Clear Sky Science · en
Structural evolution and optical tailoring of Mg-doped ZnO: Insights into doping-induced modifications
Why tiny crystals matter for future gadgets
From phone screens to solar panels, many modern devices rely on materials that can carefully control how they absorb and emit light. This study looks at zinc oxide, a common, low-cost material, and shows how adding a small amount of another element, magnesium, can fine‑tune its structure and light‑handling abilities. That kind of control could help build more efficient solar cells, sensors, and ultraviolet light devices.
Building a better light‑friendly material
The researchers focused on zinc oxide nanoparticles—grains thousands of times smaller than the width of a human hair. Zinc oxide is already popular because it is stable, non‑toxic, and good at interacting with ultraviolet light. The team explored what happens when some zinc atoms are replaced with magnesium atoms, in amounts up to 15 percent. Their goal was to see how this tiny chemical adjustment changes both the internal crystal structure and the way the material responds to light, with an eye toward future use as an electron‑conducting layer in high‑efficiency perovskite solar cells and in other optoelectronic devices.
Cooking up nanoparticles in the lab
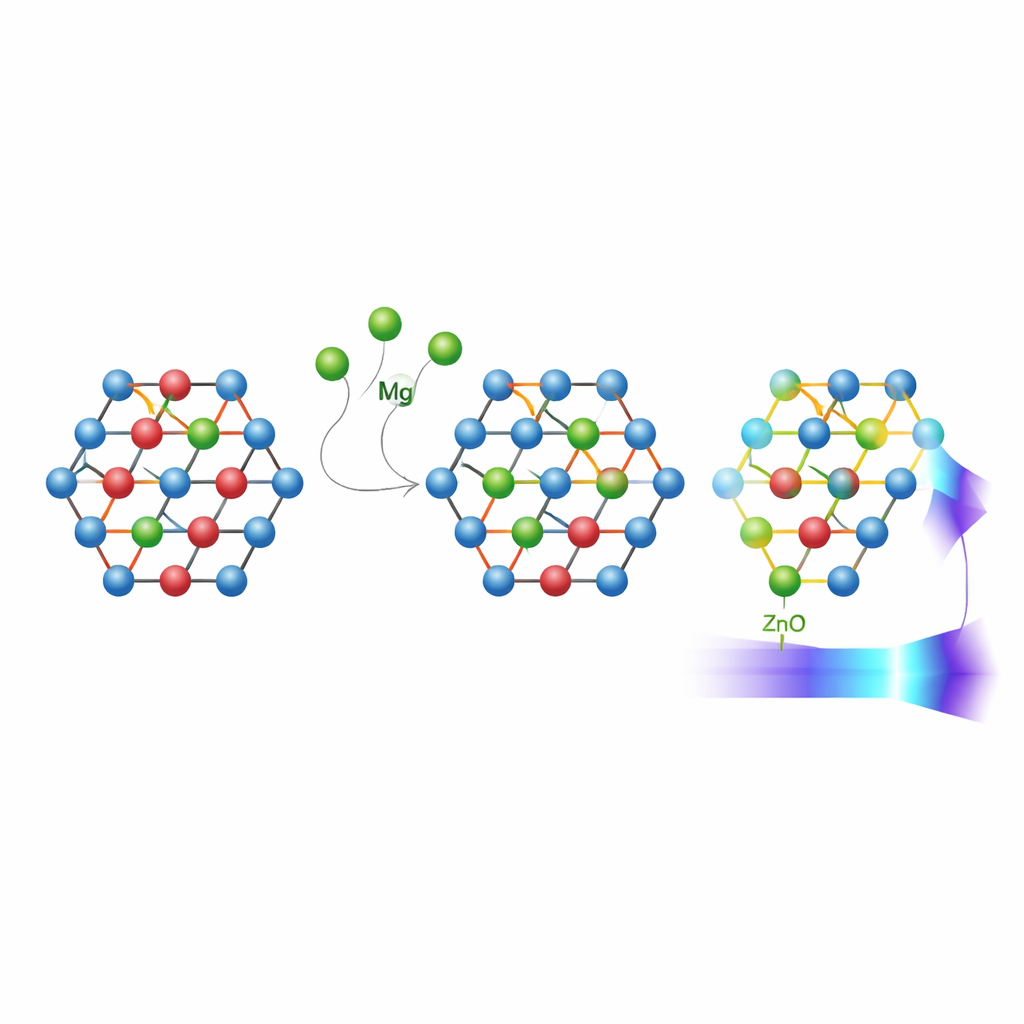
To make the materials, the team used a relatively simple and inexpensive sol‑gel process, mixing water‑based solutions of zinc and magnesium salts with citric acid and then heating them through several steps. This route produced fine powders of mixed zinc‑magnesium oxide nanoparticles. X‑ray measurements showed that, even as magnesium content increased, the particles kept the same underlying hexagonal crystal pattern typical of zinc oxide. Magnesium atoms slid into the zinc positions without forming unwanted extra phases, and the average crystal size stayed in the tens of nanometers, growing modestly as more magnesium was added.
How shape and bonds quietly change
Microscope images revealed that the particles tended to cluster into roughly spherical or hexagonal aggregates. At low magnesium content the clusters were denser and made up of smaller grains, while higher magnesium levels produced more open, porous clumps of somewhat larger grains. Infrared measurements, which probe atomic vibrations, confirmed that the basic zinc‑oxygen framework stayed intact, with subtle shifts in the vibration frequencies as lighter magnesium atoms and slightly shorter magnesium‑oxygen bonds appeared. These changes went hand‑in‑hand with a reduction in certain structural defects, meaning the crystals became more orderly as magnesium was introduced.
Tuning how the material handles light
The most technologically important shifts showed up when the team examined how the powders absorbed and emitted light. By analyzing reflected ultraviolet and visible light, they found that the energy gap between filled and empty electronic states—the band gap—grew slightly as magnesium content rose from zero to about 6 percent, then dipped a bit at higher levels but still stayed above that of pure zinc oxide. This means the material can be pushed to interact more strongly with higher‑energy ultraviolet light. A related quantity, called the Urbach energy, shrank with magnesium addition, signaling fewer disordered states at the edges of this gap and a sharper onset of absorption. Light‑emission measurements told a complementary story: at low magnesium levels, the nanoparticles glowed mainly in the near‑ultraviolet, while higher magnesium contents shifted and broadened the glow and highlighted the role of defects such as missing oxygen atoms. Together, these effects show that the brightness, color, and sharpness of emission can all be adjusted through careful control of magnesium content.
What this means for real‑world devices
By demonstrating that magnesium can smoothly substitute into zinc oxide nanoparticles while subtly reshaping both their crystal structure and optical response, the study points to a practical way of "dialing in" desired properties for specific technologies. Materials engineers can choose a magnesium level that balances crystal quality against useful defect‑related light emission, or that matches the energy levels needed in a solar cell or light‑emitting device. In simple terms, the work shows how a small chemical tweak can act like a fine adjustment knob on a familiar material, making it a more versatile building block for the next generation of energy and light‑based technologies.
Citation: Kumar, M., Kumar, A., Dabas, S. et al. Structural evolution and optical tailoring of Mg-doped ZnO: Insights into doping-induced modifications. Sci Rep 16, 8919 (2026). https://doi.org/10.1038/s41598-026-40403-y
Keywords: zinc oxide nanoparticles, magnesium doping, optical band gap, perovskite solar cells, optoelectronic materials