Clear Sky Science · en
Lectin-based detection and expression profiling of native glycoRNAs
RNA Wear Sugar Coats
Our cells carry vast numbers of RNA molecules, best known as the messengers that help turn genes into proteins. In recent years, scientists have discovered a surprising twist: some RNAs are decorated with complex sugars and even appear on the outside of cells. These “sugar‑coated” RNAs, called glycoRNAs, seem to influence how immune cells move, how cancers spread, and how our bodies respond to infection. The study summarized here introduces a simpler way to spot these elusive molecules and maps where they appear in the body, opening doors to new diagnostics and therapies.
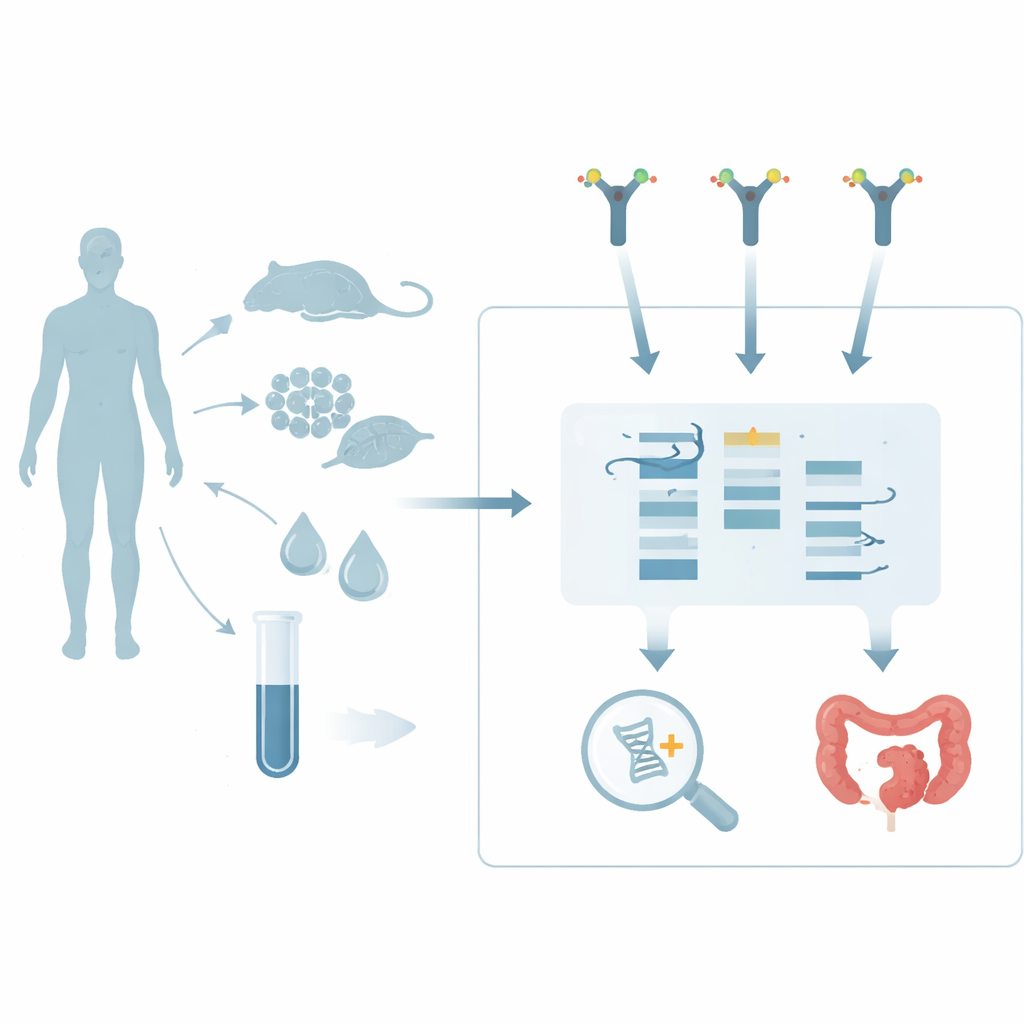
A New Way to Spot Sugar‑Coated RNA
Until now, finding glycoRNAs has required technically demanding methods. One approach forces living cells or animals to take up artificial sugar building blocks that later show up on newly made glycans, which can then be tagged and visualized. Another, called rPAL, chemically tweaks certain sugars on purified RNA so they can be detected. Both strategies are sensitive but have drawbacks: one depends on live systems that absorb the probe, the other only sees a subset of glycoRNAs with specific sugar tips. The authors of this paper developed a more straightforward alternative that works directly on extracted RNA. Their method, lectin‑based detection (LBD), relies on lectins—naturally occurring proteins that recognize particular sugar shapes—to bind glycoRNAs on a blot, much as antibodies bind proteins.
Fine‑Tuning a Simple Laboratory Workflow
To build LBD, the team first isolated total RNA from cultured human monocytes and separated the molecules by size using a standard northern blot procedure. They then tested 24 different lectins for their ability to highlight a distinct glycoRNA band, comparing the results to rPAL as a reference. Several lectins worked, but one derived from tomato, called LEL, gave especially strong signals and became their probe of choice. The researchers then optimized routine steps—how RNA is transferred to membranes, which membrane material to use, how to block background sticking, how long to incubate, and how much lectin to apply—until the signals were both sharp and reproducible, all while keeping the protocol simple enough for a typical molecular biology lab.
Checking Sensitivity, Specificity, and What Is Being Seen
A key question was whether LBD is both sensitive and truly specific for glycoRNAs. By comparing LBD side‑by‑side with metabolic labeling and rPAL across a range of RNA amounts, the authors found that LBD could detect as little as about half a microgram of total RNA, on par with the established methods. To test specificity, they treated RNA samples with enzymes that cut RNA, DNA, proteins, or the N‑linked sugars that decorate glycoRNAs. Only treatments that destroyed RNA itself or removed its sugar chains erased the signal, while DNA‑cutting or protein‑digesting enzymes had no effect. Drugs that block glycan assembly inside cells also sharply reduced the LBD signal. Together, these tests show that the lectin‑based method is truly responding to sugar‑modified RNAs rather than to contaminants.
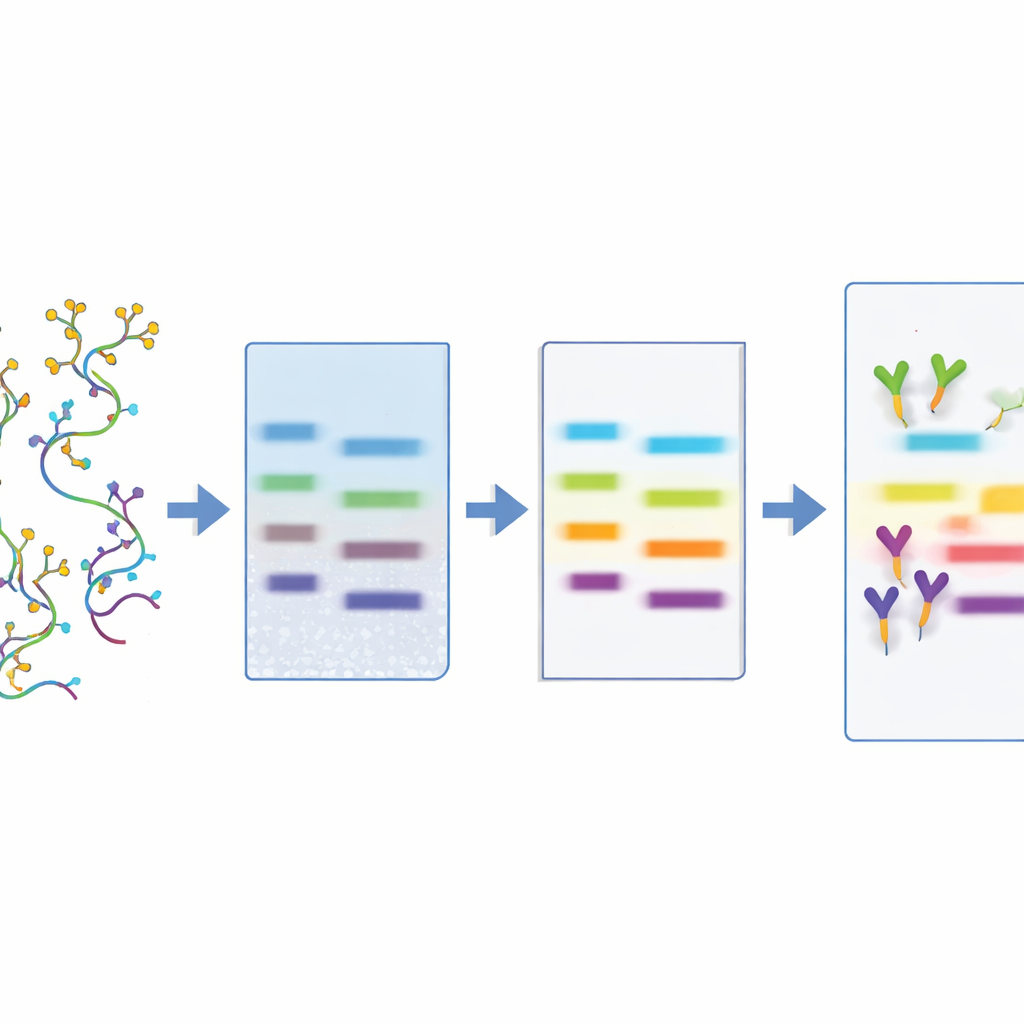
Where Sugar‑Coated RNAs Show Up in Health and Disease
With the method in hand, the team surveyed where glycoRNAs appear in a broad set of cells, tissues, and body fluids from mice, rats, and humans. They found especially strong signals in immune‑related cells (such as monocytes, neutrophils, and white blood cells), in organs that form barriers to the outside world (like the gut and respiratory tract), and in certain regions of the brain and heart. Intriguingly, glycoRNAs were absent or very low in some major organs, including liver, kidney, and skeletal muscle. LBD also revealed multiple bands in some tissues, and different lectins recognized different subsets of bands, hinting that there are diverse glycoRNA “flavors” with distinct sugar decorations. Importantly, the authors detected free glycoRNAs in human plasma, urine, feces, and amniotic fluid, implying that these molecules circulate outside cells and might be sampled through relatively noninvasive tests.
Cancer Links and Future Medical Uses
The study also probed how glycoRNA patterns change in cancer. In matched human tissue samples, breast and colon tumors showed higher levels of glycoRNAs than their normal counterparts, with the strongest signals in metastatic disease. In metastatic breast cancer, the glycoRNA bands migrated differently on gels compared with those from primary tumors, suggesting structural changes that might track with disease progression. Such tumor‑associated variants could ultimately help distinguish aggressive cancers or guide targeted therapies. Because LBD uses readily available reagents and avoids live‑animal labeling steps, it provides a practical way to screen many samples and compare them across methods, revealing how different detection strategies highlight overlapping but not identical sets of glycoRNAs.
Why This Matters for Everyday Health
In plain terms, this work offers a toolkit for seeing a newly recognized class of biomolecules that help cells talk to each other, especially in the immune system and at body surfaces that face microbes and toxins. By showing that a simple sugar‑binding probe can reliably reveal glycoRNAs across tissues, in body fluids, and in cancers, the study lays groundwork for turning these molecules into indicators of inflammation, infection, or tumor spread. While more sensitive clinical assays and deeper functional studies are still needed, lectin‑based detection makes it far easier for many laboratories to explore where glycoRNAs live and what roles they play in health and disease.
Citation: Li, Y., Qian, Y., Li, X. et al. Lectin-based detection and expression profiling of native glycoRNAs. Sci Rep 16, 9031 (2026). https://doi.org/10.1038/s41598-026-40291-2
Keywords: glycoRNA, lectin-based detection, cell-surface RNA, biomarkers, cancer metastasis