Clear Sky Science · en
Comparison of innate and adaptive immune responses to joint injury during the onset and progression of post-traumatic osteoarthritis in common murine models
Why joint injuries don’t always end the same way
Many people who tear a knee ligament or damage their meniscus go on to develop painful, long‑lasting osteoarthritis—but others with very similar injuries do not. This study in mice asks a simple but important question: beyond the mechanical damage, what is happening inside the joint that tips some injuries toward chronic arthritis while others heal? By tracking immune cells over time after different kinds of knee injuries, the researchers show that the body’s own inflammatory defenses can help predict, and possibly drive, whether post‑traumatic osteoarthritis takes hold.
Different knee injuries, different long‑term futures
The team used several well‑established mouse models that mimic common human knee injuries. Some procedures damaged the anterior cruciate ligament (ACL), which stabilizes the knee, either surgically or by a controlled mechanical rupture. Another surgery destabilized the medial meniscus, the cushioning pad on the inner side of the joint. Two comparison groups had either sham surgery, where the joint was opened but key structures were left intact, or a mild ACL sprain that stressed but did not tear the ligament. Over ten weeks, the researchers repeatedly examined joint tissue under the microscope to score cartilage wear, bone changes, and inflammation in the joint lining.
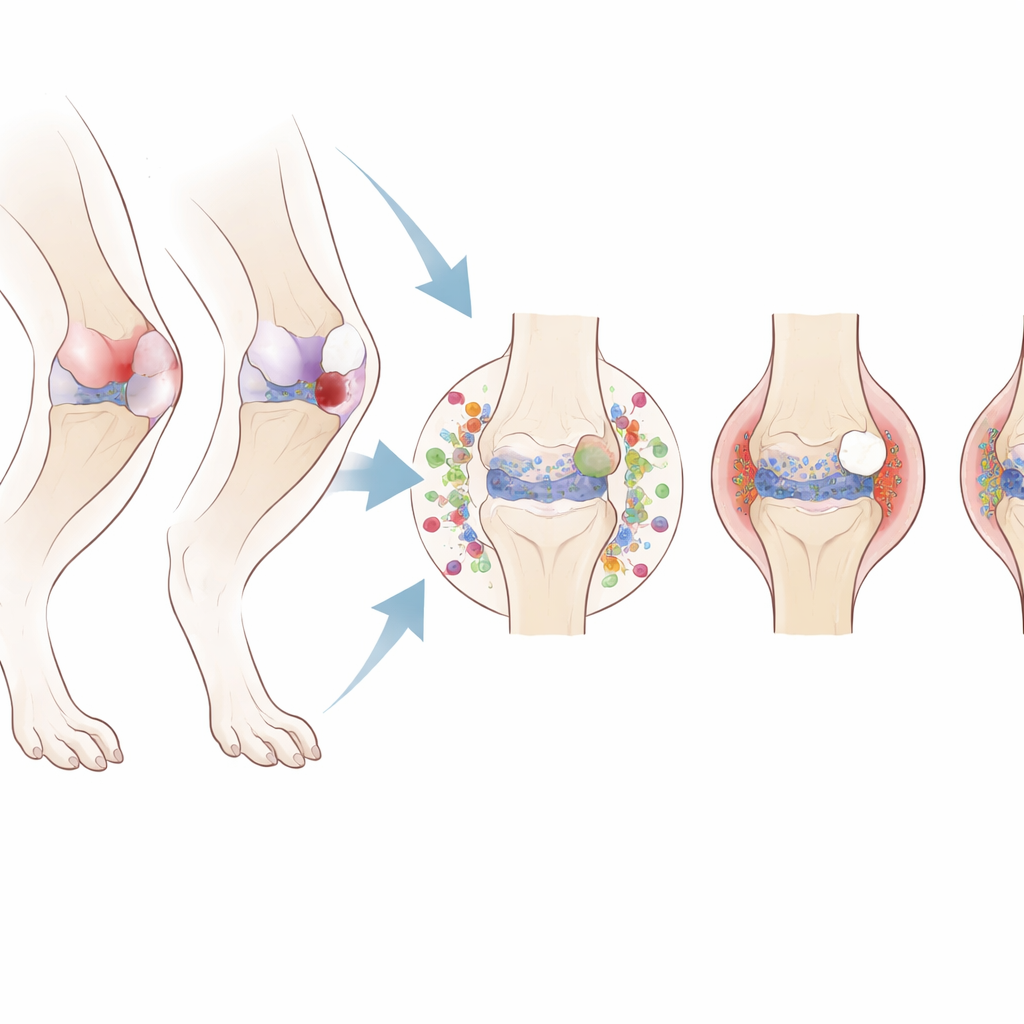
How the joint slowly breaks down
The structural changes in the knee followed distinct patterns depending on which tissue was injured. Both surgical and non‑surgical ACL ruptures produced rapid and severe cartilage damage, bony outgrowths at the joint edge, and erosion of the bone‑cartilage interface. In contrast, meniscus injury led to slower, milder cartilage wear but more thickening and hardening of the underlying bone. Sham surgery and the non‑tearing ACL sprain caused only minor, late‑appearing changes. These findings show that simply opening the joint or briefly over‑loading it is not enough to trigger full‑blown post‑traumatic osteoarthritis; substantial damage to either the ACL or the meniscus is required.
The immune system moves into the joint
To understand the biology behind these different outcomes, the investigators used flow cytometry—an advanced cell‑counting method—to track immune cells in the joint lining, nearby lymph nodes, spleen, and bone marrow. All injuries caused some early inflammation, but joints destined for osteoarthritis showed a much stronger and more sustained response. Right after ACL or meniscus damage, large numbers of circulating monocytes—precursor cells that can become inflammatory macrophages—left the bone marrow and spleen and appeared in the synovial lining of the injured knee. There they adopted a pro‑inflammatory state, known as M1‑like, and remained elevated for weeks. At the same time, certain helper T‑cell subsets associated with a more aggressive immune profile (often called Th1 and Th17) accumulated in the joint, especially after ACL rupture.
Local fire, limited spillover
One might expect such a strong reaction to spread throughout the body, but the study found that most of the action stayed near the injured joint. Changes in immune cells within the spleen and distant bone marrow were generally short‑lived, while the synovium and the draining lymph node next to the injured leg showed the clearest and most persistent shifts. Interestingly, how the ACL was damaged—by surgery or by mechanical loading—mattered far less than which tissue was injured. ACL‑tearing models behaved similarly to each other and quite differently from the meniscus model, underscoring that “what you injure” is more important than “how you injure it” for shaping the immune response and the eventual pattern of joint damage.
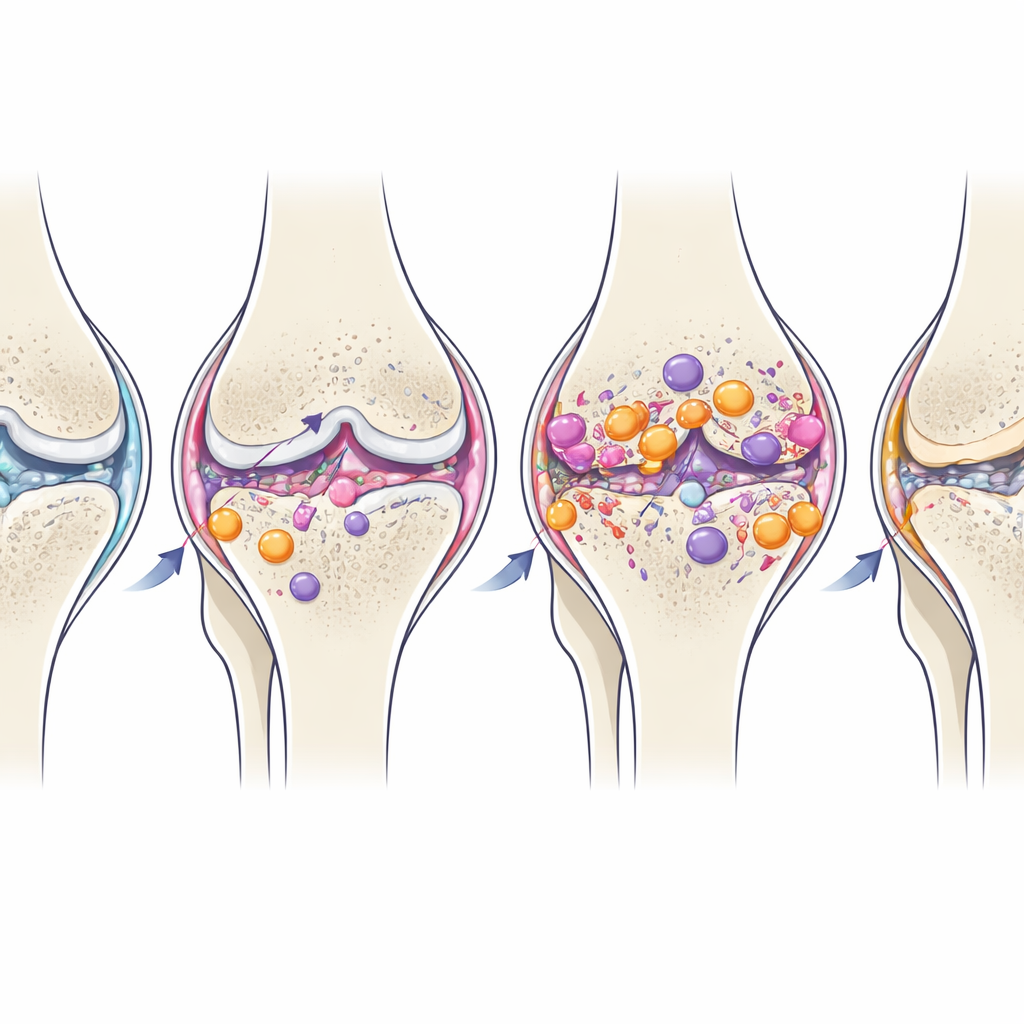
When inflammation turns from helper to harm
Over time, some of the joint’s resident macrophages gradually shifted toward a more soothing, M2‑like profile, even though structural damage continued to worsen. This natural late switch suggests the joint attempts to resolve inflammation, but in post‑traumatic osteoarthritis it comes too late or is too weak to prevent ongoing cartilage loss and bone remodeling. Persistent elevation of Th17‑type T cells in the joint, long after the initial injury, hints that these cells may help keep the inflammatory environment smoldering.
What this means for injured knees
For a general reader wondering why one person’s sports injury heals while another’s becomes a lifelong problem, this work offers a key insight: the immune system’s early, local reaction to injury can help decide the joint’s fate. In these mouse models, injuries that ultimately caused post‑traumatic osteoarthritis were marked by a strong, prolonged influx of inflammatory monocytes and macrophages and a skewed helper T‑cell response in and around the injured knee, with only modest whole‑body involvement. These findings point toward new treatment strategies that go beyond surgery and rehabilitation—such as timely drugs that rebalance joint‑resident immune cells in the days and weeks after injury, potentially reducing the chance that a bad sprain or tear will turn into chronic osteoarthritis.
Citation: Haubruck, P., Colbath, A.C., Blaker, C.L. et al. Comparison of innate and adaptive immune responses to joint injury during the onset and progression of post-traumatic osteoarthritis in common murine models. Sci Rep 16, 9767 (2026). https://doi.org/10.1038/s41598-026-40338-4
Keywords: post-traumatic osteoarthritis, knee injury, joint inflammation, immune cells, mouse model