Clear Sky Science · en
Sustainable removal of Cd(II) and Cr(VI) from aqueous solution via agro-waste derived biochar
Turning Farm Waste into a Water Cleaner
Across the world, industrial activities release toxic metals into rivers and groundwater, threatening drinking supplies and aquatic life. At the same time, mountains of agricultural leftovers like corn cobs are burned or discarded. This study explores a way to tackle both problems at once: transforming corn cobs into a simple charcoal-like material, called biochar, that can pull dangerous metals out of water without expensive chemicals or complex technology.
Why Certain Metals in Water Are So Worrying
Two metals sit at the heart of this work: cadmium and chromium. They seep into water from processes such as electroplating, leather tanning, battery making, and dye production. In tiny doses they can already harm kidneys, bones, and the nervous system, and some forms are strongly linked to cancer. Because these metals do not break down and can build up in the food chain, even low levels in wastewater can become a long-term health risk. Many current treatment methods work, but they can be costly, energy-hungry, or create new waste streams like toxic sludge. A cheap, reusable filter made from waste biomass would therefore be highly attractive, especially in regions with limited resources.
From Corn Cob to Porous Filter
The researchers collected discarded corn cobs from farmland, washed them with a mild acid to remove impurities, dried them, and then heated them in the absence of oxygen in a process known as pyrolysis. This turned the pale, fibrous cobs into dark, porous biochar granules. Detailed imaging showed a rough, rod-like surface riddled with tiny channels where water and dissolved metals can enter. Chemical tests revealed that the surface is rich in oxygen-containing groups that can interact with metal ions. In other words, the team converted a low-value farm residue into a structured carbon sponge with many microscopic nooks and chemically active spots suited for trapping pollutants.
How Well the Corn-Based Sponge Captures Metals
To see how effective this material is, the team soaked measured amounts of biochar in water containing known levels of cadmium or hexavalent chromium and varied conditions such as contact time, temperature, acidity, and dosage. They found that the metal uptake rose quickly within the first hour and then leveled off as the available sites became filled. Under favorable conditions, a single gram of corn cob biochar could hold up to about 70 milligrams of cadmium or 55 milligrams of chromium, values that compare well with or surpass many other low-cost adsorbents reported in the literature. Mildly acidic water worked best: around pH 4.5 for cadmium and pH 5.0 for chromium, where the biochar surface charge and the dissolved forms of the metals line up to favor attraction rather than repulsion.
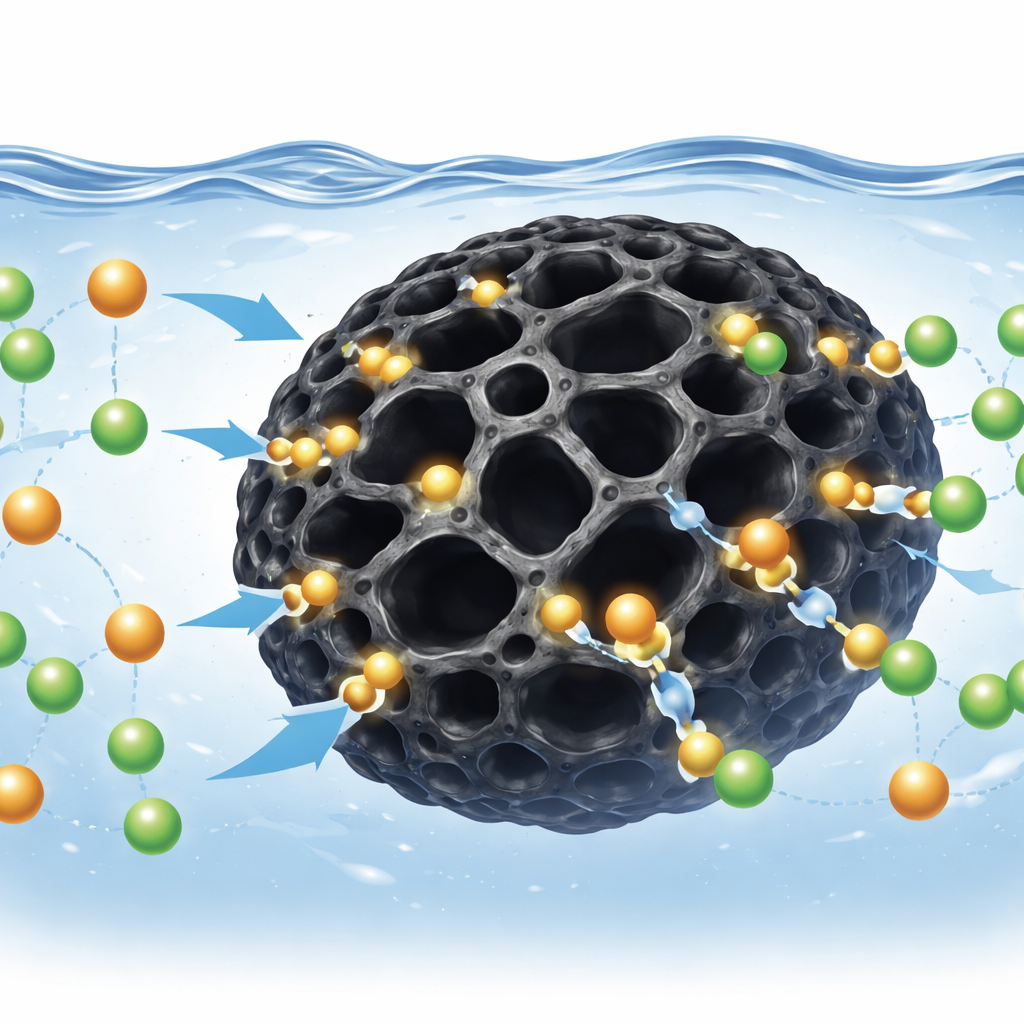
Peering into the Hidden Mechanism
By tracking how fast metals disappeared from solution and how the biochar’s surface bonds shifted after use, the authors inferred what happens at the microscopic level. The rate at which metals were captured matched a model where attachment is governed by interactions between the ions and specific surface sites, rather than by simple diffusion alone. Spectroscopic fingerprints showed that certain surface groups, such as those containing oxygen, changed when metals were present, pointing to a combination of electrostatic attraction, swapping of ions at the surface, and hydrogen-like bonding. Heat helped the process along: higher temperatures generally made adsorption more favorable, and thermodynamic analysis indicated that binding is spontaneous and mildly heat-absorbing. Even after several cycles of loading and chemical rinsing, the biochar retained much of its ability to capture metals, especially cadmium, hinting at practical reusability.
Real-World Robustness and Competition
Industrial wastewaters rarely contain a single pollutant, so the study also examined how other common ions, like calcium, magnesium, nitrate, and sulfate, affect performance. These extra species did compete with cadmium and chromium for space and charge on the biochar surface, lowering removal to some degree. Yet the corn cob material still removed a substantial share of the target metals, suggesting that it can function in complex, real-world mixtures rather than only in ideal laboratory solutions. The influence of these background ions also helped confirm that both charge-based attraction and ion exchange are central to how the system works.
A Simple Path to Cleaner Water
Overall, the work shows that an abundant farming by-product can be turned into an efficient, low-cost tool for cleaning water polluted with particularly stubborn and toxic metals. Without resorting to expensive chemical activation or high-tech processing, the corn cob biochar combines a porous structure with a chemically active surface that binds cadmium and chromium strongly under realistic conditions. For communities facing both agricultural waste and contaminated effluents, this kind of material offers a promising route toward more sustainable, locally sourced water treatment.
Citation: Din, S.U., Al-Ahmary, K.M., Al-Mhyawi, S.R. et al. Sustainable removal of Cd(II) and Cr(VI) from aqueous solution via agro-waste derived biochar. Sci Rep 16, 9792 (2026). https://doi.org/10.1038/s41598-026-40608-1
Keywords: biochar, corn cob, heavy metal removal, wastewater treatment, sustainable adsorbent