Clear Sky Science · en
Identification of an age-related monocyte-derived macrophages characteristic genes-based signature for high-grade diffuse glioma
Why this brain tumor study matters
Brain tumors called diffuse gliomas are among the deadliest cancers in adults, and outcomes are especially poor for older patients. Doctors know that immune cells inside tumors can either help fight cancer or secretly support its growth, but they lack simple tools to tell which patients might benefit from therapies that target these cells. This study explores how a particular group of immune cells linked to aging might worsen aggressive brain tumors, and builds a gene-based score that could help guide treatment decisions.
Immune cells that move into the brain
Our brains contain resident immune cells, but tumors also recruit reinforcements from the bloodstream. These incoming cells, called monocyte-derived macrophages, can settle around the tumor and shape its local environment. Earlier work showed that some of the most aggressive brain tumors, especially in older adults, tend to be packed with these cells and that patients with higher levels often live for a shorter time. The challenge is that directly measuring these cells in every patient requires complex single-cell analyses that are not practical in routine care.
Finding a genetic fingerprint of harmful immune cells
The researchers turned to large public datasets of brain tumor samples that include both clinical information and patterns of gene activity. They compared tumors from younger and older adults to find genes that were switched on more strongly with age, then intersected these with genes known from single-cell studies to mark monocyte-derived macrophages. This process produced a set of nearly one hundred genes that seemed to capture how these immune cells change with aging inside high-grade diffuse gliomas. Because so many genes overlap and interact, they used statistical methods to distill this long list down to a few that carry most of the predictive power.
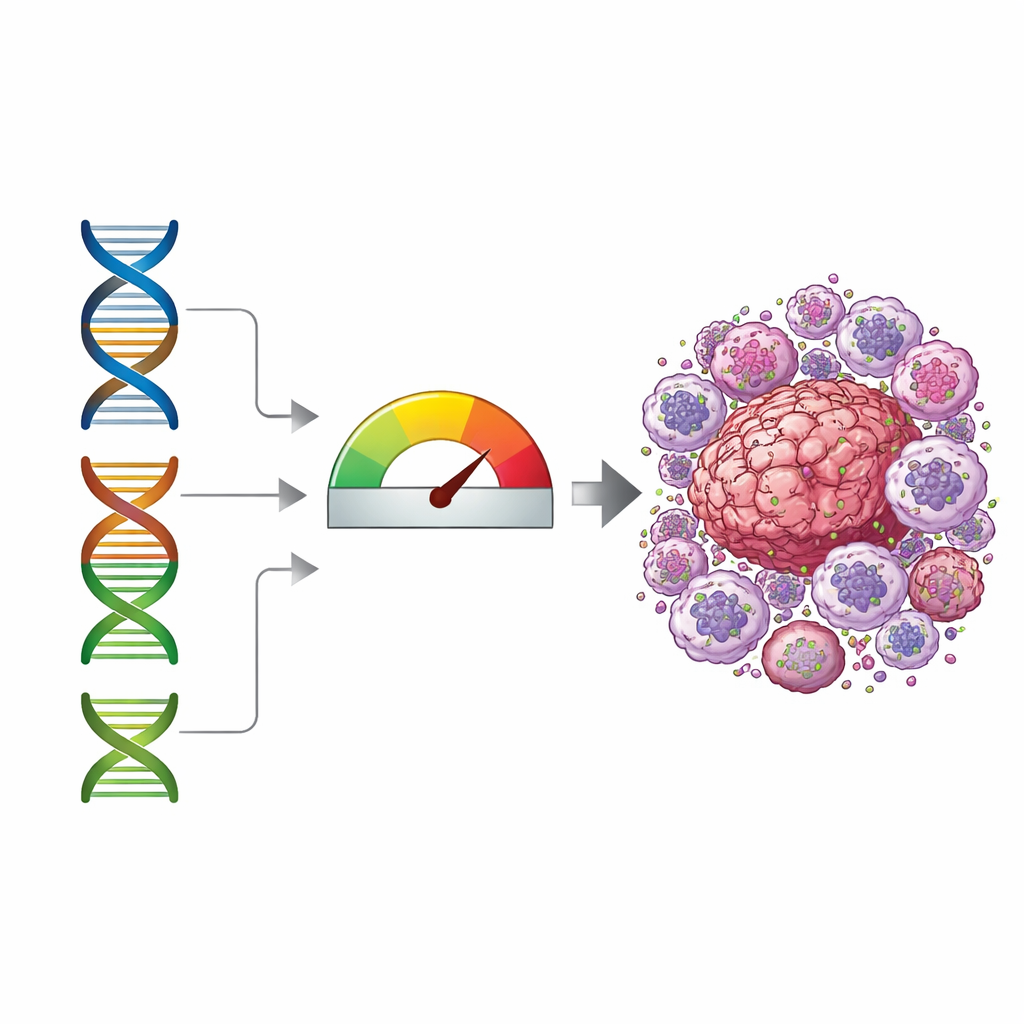
A three-gene score that tracks risk
From this analysis, three genes emerged as key markers: G0S2, PLAUR, and SPAG4. The team built a "risk score" by combining how strongly each of these genes was expressed in a tumor. They first tested this score in a large U.S. patient cohort and found that people with higher scores tended to have shorter overall survival, even after accounting for age and common tumor mutations. They then checked the result in an independent Chinese cohort and again saw that the score separated patients into higher- and lower-risk groups. While the score is not a perfect yes-or-no test, it behaves as a continuous indicator that reflects how strongly the tumor appears to be influenced by these macrophage-like cells.
Linking the score to the tumor’s immune climate
To understand what the score represents biologically, the authors examined patterns of immune cell infiltration. Tumors with higher scores showed more macrophages of multiple types and more cells associated with suppressing immune responses, such as regulatory T cells and certain myeloid cells. At the same time, cell types that usually mount active anti-tumor attacks, such as natural killer cells and specific T cell subsets, tended to be less prominent. This suggests that the three-gene score is not just tracking age, but is capturing a shift toward an immune environment that protects the tumor rather than the patient.
Age, sex, and who might benefit most
The study also explored how the score relates to other known tumor features. It tends to be lower in tumors carrying mutations in the IDH gene and several other markers that are usually linked to better outcomes, and higher in more malignant, IDH-wildtype tumors. The score rises with patient age and still distinguishes risk even among older patients alone. When the team split the data by sex, they found that the individual genes contributed differently in men and women, but the combined score remained a strong predictor in both groups. These patterns hint that older patients, especially those with aggressive tumors and high scores, may be the ones most likely to benefit from treatments that target these macrophage populations.
What this means for patients and future care
In everyday terms, this work proposes a bloodless "thermometer" for a hidden part of brain tumors: the aging, invasive immune cells that can help cancers thrive. By reading out the activity of just three genes from a tumor sample, clinicians could estimate how heavily the tumor depends on these cells and how strongly its immune environment is tilted toward protecting the cancer. While more testing is needed before it can guide individual treatment choices, this gene-based score offers a practical way to identify patients who might gain the most from future drugs that disarm or reduce these harmful immune cells, particularly older adults with the most aggressive forms of diffuse glioma.
Citation: Chen, W., Xie, Y., Pan, H. et al. Identification of an age-related monocyte-derived macrophages characteristic genes-based signature for high-grade diffuse glioma. Sci Rep 16, 10328 (2026). https://doi.org/10.1038/s41598-026-40485-8
Keywords: glioblastoma, brain tumor immunity, tumor-associated macrophages, aging and cancer, gene risk signature