Clear Sky Science · en
Stimuli-responsive chitosan-coated ferrite nanocarriers for targeted capsaicin delivery and core-dependent HepG2-selective bioactivity
Spice with a Hidden Power
Chili peppers add more than heat to our food: their burn comes from capsaicin, a natural compound that can also kill cancer cells. Yet using capsaicin as a medicine has proven difficult because it dissolves poorly in water, disappears quickly from the body, and irritates healthy tissues at high doses. This study explores a way to tame that fiery molecule by tucking it inside tiny, smart particles that can carry capsaicin safely through the bloodstream and release it mainly inside liver tumors.
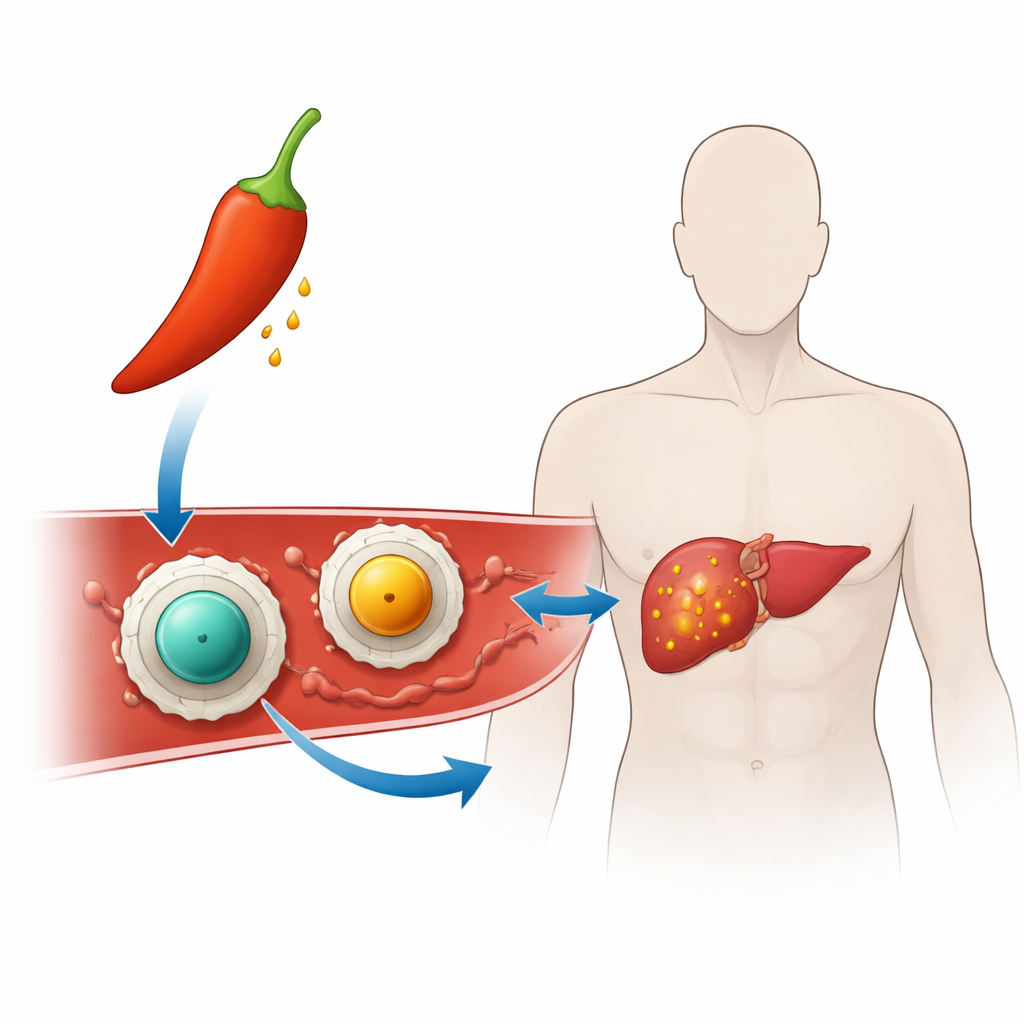
Turning Hot Pepper into Targeted Therapy
The researchers built “nanocarriers” – particles thousands of times smaller than a grain of sand – to ferry capsaicin to cancer cells. Each nanocarrier has a solid core made of a magnetic mineral called ferrite (either zinc ferrite or manganese ferrite) wrapped in a thin coat of chitosan, a sugar-like substance derived from shellfish that is already used in medical and food products. This coating makes the particles easier to suspend in water and helps them stick to cell surfaces and DNA, which is important for cancer killing. By loading capsaicin into this chitosan shell, the team hoped to keep the drug stable and hidden until it reaches the harsh environment inside a tumor.
Smart Release in Tumor Conditions
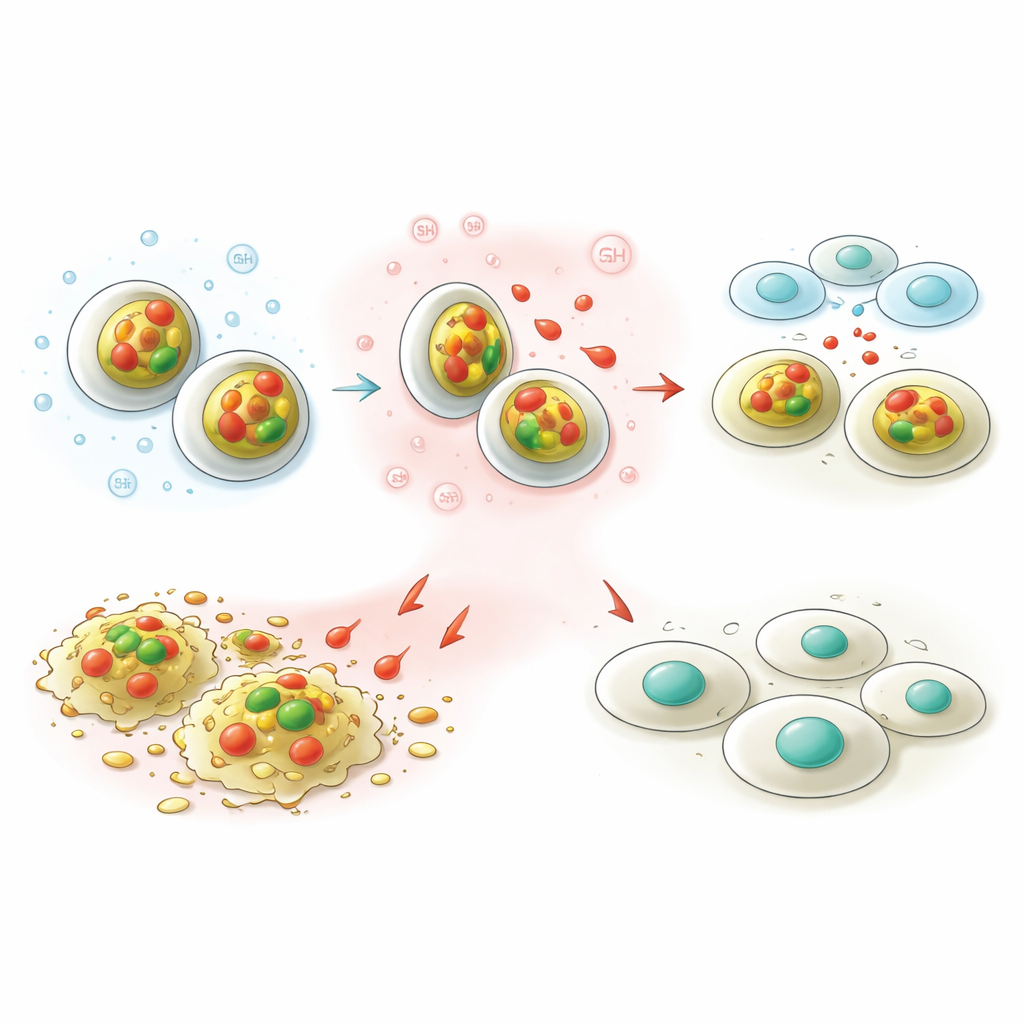
A key feature of tumors is that they tend to be more acidic and richer in certain antioxidants, such as glutathione, than healthy tissues. The team designed their nanocarriers to sense these conditions. In lab tests at normal blood pH, less than 10 percent of the stored capsaicin leaked out over two days, suggesting the particles would carry their cargo safely during circulation. Under tumor-like conditions – more acidic and with abundant glutathione – the particles released about a third of their capsaicin. This "on–off" behavior means the drug is more likely to be dumped inside cancer cells rather than along the way, potentially boosting benefit while lowering side effects.
How Particle Ingredients Shape Safety and Strength
Although the two nanocarriers looked similar from afar, their inner cores made a real difference. Zinc-based particles tended to be smaller and held on to capsaicin more tightly, while manganese-based particles were more porous and let the drug out more readily. Both types carried a strong positive surface charge, which helps them interact with the negatively charged surfaces of cells and DNA. Tests with human blood showed very low damage to red blood cells, and the empty particles were gentle to normal lung cells, supporting their basic safety. When loaded with capsaicin, both systems became more potent against liver cancer cells, but the manganese version achieved the best balance: it harmed cancer cells more strongly while remaining relatively kind to normal cells.
Beyond Cancer: Extra Protective Effects
The nanocarriers did more than deliver capsaicin. They also slowed the growth of several disease-causing bacteria as effectively as a standard antibiotic in some tests, and they helped neutralize harmful free radicals, which are linked to inflammation and tissue damage. Careful light-based measurements showed that, once capsaicin was packed into the ferrite–chitosan particles, it attached more strongly and more stably to DNA. Fast kinetic experiments revealed a two-step dance: DNA first latches onto the positively charged particle surface, then the complex rearranges into a tighter, longer-lasting association. These strengthened interactions likely help explain why the loaded particles show stronger anticancer, antibacterial, and antioxidant effects than capsaicin or ferrite particles alone.
What This Could Mean for Patients
For now, these findings come from lab dishes, not from patients. Still, they suggest that carefully engineered nanocarriers can turn a spicy food ingredient into a more precise cancer weapon. By choosing the right core material and shell, the researchers created tiny packages that keep capsaicin stable, carry it safely in blood, and unload it preferentially in tumor-like conditions, especially in liver cancer cells. Among the designs tested, manganese-based particles offered the most promising mix of targeted cancer killing, low harm to normal cells, and added antibacterial and antioxidant benefits. With further animal and safety studies, such “smart” pepper-inspired particles could one day contribute to gentler, more focused therapies for liver cancer and potentially other diseases.
Citation: Bakr, E., Elshami, F.I., Okba, E.A. et al. Stimuli-responsive chitosan-coated ferrite nanocarriers for targeted capsaicin delivery and core-dependent HepG2-selective bioactivity. Sci Rep 16, 8957 (2026). https://doi.org/10.1038/s41598-026-40433-6
Keywords: capsaicin, liver cancer, nanoparticles, targeted drug delivery, stimuli-responsive carriers