Clear Sky Science · en
In ovo sexing and genotyping using PCR techniques: a contribution to the 3R principles in chicken breeding
Why what happens inside an egg matters
Every year, millions of male chicks are killed shortly after hatching because they are not useful for egg production. At the same time, research labs around the world hatch more animals than they actually need, simply because they cannot tell an embryo’s sex or genetic makeup early enough. This study presents a practical way to “read” the sex and genes of a chick while it is still developing inside the egg, using standard DNA tests. By doing this early and gently, the method aims to avoid hatching surplus animals and to reduce suffering, without requiring expensive industrial equipment.
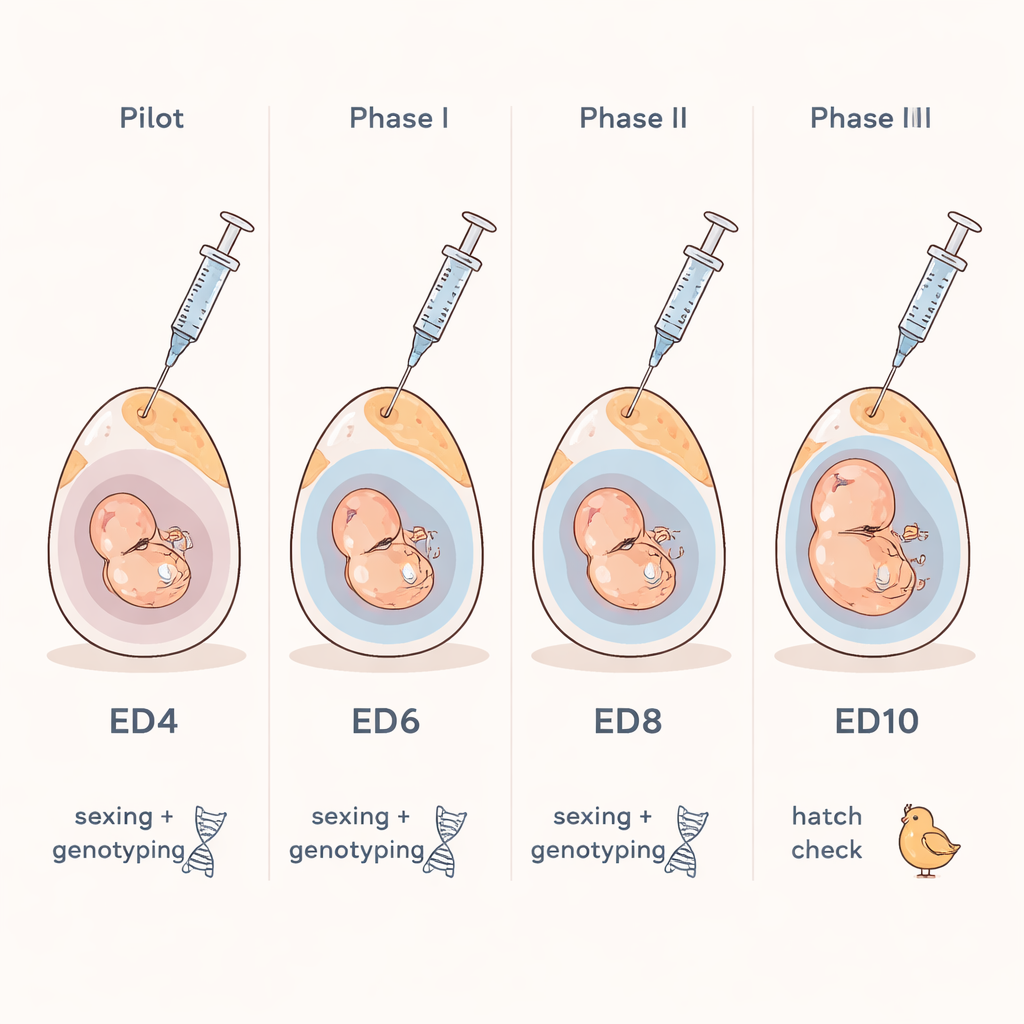
Looking inside the egg without opening it
The researchers focused on a simple question: can we safely take a tiny fluid sample from a fertilized chicken egg and use it to identify the embryo’s sex and key genetic traits? They drew on a concept familiar from human medicine, where doctors test fluid around a fetus to look for genetic conditions. In chickens, a thin, fluid-filled sac called the allantois forms around the embryo as it grows. By day seven of incubation, this sac holds enough fluid that a fine needle can reach it without touching the embryo itself. The team used a bright light to locate the air space at the blunt end of the egg, made a pin‑sized hole in the shell, and drew out just a few drops of fluid with a tiny syringe.
From a few drops to a genetic fingerprint
The fluid drawn from eggs early in development contains only minute amounts of DNA. To work around this, the scientists first amplified all the genetic material in each sample, creating many copies without changing its content. They then applied two routine laboratory tests: a standard polymerase chain reaction (PCR) and a variant called Kompetitive Allele Specific PCR (KASP). Both tests can distinguish between the sex chromosomes of male and female chicks and can also spot specific genetic markers, such as the mutation that causes blue eggshells or the presence of a laboratory‑inserted gene in specialized research lines. Across more than 800 eggs from several breeds—including commercial layers, colorful Araucana crosses, and a genetically modified research line—these methods produced correct identifications in roughly 92–100% of cases.
Finding the safest moment to sample
Sampling too early risks damaging fragile structures that are vital for the embryo, while sampling too late leaves little time to act on the results. To pinpoint the ideal window, the team first grew embryos in a shell‑less culture to watch how the membranes and fluid spaces expand over time. They then ran a series of staged trials in intact eggs. Fluid could be collected from day four onward, but success rates for DNA testing were lower at these early days, and the procedure was technically more difficult. When they compared hatch rates between punctured and untouched control eggs, they found that day seven struck the best balance: there was ample clear allantoic fluid, DNA tests worked reliably, and most embryos survived to hatch. At this point, there are still several days before the likely onset of pain perception in the chick embryo, giving an ethical buffer for decisions.
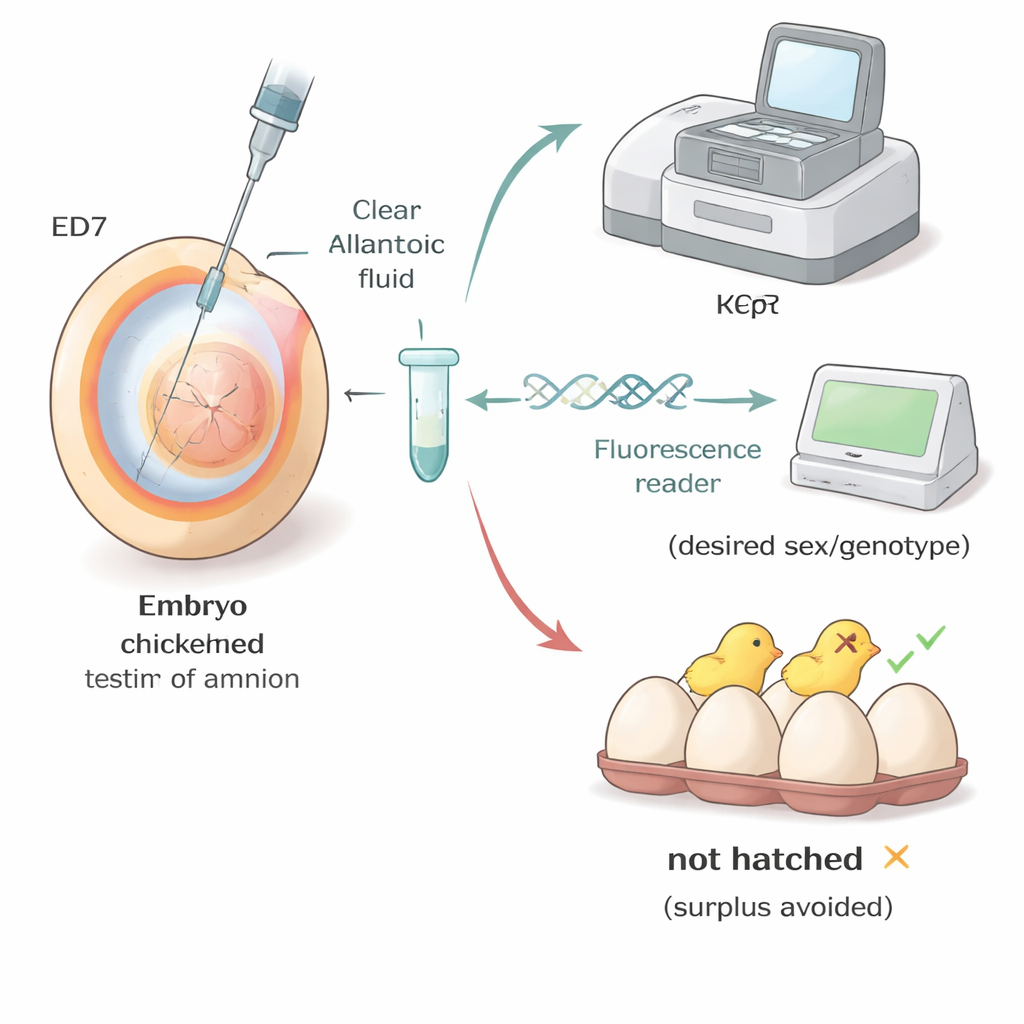
Putting the method to work in real flocks
The researchers tested their workflow under conditions similar to those in breeding facilities and university labs. In commercial brown and white layer lines, eggs sampled between days four and seven showed that hatchability generally improved when sampling was done later, with the best results again at day seven. In a specialized “surrogate host” line used for advanced genetics, the team went a step further: they sexed and genotyped embryos at day seven, then chose only those with the desired sex and genetic combination to continue to hatch. Almost all selected embryos hatched into healthy chicks that grew as expected, showing that early selection can sharply reduce the number of unwanted animals without compromising chick quality.
What this means for animal welfare and research
To a non‑specialist, the core message is straightforward: with a pin‑prick and routine lab tools, it is now possible to know, while the chick is still safely inside the egg, whether it is male or female and whether it carries particular genetic traits. The study shows that doing this around day seven of incubation is both technically reliable and gentle enough that most embryos survive. By allowing unwanted embryos to be removed before they are likely capable of feeling pain, the approach supports the widely accepted 3R principles: replacing, reducing, and refining the use of animals in science. For hatcheries and research labs alike, this technique offers a realistic path to fewer surplus animals, better use of resources, and higher welfare standards—without the need for complex industrial machines.
Citation: Dierks, C., Förster, A., Meunier, D. et al. In ovo sexing and genotyping using PCR techniques: a contribution to the 3R principles in chicken breeding. Sci Rep 16, 7464 (2026). https://doi.org/10.1038/s41598-026-40562-y
Keywords: chicken embryo, in ovo sexing, PCR genotyping, animal welfare, 3R principles