Clear Sky Science · en
Functional analysis of major detoxification gene superfamilies driving flubendiamide resistance in South Indian Tuta absoluta field populations
Why this tiny moth matters for your tomatoes
The tomato pinworm, Tuta absoluta, is a tiny moth whose larvae can wipe out entire tomato fields by tunneling through leaves and fruits. Around the world, farmers have turned to powerful modern insecticides to keep it in check. One of the most important of these chemicals, flubendiamide, is now losing its punch in parts of South India. This study asks a pressing question for food security: how is this pest learning to survive a once-reliable pesticide, and what does that mean for keeping tomatoes on our tables?
From miracle spray to fading shield
When flubendiamide was introduced to Indian tomato fields in 2009, it quickly became a frontline defense because it is highly selective for insects and viewed as safer for people and beneficial organisms. But heavy, repeated use created strong evolutionary pressure on Tuta absoluta populations. In South India, one population from the Krishnagiri region now shows especially high survival when exposed to this chemical. The researchers compared this resistant population with a laboratory strain that still dies at low doses, exposing both to carefully chosen sublethal amounts of flubendiamide to see how their internal chemistry responded.
Digging into the pest’s internal toolbox
Like people and other animals, insects rely on a battery of enzymes to break down foreign chemicals. Three major enzyme families are known detoxifiers: cytochrome P450s, glutathione S-transferases (GSTs), and carboxyl/choline esterases. The team first mapped where Tuta absoluta’s versions of these genes sit on the broader insect family tree. They showed that the moth carries P450 and GST genes closely related to detoxification workhorses in other crop pests, hinting that similar survival tricks might be at play. Carboxylesterase genes, another common resistance route in insects, split into two groups—those tied to detoxification and those involved in nerve and developmental functions.
Which genes switch on when the spray hits
To see which detox tools the resistant moths actually deploy, the scientists measured gene activity in larvae 24 and 48 hours after flubendiamide exposure. In the resistant Krishnagiri population, several P450 genes surged far above the levels seen in the susceptible strain. One gene in particular, CYP248f, jumped more than tenfold at 24 hours and rose even higher by 48 hours, while CYP724c and CYP272c also showed strong, sustained increases. A few GST genes behaved in a similar way: members of the epsilon and delta classes (TaGSTe and TaGSTd) became markedly more active in resistant larvae, especially soon after exposure. In contrast, the carboxylesterase genes tested (TaCCE1 and TaCCE2) barely changed, suggesting they contribute little to this specific form of resistance.
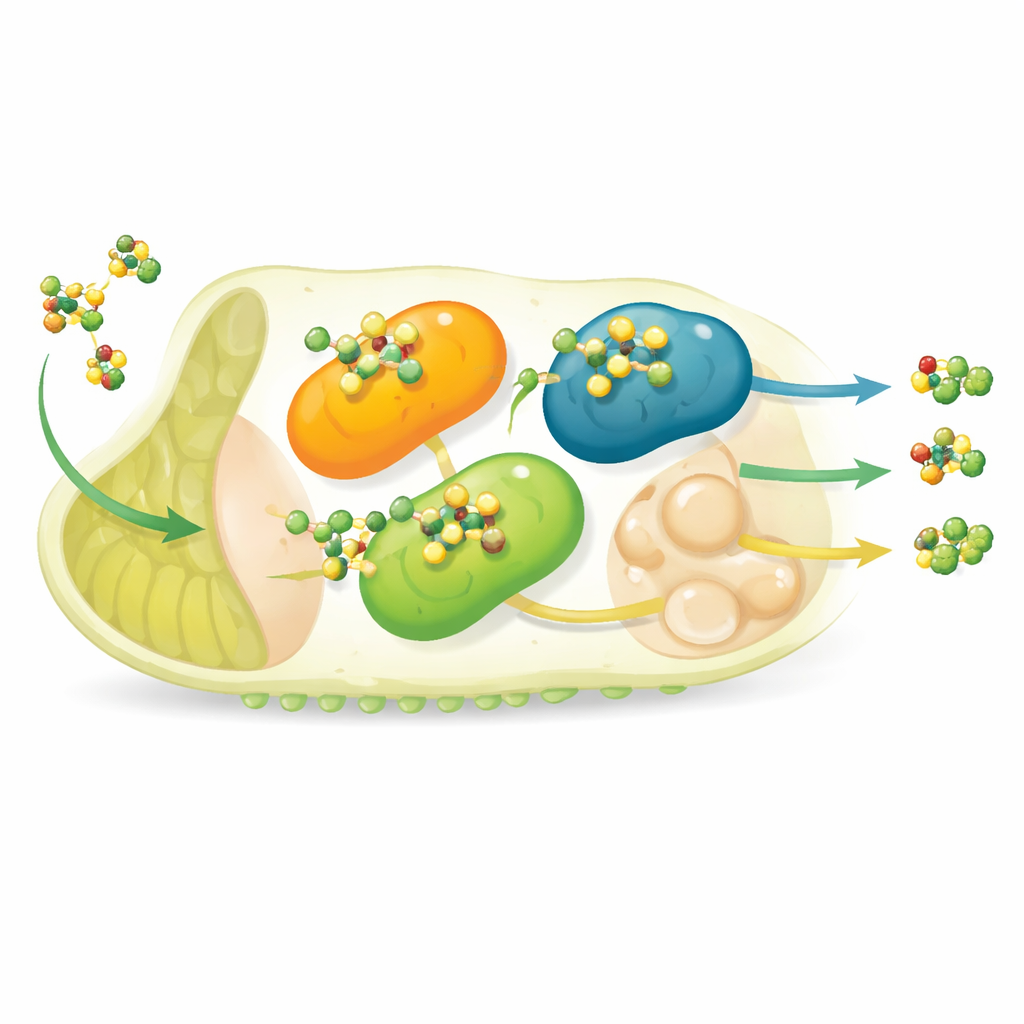
Testing how tightly the insecticide sticks
Beyond gene activity, the team wanted to know how well the proteins encoded by these genes physically interact with flubendiamide. Using computer-based molecular docking, they modeled how the insecticide fits into the three-dimensional shapes of each detox enzyme, estimating how tightly it would bind. The standout again was CYP248f, which showed the strongest predicted binding and formed several stabilizing hydrogen bonds with the compound—features consistent with an efficient detox machine. Among the GSTs, TaGSTe and TaGSTd showed similarly strong binding, while the others and the carboxylesterases bound more weakly. Together with the expression data, this points to a small set of P450 and GST proteins as the main engines of flubendiamide breakdown inside resistant larvae.
What this means for future tomato crops
For non-specialists, the key message is that Tuta absoluta is not just "getting used to" flubendiamide in a vague sense. Its cells are rewiring which genes they use, dialing up specific detox enzymes that grab and neutralize the insecticide before it can do harm. By pinpointing the main culprits—especially CYP248f and certain GSTs—this work provides molecular fingerprints that can be monitored in field populations to spot resistance early. It also offers targets for designing better control strategies, such as rotating chemicals with different weak points or combining treatments that block these detox pathways. In short, understanding the pest’s internal chemistry offers a roadmap for staying one step ahead in the fight to protect tomato harvests.
Citation: Mohan, M.L.B.C., Marimuthu, M., Venkatasamy, B. et al. Functional analysis of major detoxification gene superfamilies driving flubendiamide resistance in South Indian Tuta absoluta field populations. Sci Rep 16, 9419 (2026). https://doi.org/10.1038/s41598-026-40310-2
Keywords: tomato pinworm, insecticide resistance, flubendiamide, detoxification enzymes, tomato pests