Clear Sky Science · en
Theoretical analysis of low-power optogenetic suppression of action potentials in human ventricular cardiomyocytes expressed with potassium-selective channelrhodopsins
A gentler light-based way to calm a racing heart
Fast, chaotic heart rhythms can trigger fainting, stroke, or sudden death. Today’s treatments—powerful drugs, implanted defibrillators, or high-energy shocks—can be life-saving but also painful and imprecise. This study explores a very different idea: using faint flashes of light and specially engineered proteins to quietly nudge heart cells back into a safe, steady rhythm, while using far less energy than current approaches.
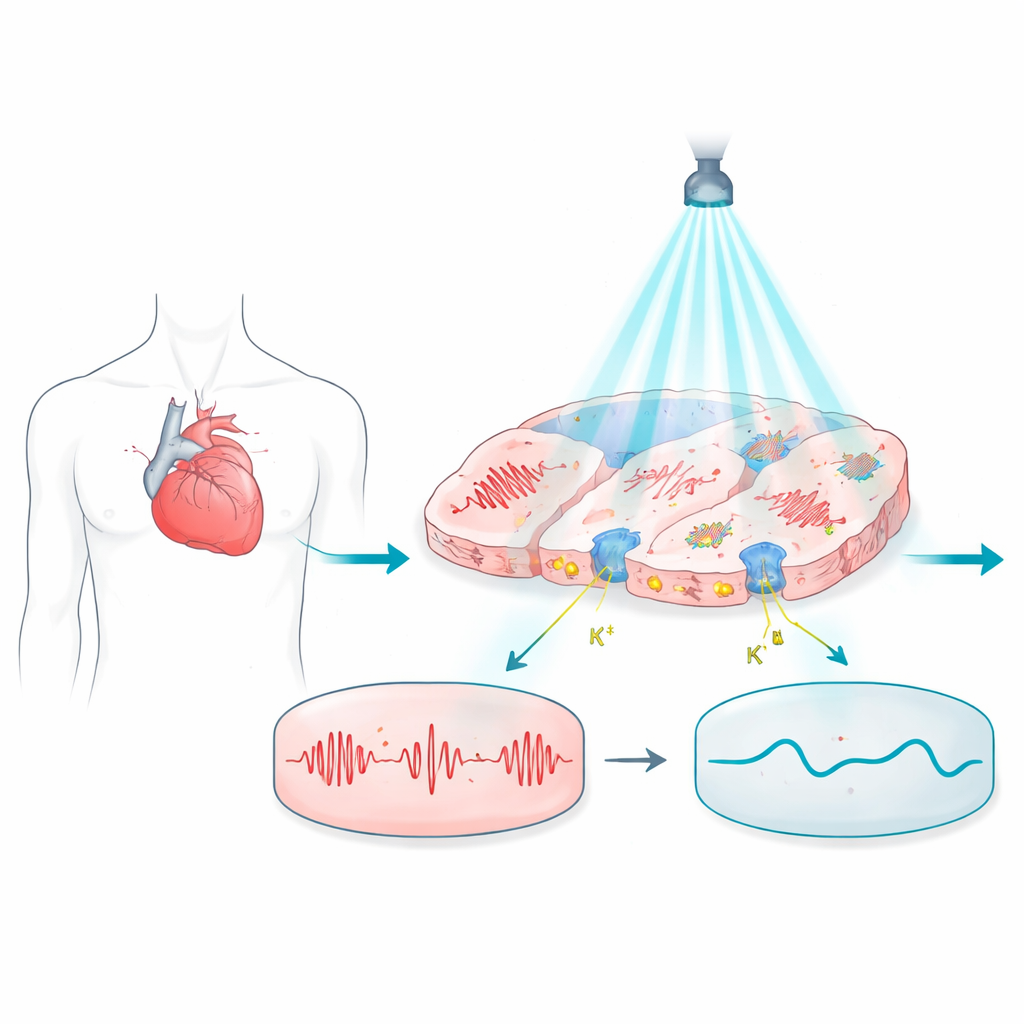
Shining light on problem heartbeats
The work builds on optogenetics, a technique in which cells are equipped with light-sensitive proteins so their electrical activity can be controlled with flashes of light. In the heart, these proteins can in principle replace metal electrodes, offering contact-free, pain-free control. But most light-gated proteins used so far tend to push the cell’s voltage upward, toward an excited state, which makes it hard to hold the cells quietly at rest or to fine-tune how long each heartbeat lasts. That limits their usefulness for safely turning off dangerous rhythms or correcting electrical disorders that depend on subtle timing.
New light switches tuned to the heart’s “resting setting”
Recently discovered proteins called potassium-selective channelrhodopsins, including WiChR and HcKCR1, promise to fix this mismatch. Unlike older proteins that pass a mixture of charged atoms, these channels strongly prefer potassium and naturally pull the cell’s voltage toward the same negative level it would adopt at rest. The authors built detailed computer models of human ventricular heart cells that express these newer channels and compared them with two well-known, more excitatory opsins, ChR2(H134R) and ChRmine. By simulating how these cells respond to different colors and intensities of light, they could safely explore conditions that would be difficult or time‑consuming to test in real hearts.
Soft light, strong control
The simulations show that potassium-selective channels provide far more economical and stable control. WiChR, in particular, could completely shut down action potentials—the brief electrical spikes that trigger each heartbeat—using light intensities hundreds to thousands of times lower than those needed for many earlier tools. Under continuous light, WiChR and HcKCR1 kept the cell’s voltage near its normal resting level, effectively holding the cell in a quiet, safe state. By contrast, ChR2 and ChRmine tended to push the voltage toward more positive values, sometimes blocking activity but only after forcing the cell into a stressed, overexcited condition. WiChR also worked well with brief light pulses, reliably preventing heart-cell spikes beat after beat, suggesting it could keep up with rapid heart rhythms without overheating tissue or wasting power.
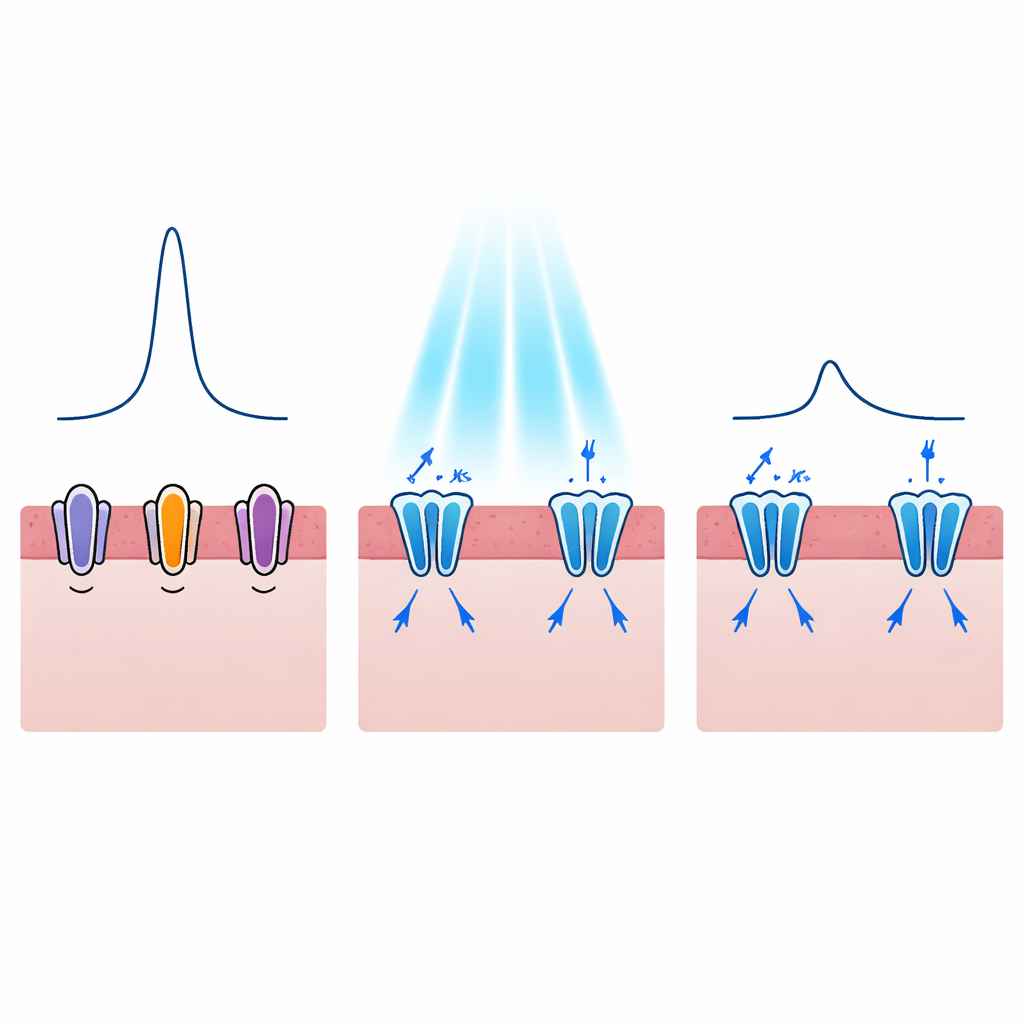
Reshaping each heartbeat, not just stopping it
Dangerous arrhythmias are often linked not only to whether a heart cell fires, but also to how long it stays excited. In disorders like long QT syndrome, the electrical pulse of each beat is stretched out, increasing the chance of spiraling into fatal rhythms. The authors therefore asked whether light-activated potassium channels could shorten this pulse in a controlled way. Their models showed that turning on WiChR or HcKCR1 during the plateau phase of the action potential created strong outward potassium currents that pulled the voltage back down sooner. As light levels increased, the duration of the electrical pulse dropped from about 300 milliseconds to roughly half that value, and this effect could be achieved even with very short light flashes. WiChR tended to produce longer-lasting quiet periods, while HcKCR1 offered quicker recovery once the light was turned off, hinting at different potential clinical uses.
From computer models to future therapies
Overall, the study concludes that potassium-selective light-gated channels, especially WiChR, are promising tools for gentle, low-power control of the heart. They can both silence runaway electrical activity and trim overly long heartbeats, all while keeping the cell’s voltage close to its natural resting state. Although the results come from detailed single-cell simulations rather than whole-heart experiments, they provide quantitative guidance for how much light might be needed, when it should be applied, and which protein variants are best suited for different goals. In the long run, this line of work points toward a future in which cardiologists might use finely tuned beams of light, rather than painful shocks, to prevent or stop life‑threatening arrhythmias.
Citation: Dixit, N., Pyari, G. & Roy, S. Theoretical analysis of low-power optogenetic suppression of action potentials in human ventricular cardiomyocytes expressed with potassium-selective channelrhodopsins. Sci Rep 16, 9765 (2026). https://doi.org/10.1038/s41598-026-40578-4
Keywords: cardiac optogenetics, arrhythmia suppression, potassium channelrhodopsins, action potential duration, long QT syndrome