption of long-range electrostatic interactions. Sci Rep 16, 8937 (2026). https://doi.org/10.1038/s41598-026-40633-0
Keywords: chronic pancreatitis, chymotrypsin C, trypsinogen, genetic variant, digestive enzymes