Clear Sky Science · en
Copper catalyzed synthesis of thiazole derivatives from enaminones, amines and CS₂
Why This New Reaction Matters
Chemists are always searching for faster, cleaner ways to build the complex molecules that power modern medicines and electronic materials. This study introduces a streamlined recipe for making a family of small ring-shaped compounds called thiazoles, which show up in many drugs and advanced materials. By combining three simple ingredients in a single pot and letting a common metal, copper, guide the transformation, the researchers show how to turn basic building blocks into a wide variety of potentially useful molecules with minimal waste and fuss. 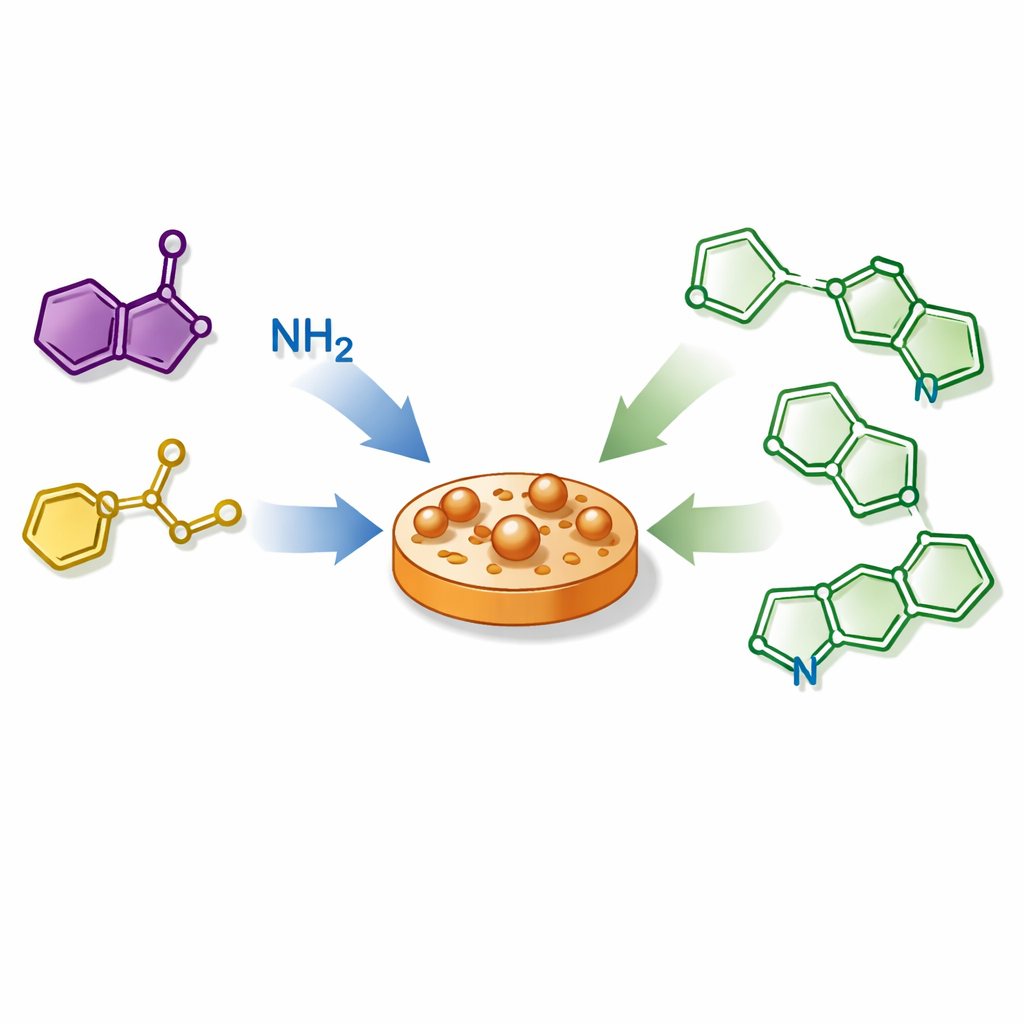
Small Rings With Big Impact
Thiazoles are compact rings that contain both nitrogen and sulfur atoms. That tiny twist in structure gives them unusual electrical and biological behavior. Thiazole-based compounds have been explored as antibiotics, anti-inflammatory agents, anticancer leads, and treatments for conditions ranging from diabetes to neurological disorders. Related structures also appear in organic electronics, where they help move charge in devices like light-emitting diodes and conductive polymers. Because of this broad impact, chemists are eager for general, dependable ways to build different thiazole shapes on demand.
Limits of Older Synthetic Routes
Traditional methods for assembling thiazoles usually start from pre‑made sulfur-rich components or require multiple separate steps, specialized reagents, or harsh conditions. Many of these routes are tuned to a specific subclass known as benzothiazoles and are less flexible when it comes to making other ring variants. Some approaches also rely on extra additives, ligands, or oxidizing agents, which add cost and generate more chemical waste. These constraints make it harder to quickly explore new thiazole designs for drug discovery or materials screening.
One-Pot Copper-Guided Assembly
The authors present a different strategy built around three readily available pieces: enaminones (simple carbon-based fragments with built‑in reactivity), common amines, and carbon disulfide, a cheap liquid that here supplies both sulfur and carbon. Under the influence of a copper salt and a basic additive in a single reaction vessel, these ingredients link together and fold into the desired thiazole ring. The process requires no special helper molecules and no added oxidant; ordinary oxygen from the air helps recycle the copper catalyst. Through systematic testing of solvents, temperatures, bases, and metal salts, the team identifies conditions that efficiently convert the starting materials into thiazoles, often in high yield. 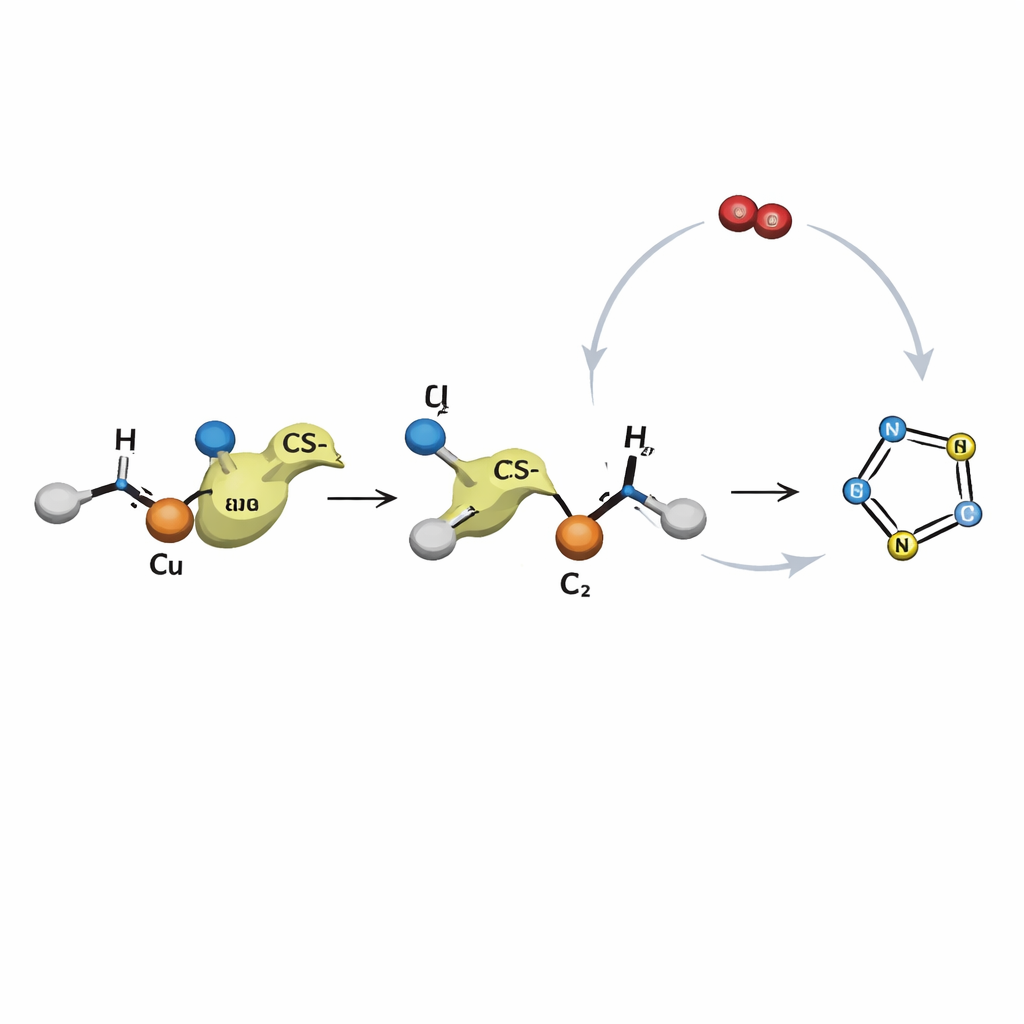
Wide Variety From Simple Inputs
Once the conditions were optimized, the researchers explored how general the reaction is by feeding in many different amines and enaminones. A broad range of secondary amines, including both flexible chain-like and rigid ring-shaped examples, produced the desired thiazoles in good to excellent yields. Primary amines also worked, though typically less efficiently. The reaction tolerated various decorative groups on aromatic rings, and even bulkier "ortho" attachments had little negative effect, suggesting that crowding near the reactive site is not a major limitation. Some strongly withdrawing or nitrogen-rich amines, however, failed to react, likely because they weakened the key sulfur-based partner needed for ring closure.
How the Copper Catalyst Does the Job
To probe how this transformation unfolds, the authors ran control experiments and proposed a step-by-step pathway. First, the amine and carbon disulfide combine under basic conditions into a sulfur-rich fragment. At the same time, the enaminone binds to copper, which activates it toward reaction. The sulfur–nitrogen fragment then attacks this activated partner, forging new carbon–sulfur and carbon–nitrogen bonds. This linkage rearranges to give a partial ring that closes on itself, ejecting hydrogen sulfide and forming the final thiazole core. Copper cycles between two oxidation states during this sequence, and oxygen from the air helps push it back to the active form so the catalyst can be reused many times within the same reaction mixture.
A Straightforward Path to Useful Molecules
Overall, the work delivers a practical and flexible route for making thiazole derivatives from simple, easy-to-handle starting materials. Because the reaction occurs in one pot, without added ligands or oxidants, it simplifies laboratory workflows and reduces chemical waste. The ability to generate many different thiazole structures by swapping in different amines and enaminones makes this method a valuable tool for chemists designing new medicines or functional materials based on this important ring system.
Citation: Arman, A., Nowrouzi, N. & Abbasi, M. Copper catalyzed synthesis of thiazole derivatives from enaminones, amines and CS₂. Sci Rep 16, 9184 (2026). https://doi.org/10.1038/s41598-026-40393-x
Keywords: thiazole synthesis, copper catalysis, heterocyclic chemistry, enaminones, carbon disulfide