Clear Sky Science · en
Temperature dependent behavior of hydrogen-bonded liquids: bridging experiments with molecular dynamics and DFT
Why warm and cold liquids behave so differently
Anyone who has tried to pour cold syrup or mix chilled oils knows that temperature can dramatically change how liquids flow and blend. This study digs into why that happens in a specific family of industrially important liquids: mixtures of an aldehyde called heptanal with several closely related alcohols. By watching these mixtures from both a laboratory and a molecular point of view, the authors show how gentle heating quietly rearranges the invisible network of attractions between molecules, with big consequences for density, thickness, and ease of flow. 
Everyday solvents with a hidden handshake
The liquids examined here are far from exotic. Aldehydes and alcohols are common in fuels, fragrances, cosmetics, and pharmaceutical formulations. When mixed, their molecules “shake hands” through directional attractions between the alcohol’s OH group and the aldehyde’s carbonyl group, forming what chemists call hydrogen bonds. These bonds pull molecules closer together than simple mixing would predict, making the blend slightly more compact and thicker than an ideal, non‑interacting mixture. The researchers focused on heptanal combined with straight‑chain alcohols from 1‑propanol to 1‑heptanol, asking two key questions: how does temperature change these molecular handshakes, and how does the length of the alcohol’s tail influence the outcome?
Measuring how liquids pack and flow
In the lab, the team carefully measured density and viscosity of each mixture over a range of temperatures near room temperature and above. They found that all mixtures show negative “extra” volume when blended, meaning that the mixed liquid occupies less space than its ingredients would separately. At the same time, the mixtures are thicker (more viscous) than a simple rule‑of‑thumb would suggest. Both effects are strongest at low temperature and for the shortest alcohol, 1‑propanol, and they steadily fade as the liquids are warmed or as the alcohol chains get longer. This pattern points to strong, efficient attractions between heptanal and short alcohols that draw molecules together and resist flow, and to weaker, more hindered interactions when the alcohol tail is bulkier.
Watching the molecules move and cluster
To see the microscopic story behind these measurements, the authors turned to molecular dynamics simulations and quantum chemical calculations. Computer models of thousands of molecules revealed how often and how closely heptanal and alcohol molecules cluster around one another. At low temperatures, the simulations show many short, well‑defined hydrogen bonds and a tightly packed structure with only small empty pockets between molecules. As the temperature rises, those bonds become less frequent and slightly longer, molecules move more freely, and the empty pockets grow larger and more connected—direct evidence of increased free space and faster diffusion. For short alcohols, the environment around heptanal is relatively uniform, while longer alcohols create a more irregular first shell, as their bulky tails partly block access to the key binding sites. 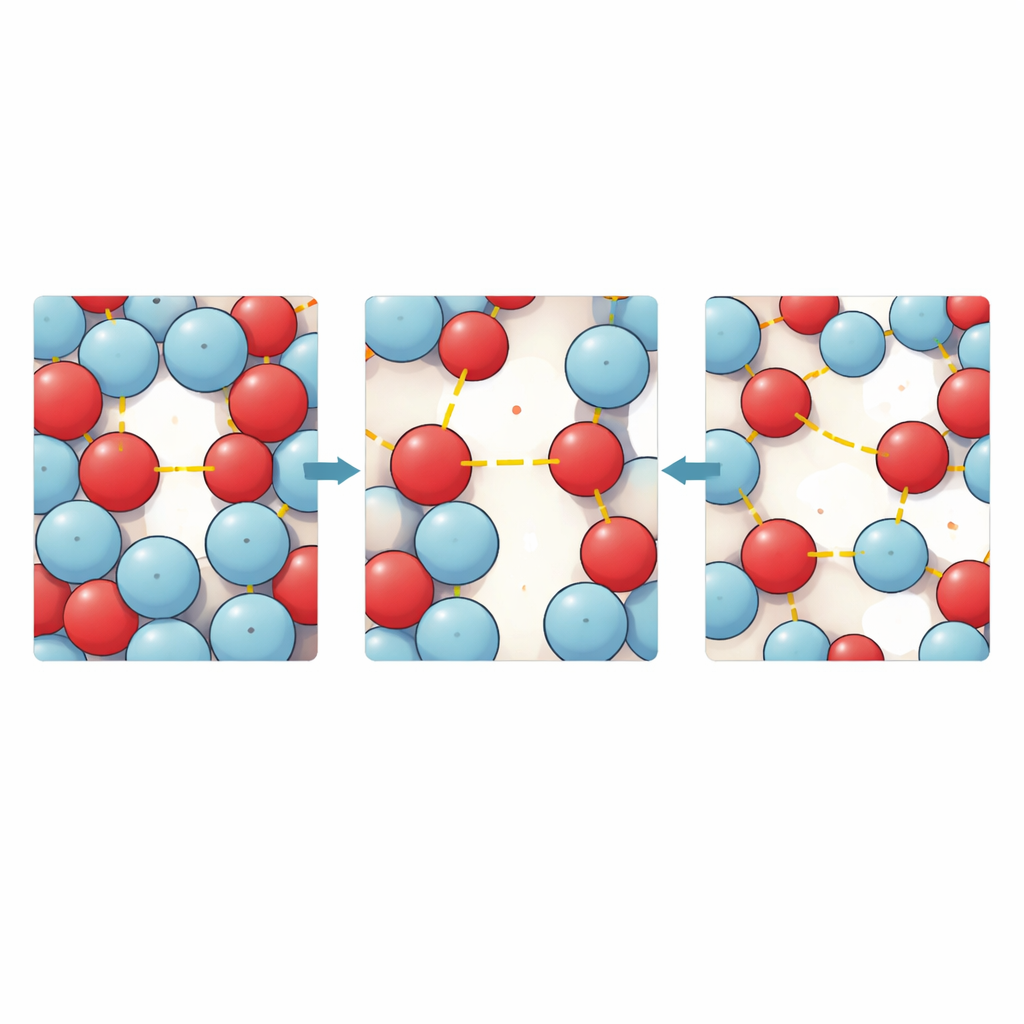
Balancing energy, order, and molecular crowding
By analyzing how the molecular arrangements change with temperature, the team could tease apart the roles of energetic stabilization and disorder. Forming close contacts between molecules releases a small amount of energy, making these interactions favorable, but it also restricts their freedom to move and rotate. The calculations show that, for these mixtures, the energetic gain from hydrogen bonding slightly outweighs the loss of freedom, so local contacts are driven mainly by attractive energy with a modest penalty in disorder. Quantum chemical models of simple pairs of molecules confirm that heptanal–alcohol pairs are generally more strongly bound than alcohol–alcohol pairs for shorter chains, reinforcing mixed bonding and compact packing. For the longest alcohol studied, this advantage nearly disappears, and the alcohol prefers to bond with itself as much as with heptanal, weakening the special contraction and thickening seen in shorter‑chain mixtures.
What this means for real‑world liquids
In simple terms, the paper shows that at low temperatures, heptanal and short‑chain alcohols interlock through many directional attractions, pulling themselves into a closely packed, relatively sluggish liquid. Heating the mixture shakes those connections loose, opening up more empty space, letting molecules slide past one another more easily, and bringing the behavior closer to that of ordinary, less interactive liquids. As alcohol chains lengthen, their bulky tails get in the way of this neat interlocking, so temperature has a milder effect and the mixtures become less compact and less viscous overall. By tying together measurements, simulations, and detailed calculations, the study offers a clear, multi‑scale picture of how changing temperature and molecular size tunes the hidden network of bonds that governs how everyday hydrogen‑bonded liquids pack, flow, and respond in industrial processes.
Citation: Almasi, M., Vatanparast, M. Temperature dependent behavior of hydrogen-bonded liquids: bridging experiments with molecular dynamics and DFT. Sci Rep 16, 9185 (2026). https://doi.org/10.1038/s41598-026-40428-3
Keywords: hydrogen-bonded liquids, temperature effects, aldehyde–alcohol mixtures, molecular dynamics, viscosity and density