Clear Sky Science · en
Synthesis and biological evaluation of 6-hydroxychromone based thiosemicarbazones as potential antidiabetic and antioxidant agents
New Molecules for a Growing Health Challenge
Type 2 diabetes and diseases linked to oxidative stress, such as heart disease and liver damage, are rising worldwide. Many existing drugs struggle to control blood sugar without side effects, and they often ignore the damage caused by harmful "oxidative" molecules inside our cells. This study explores a new family of laboratory-made compounds designed to tackle both problems at once—helping to manage blood sugar and neutralize damaging reactive molecules—offering a glimpse of what next-generation antidiabetic medicines could look like.
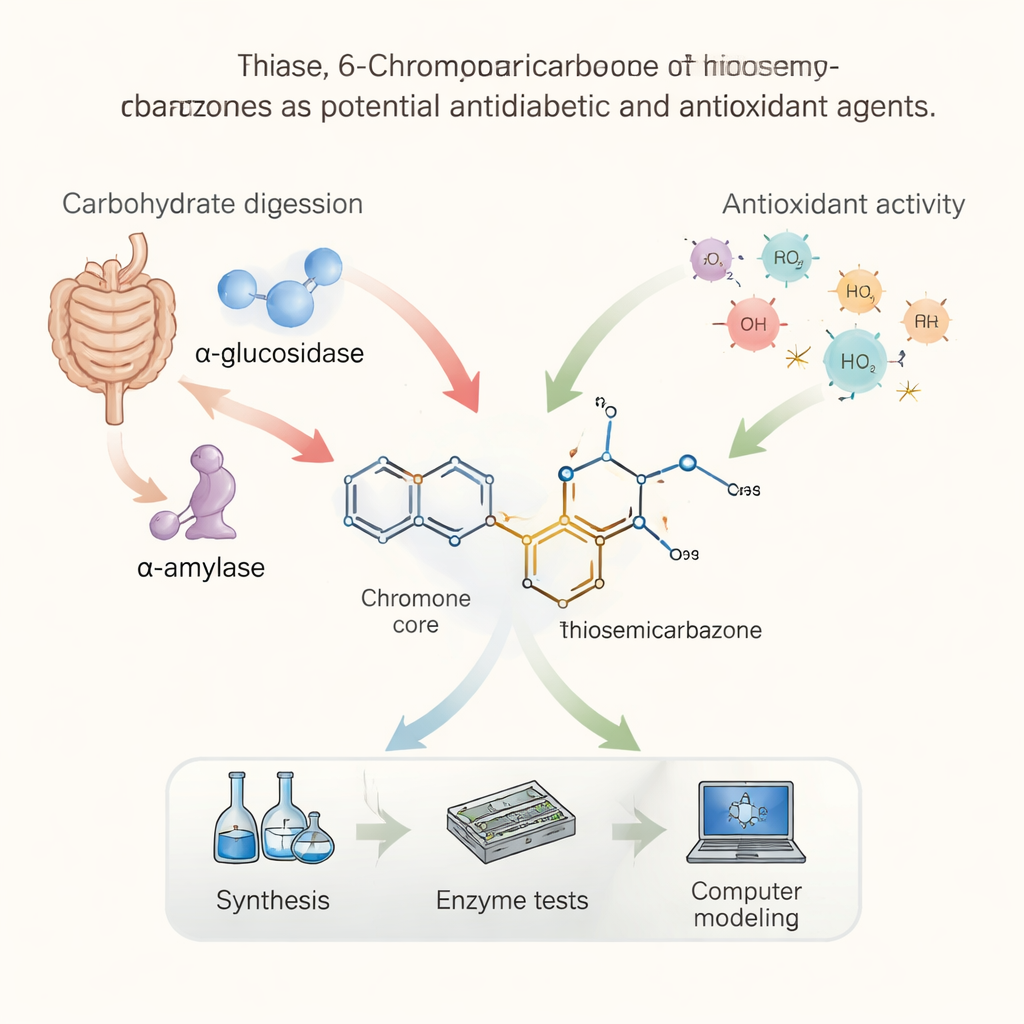
A Two-Front Attack on Blood Sugar
When we eat carbohydrates, our digestive system uses enzymes to chop long chains of starch into simple sugars that enter the bloodstream. Two key enzymes, called α-glucosidase and α-amylase, help drive this process. Current diabetes drugs sometimes work by slowing these enzymes down so that sugar is released more gradually. The researchers in this study set out to build new compounds that could block both enzymes more powerfully than today’s medicines, with the hope of smoothing out post-meal blood sugar spikes that are especially harmful in type 2 diabetes.
Designing a Hybrid Chemical Scaffold
The team combined two well-known chemical backbones that already show medical promise. One, called a chromone, is related to structures found in many plant-based natural products with anti-inflammatory and antidiabetic effects. The other, a thiosemicarbazone, is a versatile unit known for strong biological activity, including blood-sugar lowering and antioxidant properties. By fusing these two pieces into a single “hybrid” molecule and varying the attached side groups, the researchers created a family of sixteen related compounds, each a small chemical tweak on the same basic design.
Stronger Than Standard Diabetes and Antioxidant Drugs
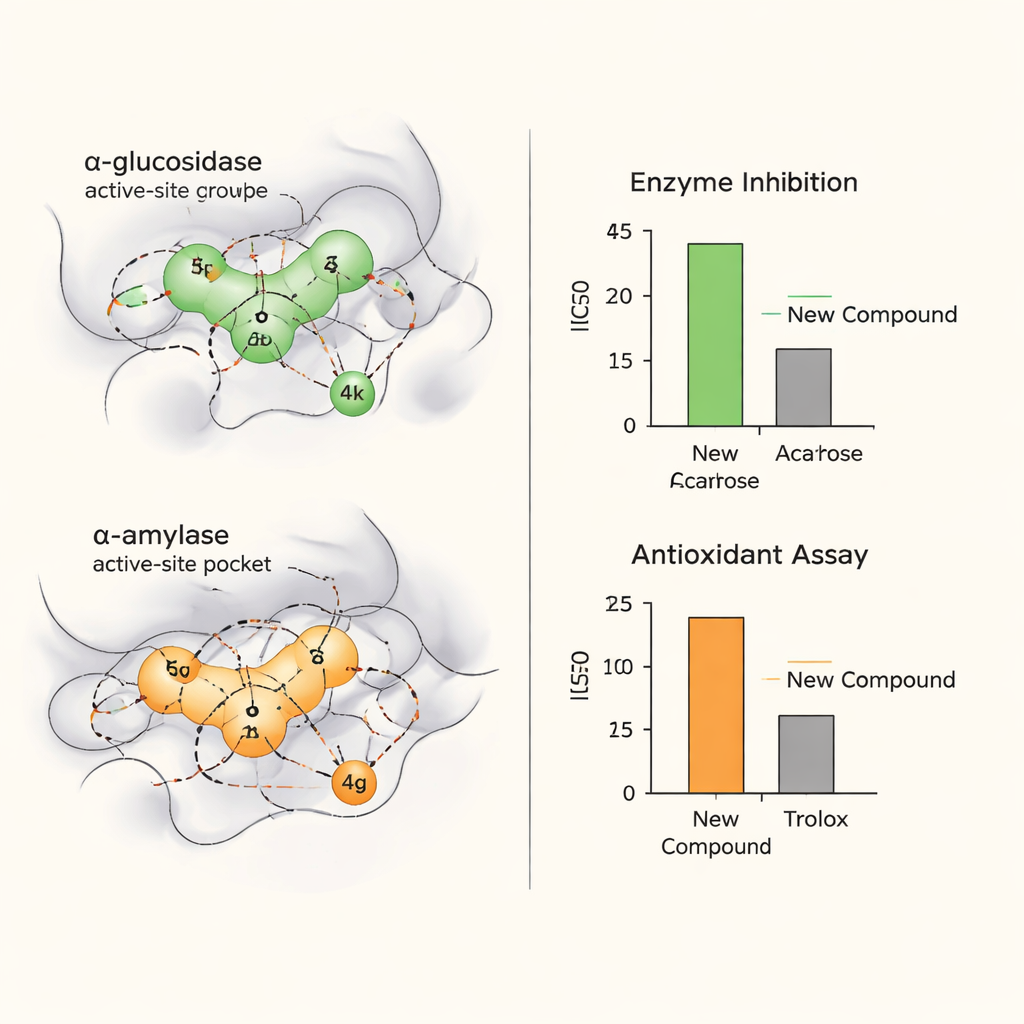
In laboratory tests, many of these hybrids were able to inhibit both α-glucosidase and α-amylase, often beating the widely used diabetes drug acarbose. One compound, labeled 4k, was especially potent against α-glucosidase, while another, 4g, excelled at blocking α-amylase. The same molecules also performed strongly in two standard antioxidant tests that measure how well a substance neutralizes unstable “free radical” species. Compounds 4o and 4g, in particular, outperformed Trolox, a vitamin E–like reference antioxidant. Together, these results mark the chromone–thiosemicarbazone hybrids as promising dual-action agents that may both slow sugar release and protect tissues from oxidative damage.
Peering into Molecular Workings with Computers
To understand how these molecules achieve their effects, the scientists used computer-based docking and molecular dynamics simulations. These tools virtually “fit” the compounds into three-dimensional models of the enzymes, showing where and how they bind. For compound 4k, simulations suggested a snug and stable fit in the α-glucosidase active site, maintaining key contacts throughout a long virtual run, while 4g behaved similarly in α-amylase. Additional network pharmacology analyses linked the most promising compounds to biological pathways that govern insulin response and the body’s handling of oxidative stress, hinting that their influence could extend beyond a single target.
What This Could Mean for Future Treatments
For non-specialists, the message is that chemists are learning to design smarter molecules that tackle diabetes on more than one front—controlling how fast sugar enters the blood and limiting the internal “rusting” caused by reactive oxygen species. While these new chromone–thiosemicarbazone hybrids are still at an early stage and have yet to be tested in animals or humans, they already surpass standard laboratory drugs in several key tests. With further safety and pharmacokinetic studies, such dual-action compounds could one day form the basis of therapies that better protect people with diabetes from both high blood sugar and the long-term tissue damage it brings.
Citation: Zareen, W., Ahmed, N., Siddique, F. et al. Synthesis and biological evaluation of 6-hydroxychromone based thiosemicarbazones as potential antidiabetic and antioxidant agents. Sci Rep 16, 7512 (2026). https://doi.org/10.1038/s41598-026-40449-y
Keywords: type 2 diabetes, enzyme inhibitors, antioxidants, drug discovery, chromone thiosemicarbazone