Clear Sky Science · en
Trabectedin plus CD13-targeted tissue factor tTF-NGR against advanced relapsed or refractory soft tissue sarcoma: translational data, clinical safety and efficacy
Why this research matters
People with advanced soft tissue sarcomas – rare cancers that grow in muscles, fat, or other supporting tissues – often run out of effective treatment options. This study explores a new two-step drug strategy designed to trap an existing chemotherapy drug inside the tumor and cut off the tumor’s blood supply, with the hope of improving control of the disease while keeping side effects manageable.
A rare cancer that needs better options
Soft tissue sarcomas make up only about 1% of adult cancers, but they are often aggressive once they spread or cannot be removed by surgery. Standard chemotherapy can slow them down, yet survival for people with advanced disease remains poor. Trabectedin is one of the main drugs used after first-line treatments fail. It works by damaging the DNA of cancer cells and also by affecting cells in the tumor’s surroundings. Even so, many tumors eventually grow again, motivating researchers to look for ways to boost its impact without greatly increasing harm to patients.
A two-step "trap" for the tumor
The team tested a concept they call the "trabectedin trap" (TRABTRAP). First, patients receive trabectedin through a 24-hour infusion. On the next two or three days, they receive a second drug, tTF-NGR. This engineered protein homes in on a marker called CD13 that is found mainly on blood vessels feeding tumors. When tTF-NGR latches onto these vessels, it triggers local blood clotting and blocks blood flow, causing parts of the tumor to die. The idea is that giving trabectedin first lets it seep into the tumor, and then tTF-NGR seals the exits by closing the tumor’s small vessels, trapping the drug where it is needed most and amplifying the impact of both treatments.
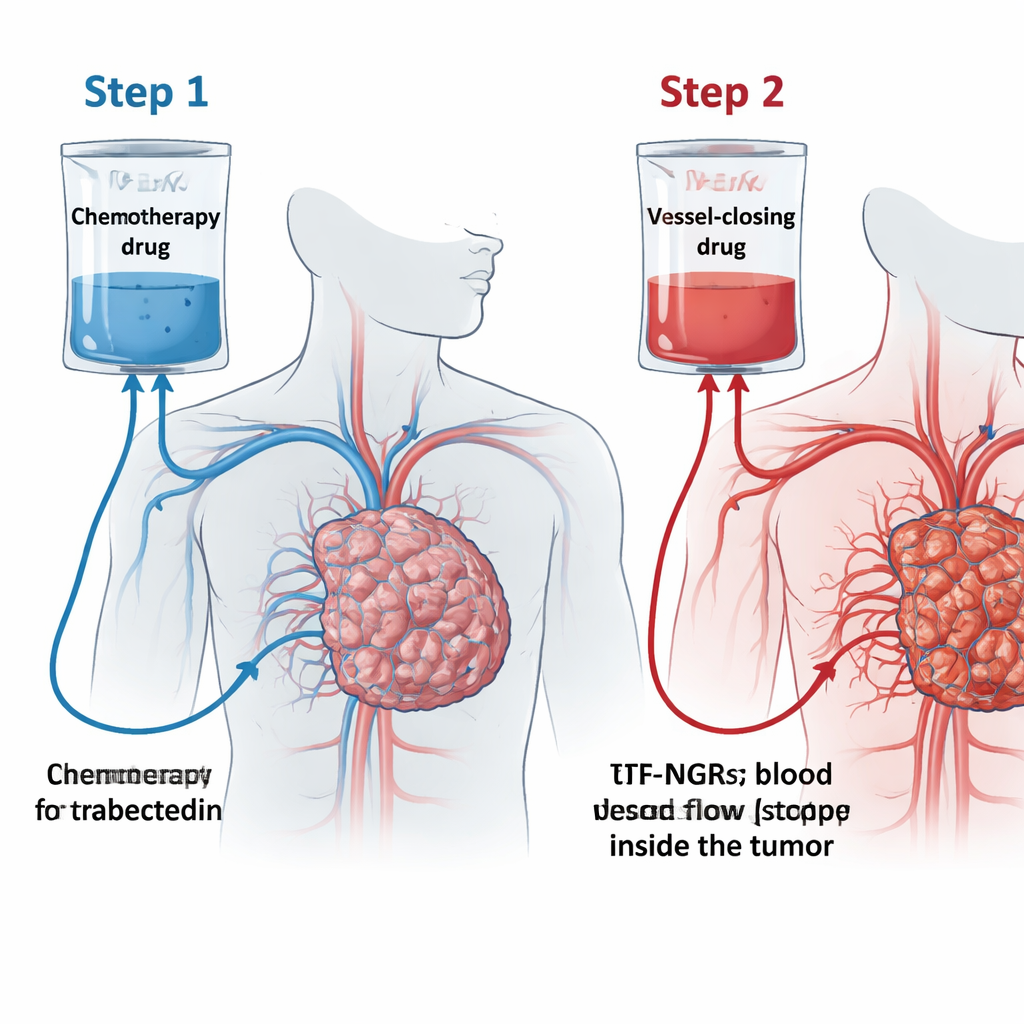
From lab bench to early patient testing
In laboratory experiments, the researchers showed that trabectedin makes blood-vessel cells and sarcoma cells display more of a certain fat molecule on their surface that supports clotting. This change made tTF-NGR more effective at triggering local clot formation on these cells, which is exactly what is needed to shut down tumor vessels. In mice bearing human sarcoma tumors, the sequence of trabectedin followed by tTF-NGR shrank tumors more than either drug alone. Because animal tumors broke down so completely under the combination, the team could not reliably measure how much extra trabectedin was trapped, but the stronger tumor control supported the basic idea of the "trap."
Finding a safe dose in real patients
The heart of this paper is the safety "run-in" phase of the TRABTRAP trial in 19 people with advanced, heavily pretreated soft tissue sarcomas. All patients received the standard dose of trabectedin. The second drug, tTF-NGR, was started at the highest dose previously tolerated when used alone, then carefully stepped down when serious side effects appeared. At higher doses, some patients developed significant heart-related or clotting problems, such as silent heart muscle injury seen on blood tests, small lung clots, or deep-vein thrombosis. These events were reversible with treatment and stopping or reducing tTF-NGR, but they signaled the need for lower doses. At 0.5 mg/m² of tTF-NGR given on two days after trabectedin, six patients completed at least two cycles without any dose-limiting toxicities, and some continued for up to 14 cycles.
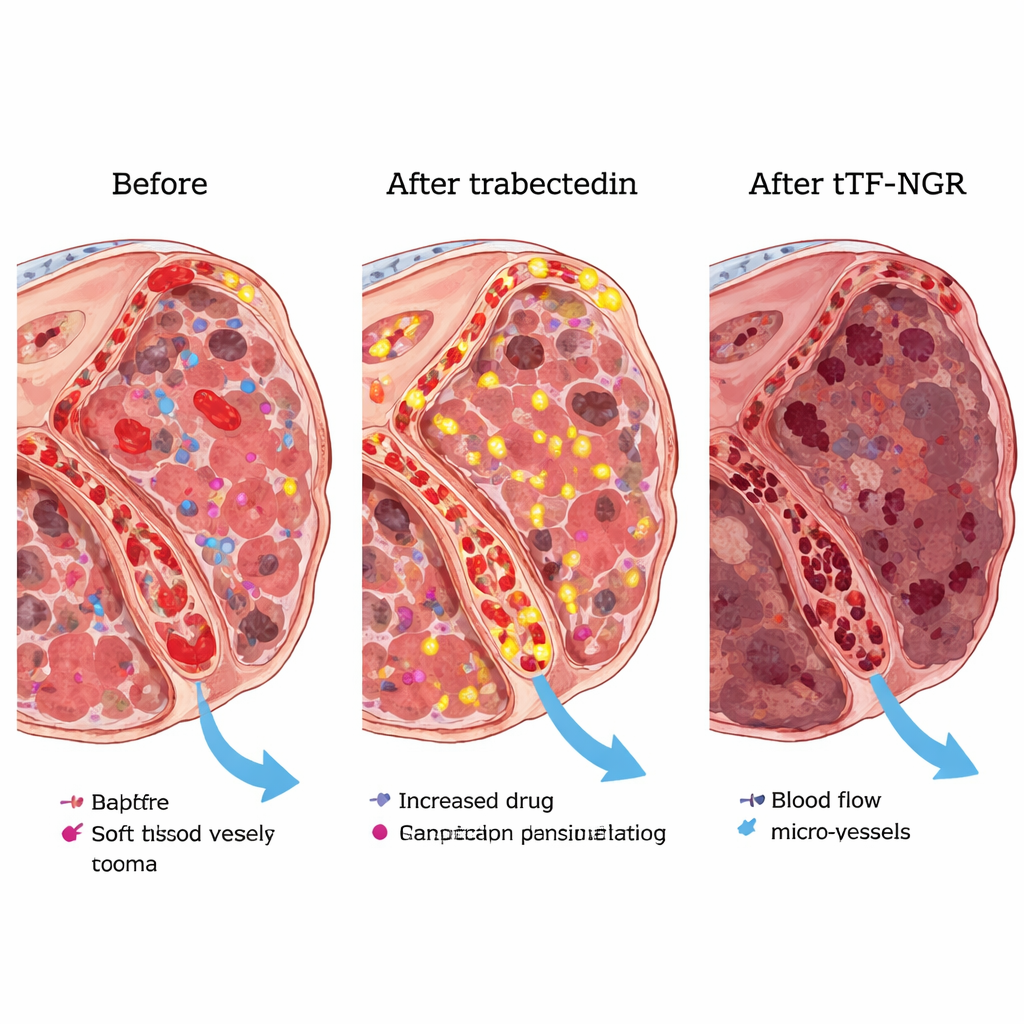
Early signs of benefit and how the drugs interact
Although this part of the study was too small to prove effectiveness, the results were encouraging. Among the 19 patients, most had at least temporary disease control, and two had clear partial shrinkage of their tumors. In people with liposarcoma or leiomyosarcoma – two common sarcoma subtypes for trabectedin – nearly two thirds had stable disease or partial responses at the first major checkup. Detailed MRI scans in one patient confirmed that tTF-NGR at the chosen dose could selectively reduce blood flow inside tumor deposits. Blood tests showed that, when given after trabectedin, tTF-NGR stayed in the circulation longer and appeared more active than when used alone, which helps explain why the safe dose in combination is lower than for the single drug.
What this means for patients
For people facing advanced soft tissue sarcoma, TRABTRAP represents a carefully engineered attempt to get more out of an existing chemotherapy by pairing it with a vessel-closing agent that targets the tumor’s own blood supply. The safety run-in shows that, at adjusted doses, this combination can be given repeatedly, with side effects that are significant but generally manageable and reversible under close monitoring. While it is too early to say whether it will extend life or consistently shrink tumors, the promising early disease control and clear effects on tumor blood flow justify moving ahead to a larger, randomized trial. If future results are positive, this two-step approach could open a new avenue for treating sarcomas and might inspire similar strategies for other hard-to-treat cancers.
Citation: Hessling, K., Brand, C., Schwöppe, C. et al. Trabectedin plus CD13-targeted tissue factor tTF-NGR against advanced relapsed or refractory soft tissue sarcoma: translational data, clinical safety and efficacy. Sci Rep 16, 7389 (2026). https://doi.org/10.1038/s41598-026-40362-4
Keywords: soft tissue sarcoma, trabectedin, tumor blood vessels, targeted coagulation therapy, combination chemotherapy