Clear Sky Science · en
Genomic characterization of multidrug-resistant Escherichia coli strains identified from patients with urinary tract infection in Egypt
Why these stubborn infections matter
Urinary tract infections are among the most common reasons people visit hospitals and clinics. Many are caused by the bacterium Escherichia coli and are usually treatable with standard antibiotics. But around the world, including in Egypt, some E. coli strains have become so resistant to medicines that doctors are running out of options. This study takes a close genetic look at two such hard‑to‑treat strains from Egyptian patients to understand how they dodge multiple drugs and how easily their resistance might spread.
Two patients, two dangerous germs
The researchers focused on two E. coli strains, named UPE7 and UPE139, isolated from women with urinary tract infections at a hospital in Mansoura, Egypt. When tested against a wide range of antibiotics, both strains withstood many of the drugs doctors would normally prescribe, including several last‑resort medicines. They showed resistance to common penicillins and cephalosporins, strong carbapenems, widely used fluoroquinolones, and other drug classes. This pattern placed them firmly in the category of multidrug‑resistant bacteria, making infections difficult and costly to treat and increasing the risk of complications.
Reading the bacteria’s DNA playbook
To find out how these strains became so tough, the team sequenced their entire genomes using next‑generation DNA technology. They then used specialized computer tools to scan the genomes for genes linked to antibiotic resistance and for features that help the bacteria cause disease. Both strains carried numerous resistance genes that matched their behavior in the lab. For example, they harbored multiple “beta‑lactamase” genes that break down key antibiotics, as well as genes that protect against drugs like aminoglycosides, tetracyclines, macrolides, sulfonamides, and trimethoprim. Changes in core DNA‑handling enzymes, the targets of fluoroquinolone drugs, aligned with their strong resistance to levofloxacin. Both strains also possessed an operon known to modify their outer surface in ways that blunt the action of polymyxin antibiotics.
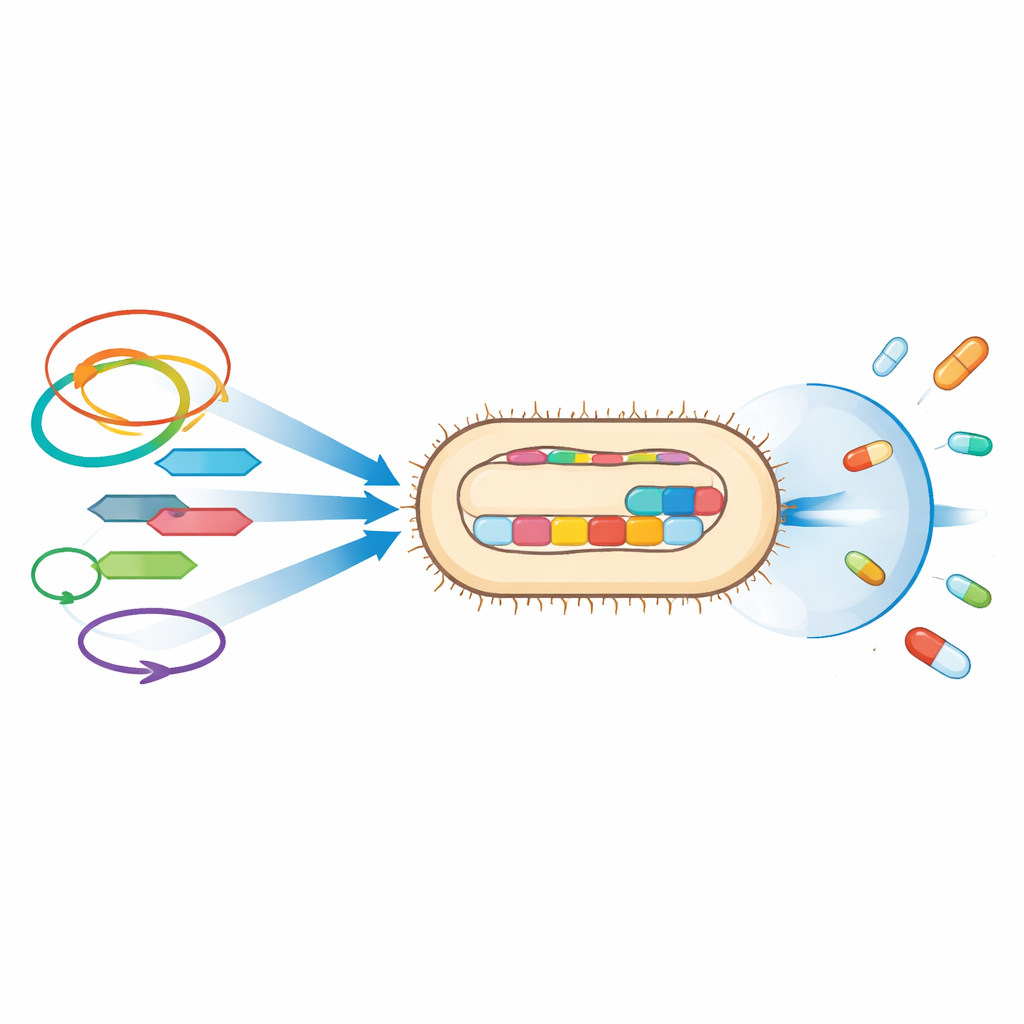
Hidden helpers that spread resistance
Beyond individual resistance genes, the study highlights the genetic vehicles that move these traits around. Many of the resistance genes sat next to mobile genetic elements such as insertion sequences and transposons—small DNA units that can hop between locations—and on plasmids, circular DNA molecules that bacteria share with one another. For instance, a widely distributed extended‑spectrum beta‑lactamase gene, CTX‑M‑15, was linked to such mobile elements in both strains, while UPE139 also carried OXA‑244, a carbapenem‑degrading enzyme embedded between jumping DNA segments on the chromosome. The same plasmid types found here have been tied elsewhere to clusters of resistance genes, raising concern that these strains can pass their defenses to other bacteria in hospitals and the community.
Tools for infection and survival
The genetic analysis also uncovered many traits that help these E. coli strains colonize the urinary tract and damage host tissues. Both UPE7 and UPE139 carried multiple adhesion structures that let them stick to cells and form biofilms, protective communities that are harder for drugs and the immune system to clear. They encoded toxins such as hemolysin, which can damage host cells, and iron‑scavenging systems that allow the bacteria to thrive in the iron‑poor environment of the body. UPE139, in particular, possessed additional genes linked to capsule formation, invasion, and immune evasion, suggesting it may have an especially strong ability to persist and spread within patients.
What this means for patients and doctors
By combining traditional drug‑sensitivity testing with full genome sequencing, this study shows how a small number of genes and mobile elements can turn ordinary urinary bacteria into formidable, multidrug‑resistant pathogens. It also reveals that these dangerous traits sit on DNA segments well suited for transfer to other strains, raising the risk of wider local and regional spread. For patients, this means some common infections may become harder to cure. For clinicians and health officials, the work underscores the need for ongoing genomic surveillance, careful antibiotic use, and broader studies of many more isolates to track high‑risk clones, fine‑tune treatment guidelines, and slow the march of resistance.
Citation: El Halfawy, N.M., Gouda, M.K., Elgayar, F.A. et al. Genomic characterization of multidrug-resistant Escherichia coli strains identified from patients with urinary tract infection in Egypt. Sci Rep 16, 8958 (2026). https://doi.org/10.1038/s41598-026-40536-0
Keywords: antibiotic resistance, urinary tract infection, Escherichia coli, genome sequencing, mobile genetic elements