Clear Sky Science · en
Large variations in total and allele-specific transcript expression in a disease mutation-independent manner
Why this matters for people with cystic fibrosis
Cystic fibrosis (CF) is often described as a textbook “single-gene” disease, yet people who carry the very same mutation can have strikingly different symptoms and life paths. This study asks a deceptively simple question with far-reaching consequences: even when the faulty gene is the same, do patients actually make different amounts of the gene’s message, and could that help explain why their disease looks so different—and how well they respond to modern drugs?
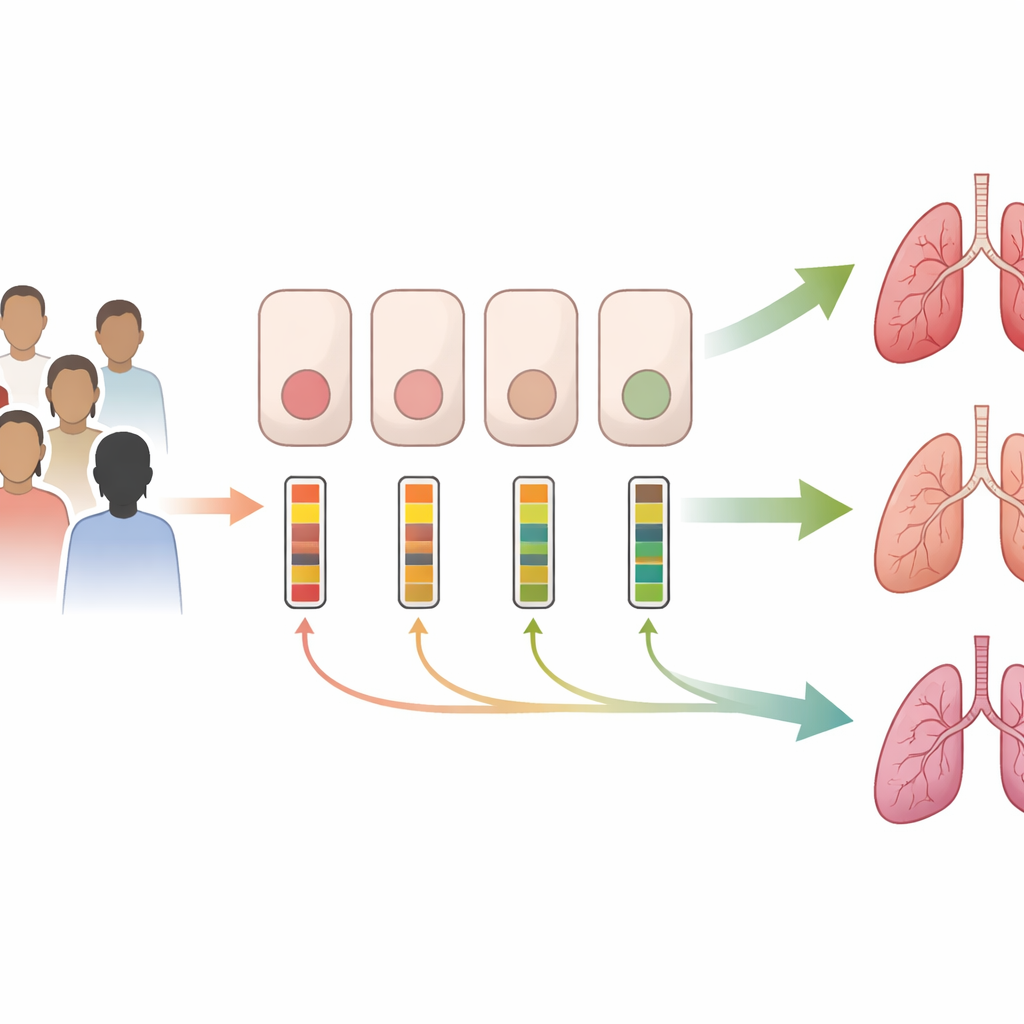
Different patients, same mutation, very different gene activity
The researchers focused on the CFTR gene, whose faulty versions cause CF. Instead of looking at DNA changes alone, they measured how much CFTR messenger RNA (mRNA)—the gene’s working “script” for making protein—is present in airway cells collected by gentle nasal brushings. Over five years they gathered samples from children and adults with CF, as well as a small group without CF. Many patients carried the common F508del mutation, either on both gene copies or on one copy paired with a different fault. Even within these groups, CFTR mRNA levels ranged widely—varying several-fold from person to person. Healthy volunteers also showed broad variation, hinting that CFTR expression naturally differs between individuals, and that this built-in variability carries over into CF.
When the two gene copies do not pull their weight equally
For patients who carried two different CFTR mutations—one F508del and one other variant—the team went a step further. Using highly specific molecular tests, they counted how much mRNA came from each gene copy separately. Instead of each allele contributing roughly half the total, they often found a strong imbalance, or “skewing”: the non-F508del allele typically produced much more mRNA than the F508del allele. In some patients, the F508del copy contributed only a tiny fraction of the total CFTR message. This uneven output appeared even among people sharing the same pair of mutations, suggesting that additional DNA changes near the gene, or broader regulatory mechanisms, shape how much message each copy produces.
Cell studies confirm built-in variability
To strip away some of the real-world complications of infections and inflammation in the airways, the scientists also grew nasal and bronchial cells from patients in the lab. There, too, they detected large differences in overall CFTR mRNA levels among people with the same genetic make-up. In a subset of cultured cells from compound heterozygous patients, expression from the two alleles was closer to balanced than in fresh nasal samples, underscoring how cell culture conditions and the body’s environment can change gene activity. Together, these observations support the idea that both total CFTR output and the relative contribution of each allele are flexible traits, not fixed by the DNA sequence of the main mutations alone.
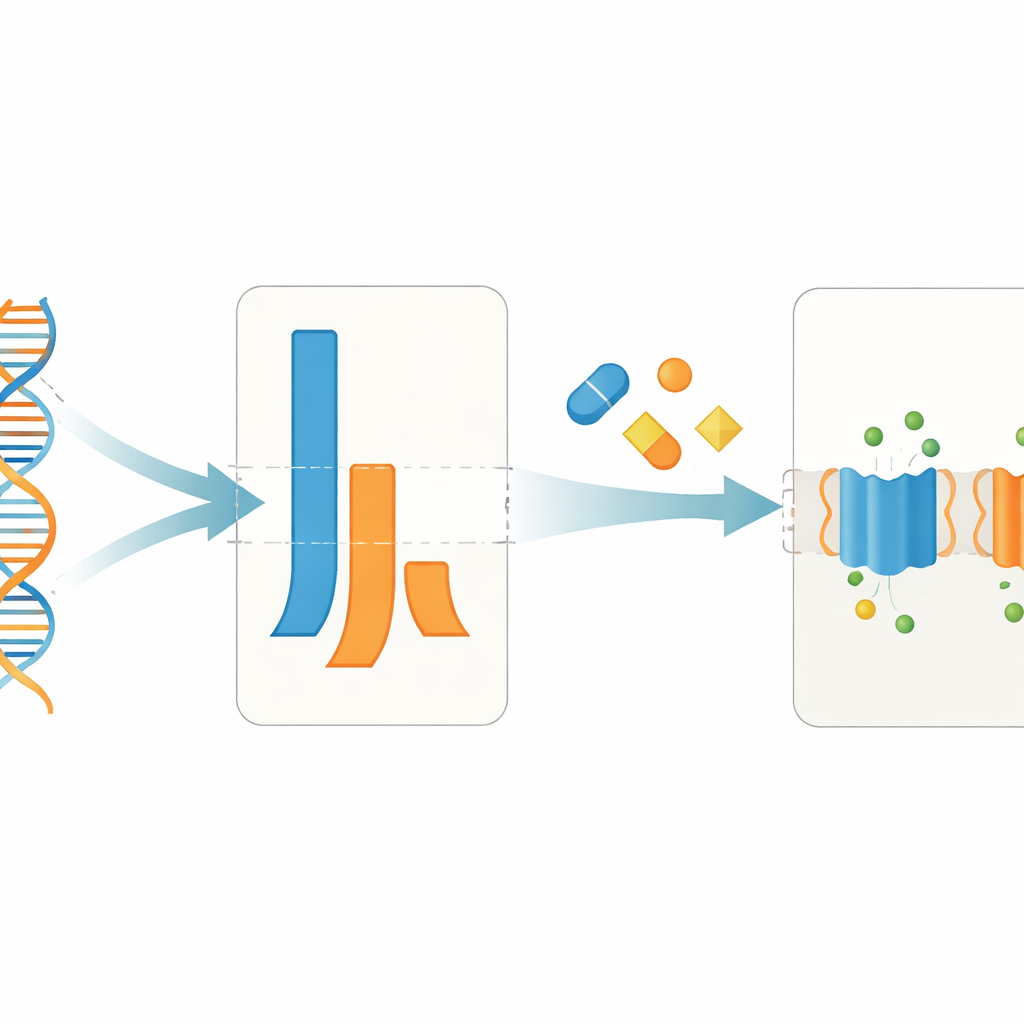
Modern CF drugs nudge the balance between gene copies
The study also examined a small group of patients before and after starting widely used CFTR “modulator” therapies, including the triple combination elexacaftor–tezacaftor–ivacaftor and a dual-drug regimen. Surprisingly, these drugs did not consistently raise or lower the overall amount of CFTR mRNA in nasal cells. Instead, they shifted which allele contributed more. After treatment, the F508del allele often produced a larger share of the total CFTR message, in some cases becoming the dominant source. This change in allelic balance occurred alongside the expected clinical improvements, such as better lung function and lower sweat salt levels, hinting that boosting message output from a rescued mutant protein may complement the drugs’ known effects on protein folding and function.
What this means for personalized treatment
For people living with cystic fibrosis, these findings highlight that DNA mutations are only part of the story. How much CFTR mRNA a person makes—and how that production is divided between their two gene copies—can vary widely and in ways not predicted by mutation type alone. This variation could help explain why patients with the same genotype experience different disease severity and respond differently to therapy. It also suggests that future personalized approaches may need to measure not just which CFTR mutations a person carries, but how actively each copy of the gene is being used, both before and during treatment. Understanding and eventually controlling this hidden layer of gene activity could help fine-tune therapies for CF and other single-gene diseases.
Citation: Freyberg, M., Bewig, M., Bampi, G.B. et al. Large variations in total and allele-specific transcript expression in a disease mutation-independent manner. Sci Rep 16, 7831 (2026). https://doi.org/10.1038/s41598-026-40624-1
Keywords: cystic fibrosis, CFTR, gene expression, allelic imbalance, CFTR modulators