Clear Sky Science · en
Alternative splicing and differential gene expression during changes in endometrial receptivity in patients with recurrent implantation failure
Why some embryos do not implant
Many couples going through in vitro fertilization face a frustrating mystery: even when high‑quality embryos are transferred, pregnancy may still not occur. This study looks closely at the lining of the uterus—the endometrium—to understand why some women experience repeated failed implantations. By examining how genes are switched on and off, how their messages are cut and rearranged, and how immune cells behave during the brief window when the womb is ready to receive an embryo, the research uncovers new molecular clues that may one day guide more precise tests and treatments.
The womb’s brief welcoming window
Implantation can happen only during a short span in each cycle when the uterine lining becomes "receptive." The researchers compared tissue samples from 90 fertile women and 73 women with recurrent implantation failure, all taken at three key times: just before the lining becomes receptive, during the receptive window, and shortly after it closes. Using high‑throughput RNA sequencing, they measured which genes were active and how their RNA messages were processed. This design let them track how a healthy lining prepares for an embryo and how that preparation goes awry in women who repeatedly fail to implant despite good embryos. 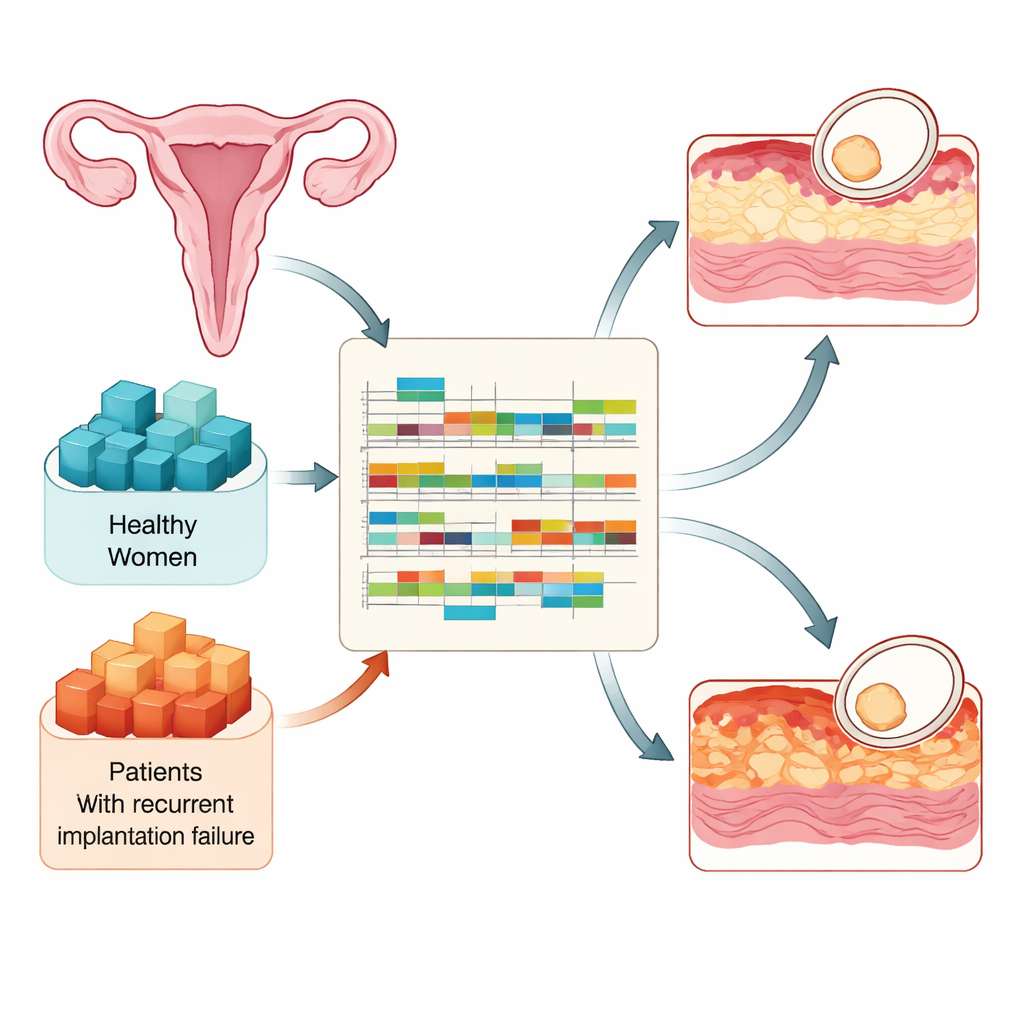
Gene messages cut and rearranged
Beyond simply counting how much of each gene is active, the team focused on alternative splicing—the process by which a single gene can be cut into different message variants, leading to distinct protein forms. They found over a million splicing events across comparisons, with two types—skipping entire segments and choosing between mutually exclusive segments—being especially common. When they looked only at changes that were both frequent and statistically robust, they saw hundreds to thousands of splicing shifts as the lining moved from the non‑receptive to the receptive phase, particularly from the early (pre‑receptive) stage into the implantation window. Many of these changes occurred even when the overall amount of the gene stayed the same, meaning the form of the protein changed without a change in total gene activity.
Cells sticking, reshaping, and talking to the immune system
Genes affected by altered splicing and by changes in activity were heavily involved in how cells stick to each other, how their internal scaffolding is rearranged, and how they send and receive immune signals. These functions are crucial as the uterine lining transforms from a simple barrier into a surface that can welcome and anchor an embryo while remodeling blood vessels. In both fertile women and those with implantation failure, the receptive phase was marked by strong remodeling in these pathways, but women with recurrent failure tended to show more frequent or differently patterned splicing changes. Some of the genes involved have already been suggested as markers of the ideal implantation window, reinforcing the idea that not just gene activity, but the exact form of their products, helps define when the lining is truly ready.
Immune cells and a key splicing regulator
The team also estimated the types of immune cells present in the tissue from the same RNA data. In women with implantation failure, the receptive phase showed fewer killer T cells and resting natural killer cells, but more monocytes and certain macrophages compared with earlier phases. Later, after the window closed, patterns shifted again, with changes in activated natural killer cells and macrophage subtypes. Several of these immune cell changes closely tracked specific splicing events, hinting that gene‑message reshaping and immune balance are linked. Among proteins that control splicing, one called KHDRBS3 stood out as a central hub connected to many of the altered events and consistently dropped in level when the lining became receptive, in both fertile and affected women. This suggests it may help drive the switch in splicing patterns that accompanies the opening of the implantation window. 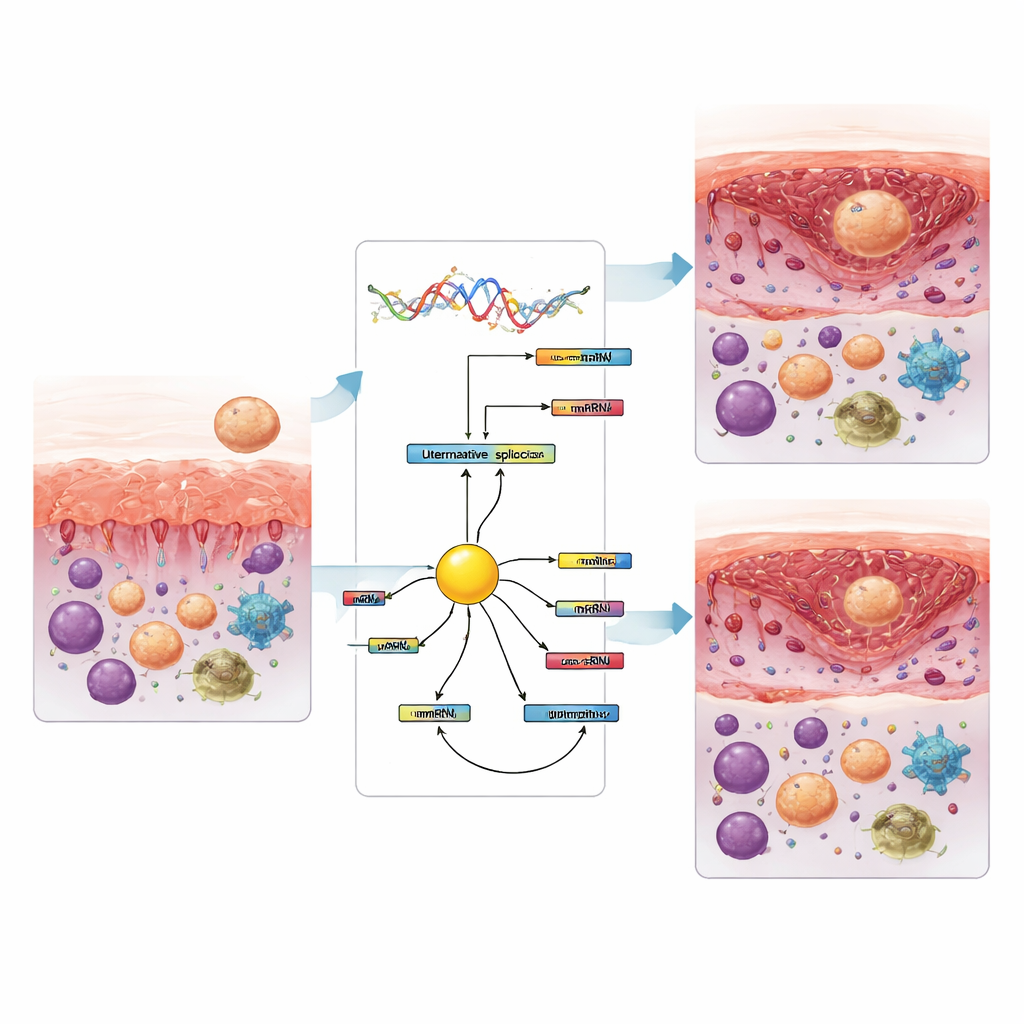
Pointing toward future tests and treatments
To translate these molecular patterns into possible therapies, the researchers asked whether existing small‑molecule drugs might counteract the abnormal splicing signatures they observed. By matching their gene lists against large drug‑response databases, they identified several approved or experimental compounds that target genes tied to implantation, including agents that influence immune responses or hormone‑related pathways. While these are far from ready for clinical use in this context, they offer a starting map for laboratory testing.
What this means for patients
Overall, the study shows that implantation failure is not simply a matter of having the "wrong" genes, but of how those genes are edited into different messages and how the immune environment of the womb is tuned over time. The work highlights KHDRBS3 and related factors as promising regulators of this process and reveals coordinated shifts in immune cells that may either support or hinder an embryo’s attempt to settle in. In the long run, such insights could lead to more refined tests that determine the true timing and quality of a woman’s receptive window, as well as targeted treatments that gently adjust gene messaging or immune balance to give embryos a better chance to implant.
Citation: Wang, Ml., Lu, Bj., Lu, X. et al. Alternative splicing and differential gene expression during changes in endometrial receptivity in patients with recurrent implantation failure. Sci Rep 16, 9754 (2026). https://doi.org/10.1038/s41598-026-40386-w
Keywords: endometrial receptivity, recurrent implantation failure, alternative splicing, uterine immune cells, assisted reproduction