Clear Sky Science · en
Structural stability of symmetric bispecific antibodies: a case study showing potential compromise near linker regions
Why tiny connectors in new cancer drugs matter
Many of today’s most exciting cancer drugs are antibodies engineered to grab two different targets at once, helping immune cells home in on tumors. These "bispecific" antibodies can be more powerful than older single-target drugs, but they are also more complex molecules. This study takes a close look at one such experimental drug and asks a simple but crucial question: do the tiny protein connectors that hold its parts together stay intact under real-world manufacturing and storage conditions, or do they become weak links?
Building a two-handed antibody
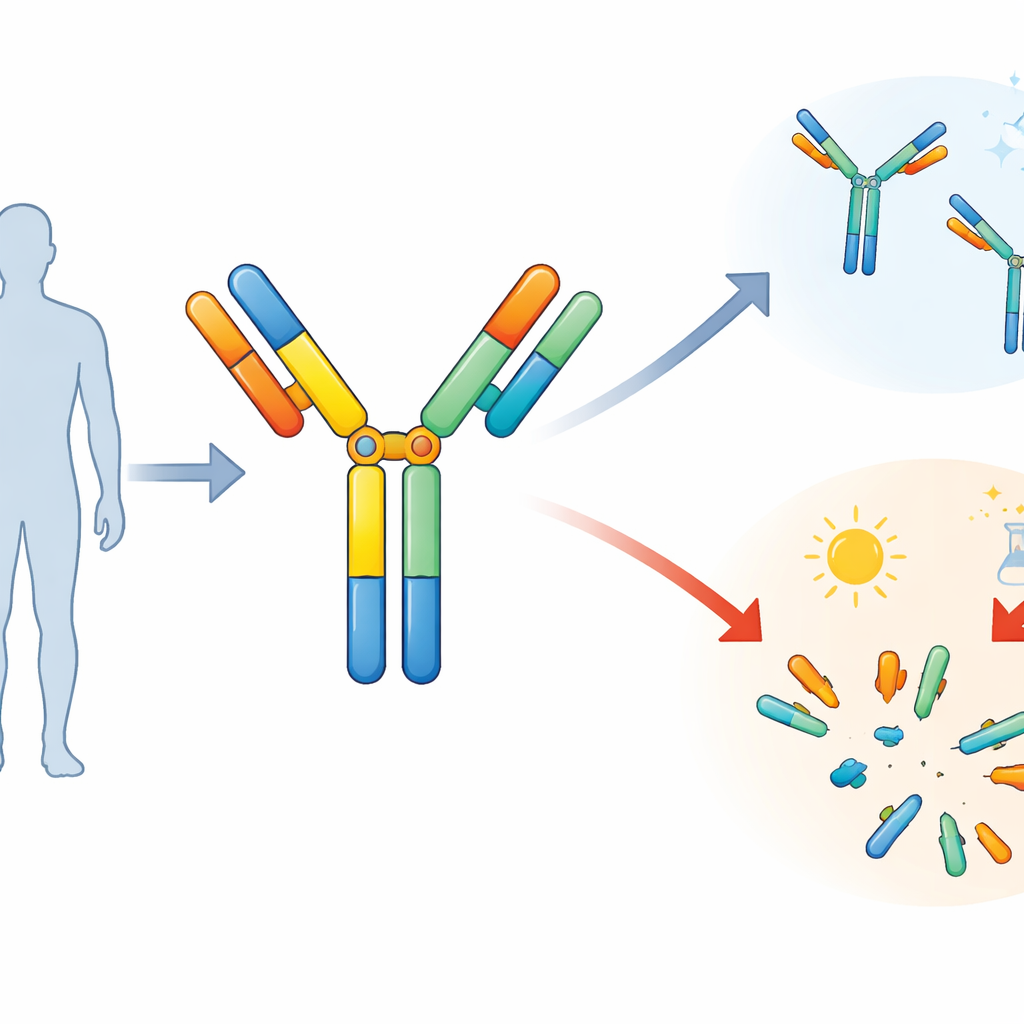
The researchers focused on a symmetric bispecific antibody designed to bind a tumor marker called HER2 and an immune-cell receptor called CD3. Symmetric here means the molecule is built in a balanced, mirror-like way, with four gripping tips instead of the usual two. To achieve this, the team stitched together familiar antibody parts with short stretches of flexible protein known as linkers. These linkers act like hinges or spacers, allowing the binding regions to adopt the right positions so the drug can grab both the cancer cell and the T cell at the same time.
The weak spots under heat and harsh conditions
To test how sturdy this design really is, the team deliberately stressed the antibody. They heated it for weeks and placed it in liquids with different acidity (pH) and salt levels, mimicking conditions the molecule might encounter during purification, formulation, or long-term storage. Using a technique that separates molecules by size, they watched as the share of intact antibody slowly dropped and the amount of smaller fragments rose, especially at higher pH. Salt alone had only a modest effect, but when high pH and high salt were combined, fragmentation increased sharply, signaling that certain parts of the molecule were coming apart.
Zooming in on the breaking points
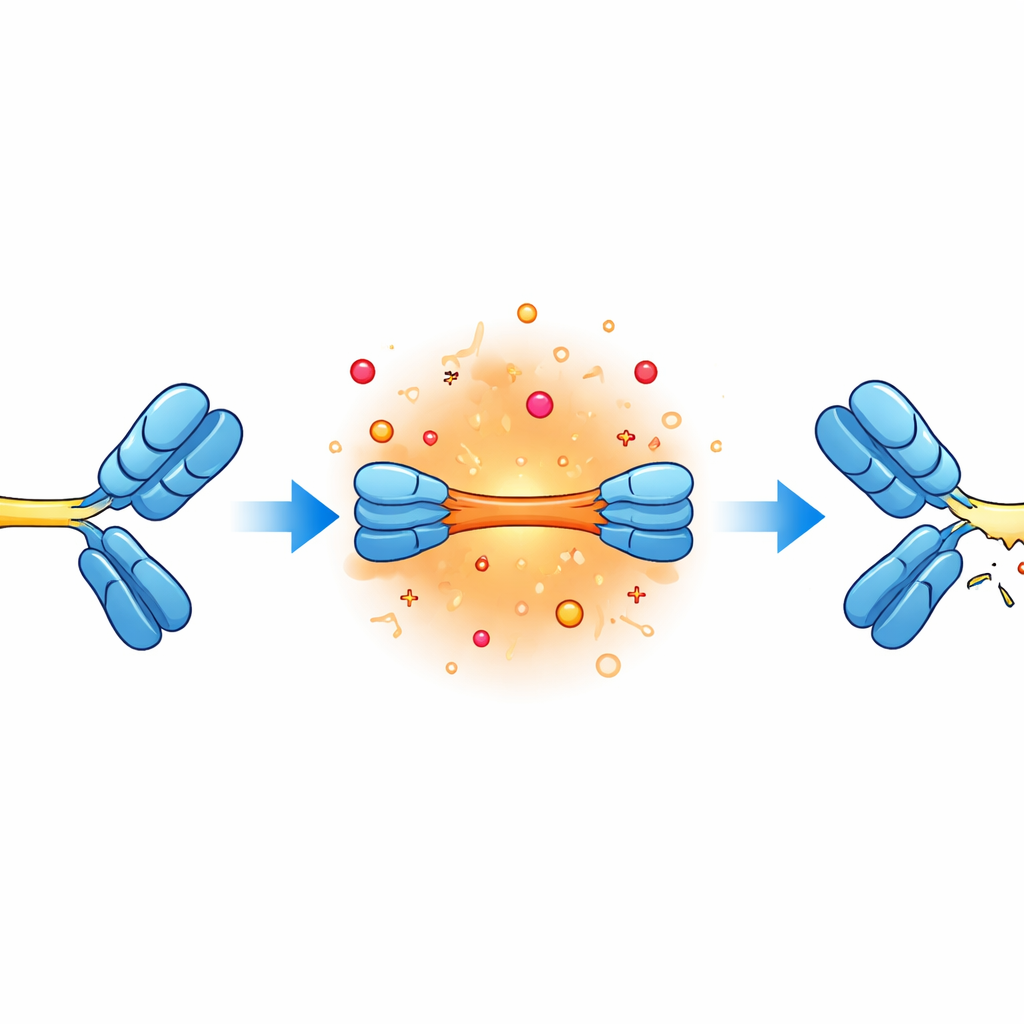
The next step was to figure out exactly where the breaks were occurring. The researchers collected the various fragments and analyzed their masses with high-resolution mass spectrometry, essentially weighing and reconstructing the pieces like a molecular jigsaw puzzle. This revealed that two specific linkers—short sequences often referred to as G4S and G4—were especially prone to being cut. These flexible connectors sit between the main antibody arms and the added binding segments. Under alkaline, salty, and warm conditions, the peptide bonds in these regions were clipped in a stepwise manner, creating a series of predictable fragments along the linker chains.
Not just the connectors: other bonds at risk
The analysis also showed damage in other vulnerable spots. Some breaks occurred near certain asparagine building blocks, which are known to undergo slow chemical changes at high pH that can eventually lead to chain scission. In addition, bonds involving sulfur-containing cysteine residues, which normally help hold the heavy and light chains of antibodies together, were disrupted. This led to detached light-chain pieces and different chemically modified variants. While these types of changes are familiar from studies of standard monoclonal antibodies, their appearance near the linker regions suggests that the added flexibility of the bispecific design may expose neighboring bonds to extra stress.
Design lessons for safer and sturdier drugs
For non-specialists, the key takeaway is that making antibodies more versatile by adding extra binding arms is not simply a matter of clever genetic engineering; it also requires careful attention to the small connector regions that tie everything together. In this case, the commonly used flexible linkers that join the functional parts of the bispecific antibody turned out to be structural weak points under realistic stress conditions. The study’s conclusion is not that bispecific antibodies are unsafe, but that their stability depends strongly on how these linkers are composed and placed. Future designs will need to optimize linker length, sequence, and local environment so that next-generation antibody therapies can deliver their full clinical promise without falling apart during manufacture, storage, or treatment.
Citation: Ingavat, N., Kok, Y.J., Dzulkiflie, N. et al. Structural stability of symmetric bispecific antibodies: a case study showing potential compromise near linker regions. Sci Rep 16, 9715 (2026). https://doi.org/10.1038/s41598-026-40607-2
Keywords: bispecific antibodies, protein stability, linker design, antibody engineering, biopharmaceutical development