Clear Sky Science · en
The effect of increased weight loading on body weight is partly dependent on Piezo1 in osteoblast-lineage cells and TrkA signaling
Why extra weight might help fight obesity
Most of us think of extra weight as something to avoid, but what if carefully added weight could actually help the body shed fat? This study explores a built-in weight-sensing system in bones that appears to tell the brain when the body is too heavy, nudging it to burn fat. By uncovering how this system works at the cellular level, the research could open doors to treatments that reduce fat while preserving valuable muscle mass.
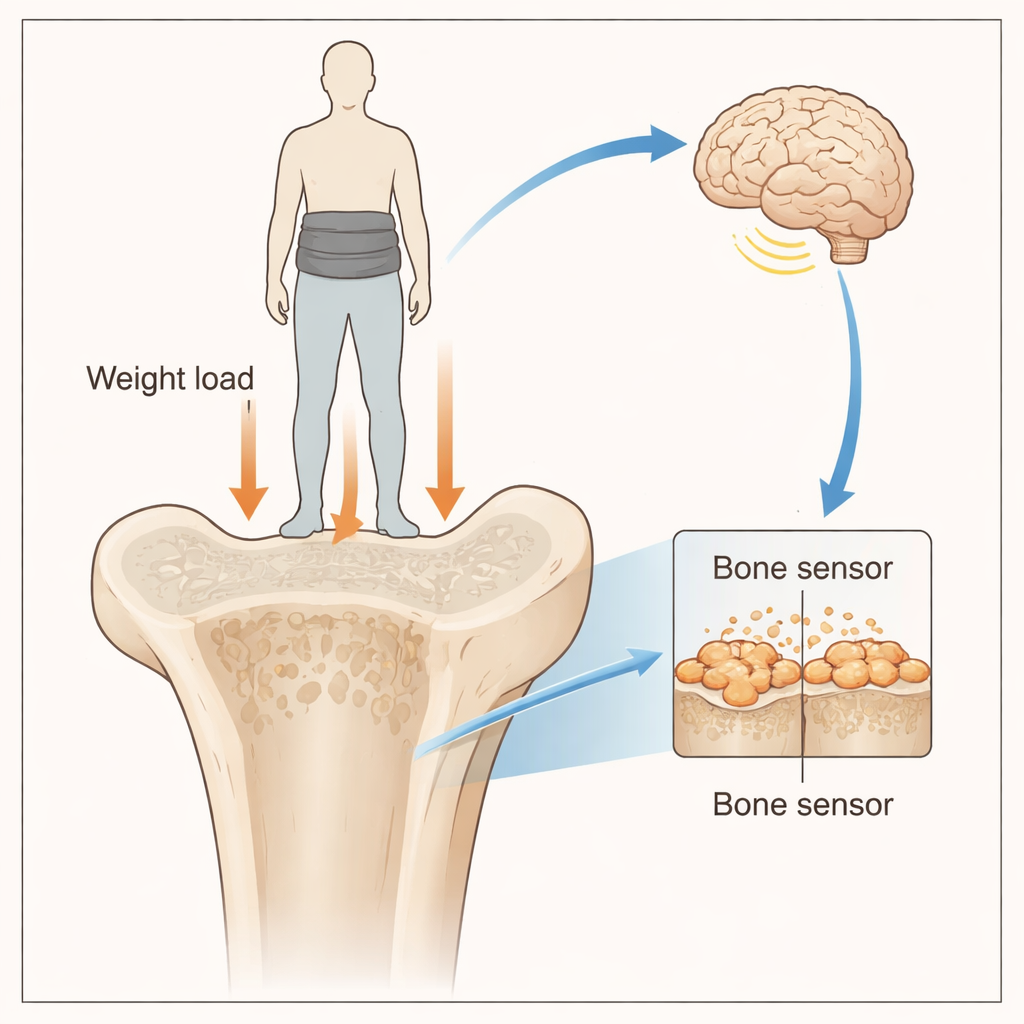
A hidden bathroom scale in your bones
Obesity is linked to diabetes, heart disease, and several cancers, and even the latest weight-loss drugs often cause loss of both fat and muscle. That is a serious problem for people with “sarcopenic obesity,” who already have weak muscles. Previous human trials showed that simply wearing a weighted vest for several weeks can lower body fat without reducing muscle. Earlier animal work led the authors to propose that bones in the legs contain a kind of internal “gravitational scale” that senses how heavy the body is and helps keep body fat in check. But which molecules actually do this sensing remained unknown.
Testing the bone weight sensor in obese mice
To investigate, the researchers used mice made obese with a high-fat diet and then surgically implanted small capsules in their abdomen. In one group, the capsule was filled with heavy material, adding about 15% to body weight; in control mice, the capsule was nearly weightless. After surgery, the scientists subtracted the capsule weight from each measurement so they could track true body weight changes. They also collected a specific fat depot—retroperitoneal white fat—from the abdomen at the end of the experiment to see how much fat the mice had lost.
A pressure-sensitive channel in bone cells
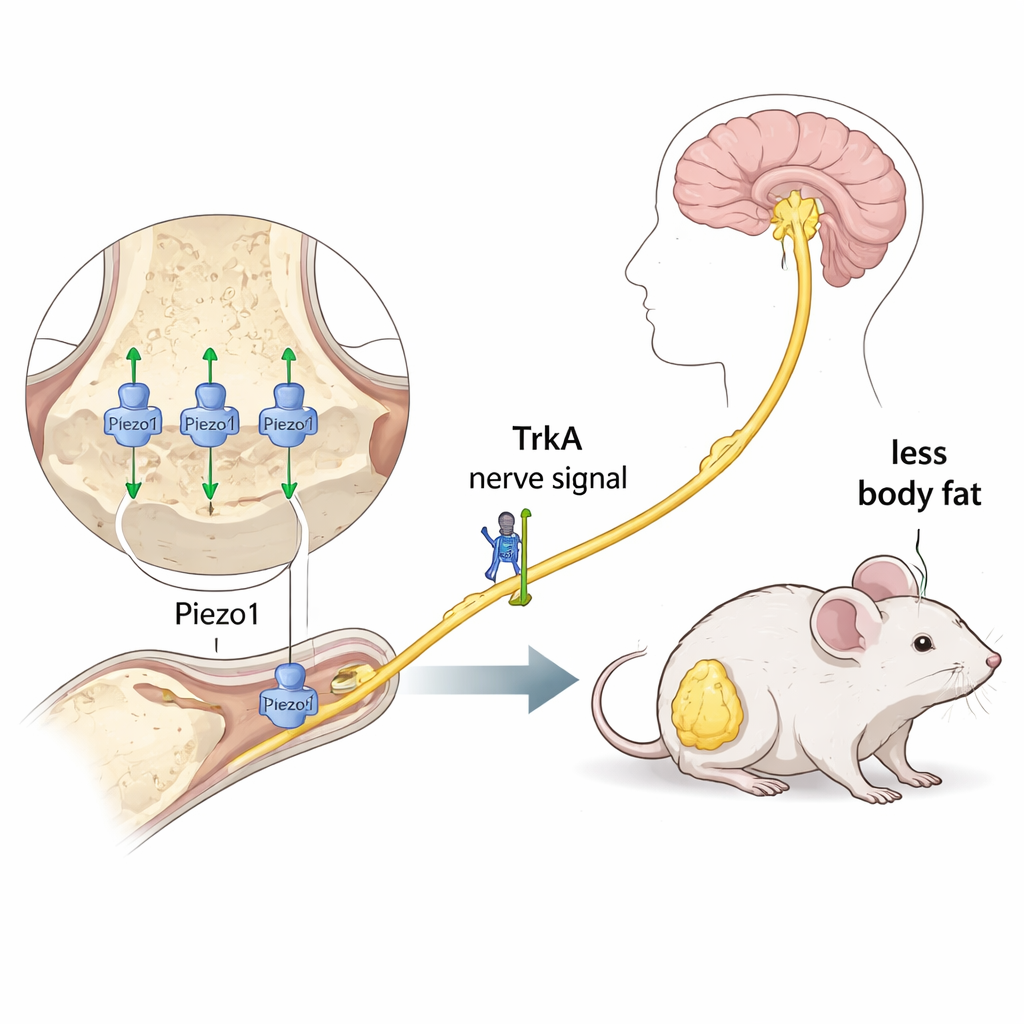
The team focused first on Piezo1, a protein channel in cell membranes that opens when cells are mechanically stretched or compressed. Using genetic engineering, they deleted Piezo1 only in early bone-forming cells (osteoblast-lineage cells) that express a marker called Runx2, mostly found in the long bones of the legs. Careful microscopic analyses confirmed that Piezo1 was strongly reduced in these bone cells but still present in nearby muscle, showing that the deletion was specific. In obese mice with normal Piezo1, the added weight led to a marked drop in overall body weight and a large reduction in abdominal fat. In contrast, mice lacking Piezo1 in bone-lineage cells showed a much weaker response: their body weight and abdominal fat barely decreased with the extra load. This indicates that Piezo1 in bone cells is a key part of the weight-sensing system.
Nerve signals that carry the message to the brain
Next, the scientists examined a second player: TrkA, a receptor found mainly on certain nerve fibers that respond to nerve growth factor (NGF). TrkA helps nerves grow and transmit pain and other signals. Using a special mouse strain, the team could turn off TrkA signaling throughout the body by adding a small molecule, 1NM-PP1, to the drinking water. A classic “tail-flick” pain test confirmed that TrkA signaling was indeed shut down: treated mice no longer became extra sensitive to heat after an NGF-like drug. When these mice were subjected to increased weight loading, animals with normal TrkA signaling lost significant body weight and abdominal fat, but mice with blocked TrkA signaling showed a much smaller and statistically weaker response. Interestingly, the added weight did not trigger obvious growth of new nerve fibers around the bone, suggesting that existing nerves, rather than newly sprouted ones, carry the signal.
What this means for future obesity treatments
Taken together, the results support a model in which extra body weight—whether from fat or from an added load—compresses the leg bones. Bone-lineage cells sense this pressure through the Piezo1 channel, then communicate via TrkA-dependent nerve pathways to brain centers that regulate appetite and energy use. The brain, in turn, helps drive down fat stores while sparing muscle. Although this work was done in mice and uses surgical implants, it helps explain why something as simple as wearing a weighted vest can reduce body fat in people. In the long run, targeting this bone–nerve–brain circuit could lead to new therapies that complement current drugs by selectively trimming fat without sacrificing muscle.
Citation: Hägg, D., Li, L., Beeve, A.T. et al. The effect of increased weight loading on body weight is partly dependent on Piezo1 in osteoblast-lineage cells and TrkA signaling. Sci Rep 16, 7162 (2026). https://doi.org/10.1038/s41598-026-40431-8
Keywords: obesity, bone mechanosensing, Piezo1, TrkA signaling, weighted loading