Clear Sky Science · en
Cardiac output-guided vs. mean arterial pressure-guided hemodynamic management in craniotomy patients with cardiovascular disease: a randomized trial
Why this matters for brain surgery patients
People undergoing brain tumor surgery already face a stressful time, and the stakes are even higher when they also have heart disease. During the operation, doctors must carefully control blood flow and blood pressure to keep the brain safe. This study asked a simple but important question: is it better to guide treatment by how hard the heart is pumping blood, or by the pressure inside the arteries? The answer could change how anesthesiologists care for some of the most fragile patients in the operating room.
Two different ways to steer the circulation
When someone is under anesthesia for brain surgery, the medical team constantly watches monitors that display heart function and blood pressure. Traditionally, most teams have focused on mean arterial pressure, a measure of how hard blood is pressing against artery walls, as a stand-in for how well the brain is being supplied. But in people with long-standing high blood pressure or heart disease, that link can be unreliable. This trial compared the usual pressure-focused approach with a newer strategy that centers on cardiac output, the actual volume of blood the heart pumps each minute. In the cardiac output group, doctors adjusted fluids and drugs to keep overall blood flow above a set level and close to each patient’s own starting value; in the pressure group, they aimed to keep blood pressure within a safe range around baseline.
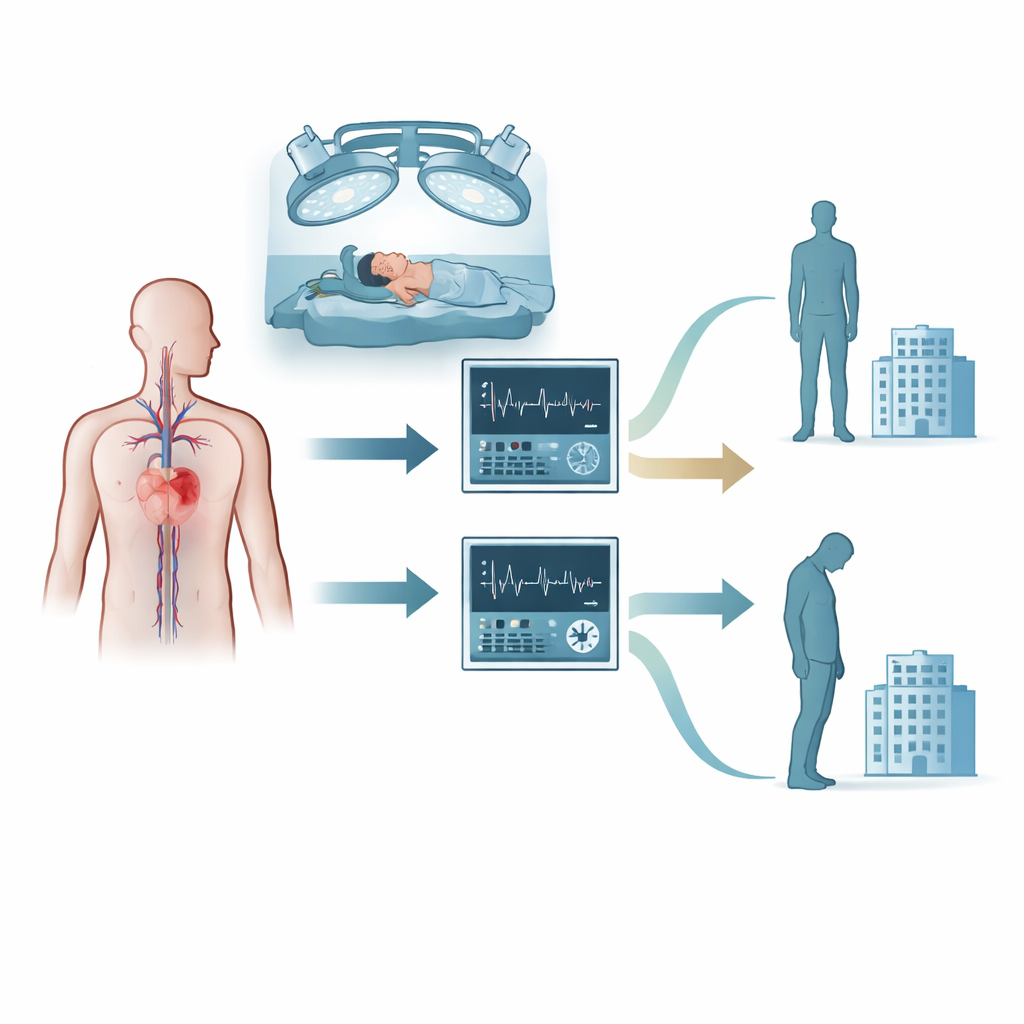
How the study was done
The researchers enrolled 202 adults scheduled for removal of tumors from the upper parts of the brain who also had diagnosed heart or blood vessel problems, such as hypertension, coronary artery disease, or weakened heart muscle. Everyone received standard anesthesia and brain tumor care, but they were randomly assigned to one of the two monitoring strategies during surgery. In the cardiac output group, a special monitor analyzed the pulse wave from an artery line to estimate how much blood the heart was moving. Clinicians then used a structured plan to decide when to give intravenous fluids, when to use drugs that strengthen the heartbeat, and when to tweak anesthesia depth. In the blood pressure group, the same monitor was covered so only routine measures like blood pressure and heart tracing were visible, and decisions were made using traditional targets.
What happened during and after surgery
The two strategies produced meaningfully different patterns in the operating room. Patients treated with cardiac output guidance received somewhat more intravenous fluid and more support from drugs that boost circulation. As a result, their hearts pumped more blood on average, and periods of low flow were almost eliminated, while blood pressure remained in a similar range to the pressure-guided group. After surgery, the team followed patients closely. Their main yardstick was a widely used five-point scale that reflects how independent and functional a person is three months after a brain injury or operation. They also tracked brain swelling on scans, new problems like weakness or seizures, heart complications, and how long patients stayed in the hospital.
Encouraging benefits, but not a final verdict
On the big-picture outcome at three months, the two approaches did not differ in a statistically firm way. Fewer people in the cardiac output group had an unfavorable recovery score, but the difference could still have been due to chance in a trial of this size. The more immediate findings, however, were striking. Only a small fraction of patients in the cardiac output group developed new brain swelling on early postoperative scans, compared with about one in ten in the pressure group. New neurological problems, such as weakness, confusion, or speech difficulties, were also less common. Patients managed with the cardiac output strategy typically left the hospital about a day earlier. These improvements appeared across many subgroups of patients, suggesting that better control of overall blood flow during surgery may ease the brain’s recovery, even if the long-term functional gains are harder to prove in a modest study.
What this means going forward
For readers, the key message is that how doctors choose to steer the circulation during complex brain surgery may influence the brain’s early healing. Paying attention to how much blood the heart is actually delivering, not just how high the pressure is, seems to reduce brain swelling and short-term complications in patients whose hearts are already under strain. At the same time, the trial was carried out in a single hospital with a limited number of participants, so its results are best viewed as promising clues rather than definitive proof. Larger, multi-center studies will be needed to confirm whether cardiac output–guided care truly improves long-term recovery and to refine the best targets. Still, this work highlights a shift in thinking: for the vulnerable brain, steady and adequate blood flow may matter more than pressure alone.
Citation: Chen, N., Yang, M., Li, R. et al. Cardiac output-guided vs. mean arterial pressure-guided hemodynamic management in craniotomy patients with cardiovascular disease: a randomized trial. Sci Rep 16, 9789 (2026). https://doi.org/10.1038/s41598-026-40615-2
Keywords: brain tumor surgery, cardiac output, blood pressure management, neurological recovery, hemodynamic monitoring