Clear Sky Science · en
Ultra-intense pulsed source of ionizing radiation based on direct laser acceleration of electrons for studying the FLASH effect
Why ultra-fast radiation pulses matter
Cancer radiotherapy walks a tightrope: deliver enough radiation to kill tumors without causing lasting harm to healthy tissue. A recent and exciting idea, called the FLASH effect, suggests that giving radiation in an ultra-short, ultra-intense burst can spare normal tissue while still attacking cancer. This study presents a new kind of experimental radiation source based on a powerful laser that creates extremely brief bursts of high-energy electrons, and uses it to watch how oxygen in water and biological liquids is suddenly used up during irradiation – a process thought to be central to the FLASH effect. 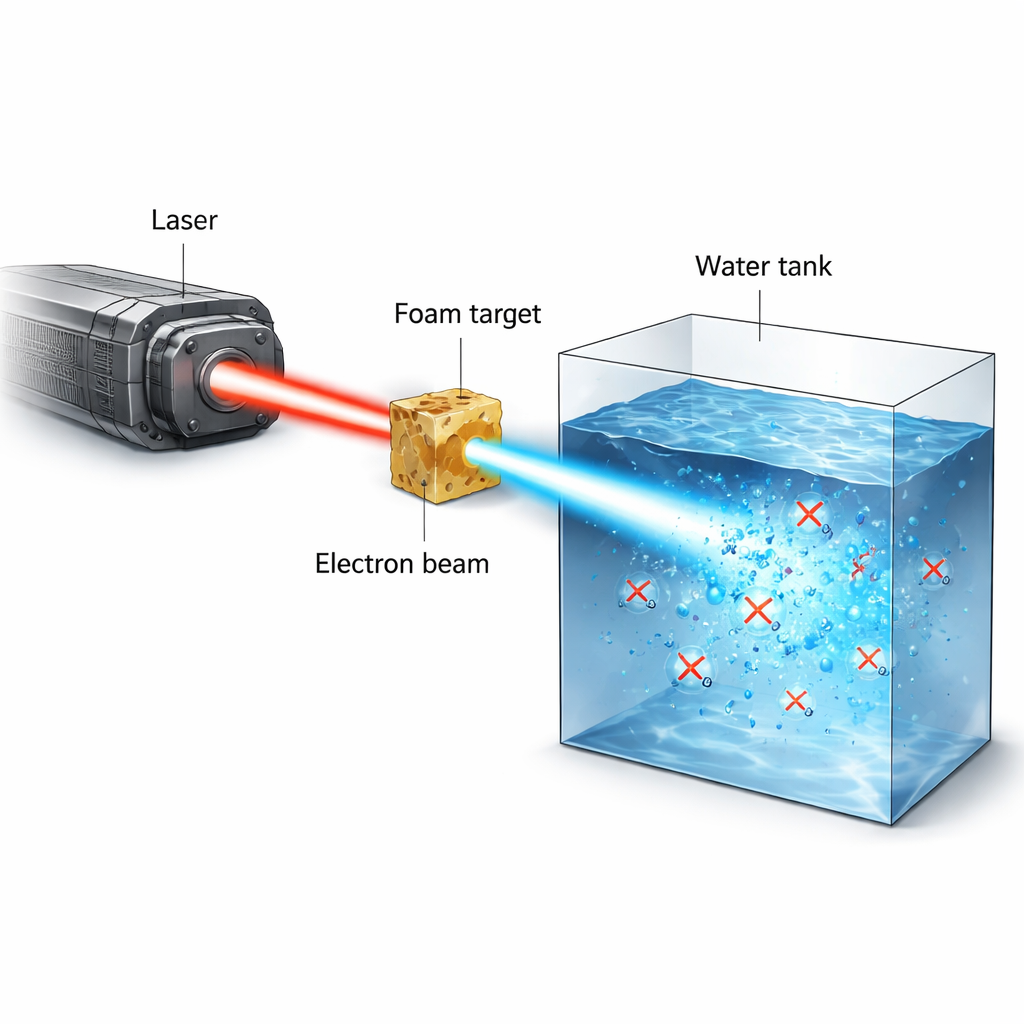
From hospital machines to laser-driven beams
Conventional radiotherapy uses X-rays, electrons, or protons from large accelerators. These machines typically deliver dose over milliseconds to seconds, and while they are very precise, they still damage normal tissue because radiation indirectly breaks DNA by splitting water molecules and generating reactive oxygen species. Oxygen in healthy, well-vascularized tissue actually makes this damage worse – the so-called oxygen effect. FLASH radiotherapy aims to flip this script by delivering the full therapeutic dose in a fraction of a second at ultra-high dose rates, which seems to temporarily protect normal tissue in animal studies without shielding tumors. However, standard medical accelerators struggle to reach these extreme dose rates, motivating the search for alternative sources such as high-power lasers.
Turning foam and light into a powerful electron burst
The researchers used the PHELIX high-power laser to create narrow, high-current beams of electrons with energies of several million electron volts (MeV). The laser first ionizes a low-density polymer foam into a near-critical plasma. In a second, ultra-short pulse lasting less than a trillionth of a second, the laser pushes and traps electrons inside a plasma channel, directly accelerating them to high energies. This process, called direct laser acceleration, produces a tightly collimated beam that can carry tens of nanocoulombs of charge in a picosecond-scale burst. When this beam strikes water or water-like material, it can deposit 20–50 gray of radiation dose in a single shot, at instantaneous dose rates above 10¹³ gray per second – far beyond what conventional machines can achieve.
Watching oxygen disappear in a flash
To probe how such intense bursts affect chemistry in tissue-like conditions, the team built sealed water tanks that could operate in vacuum and be filled with either pure water, cell culture medium, or lysed cells, all fully oxygenated beforehand. The electron beam was shaped and filtered so that high-energy electrons dominated the dose inside the tank, while protons and X-rays were largely suppressed or carefully accounted for using layers of shielding and radiochromic films. An optical sensor placed on the inner wall of the tank monitored dissolved oxygen by measuring how oxygen molecules quenched the luminescence of a dye spot. After each single-pulse irradiation, the sensor recorded a sudden drop in oxygen concentration that then slowly relaxed as oxygen diffused within the sensor region. By combining film data, simulations, and the known stopping power of electrons, the authors reconstructed the dose delivered to the irradiated volume and linked it directly to the measured oxygen loss. 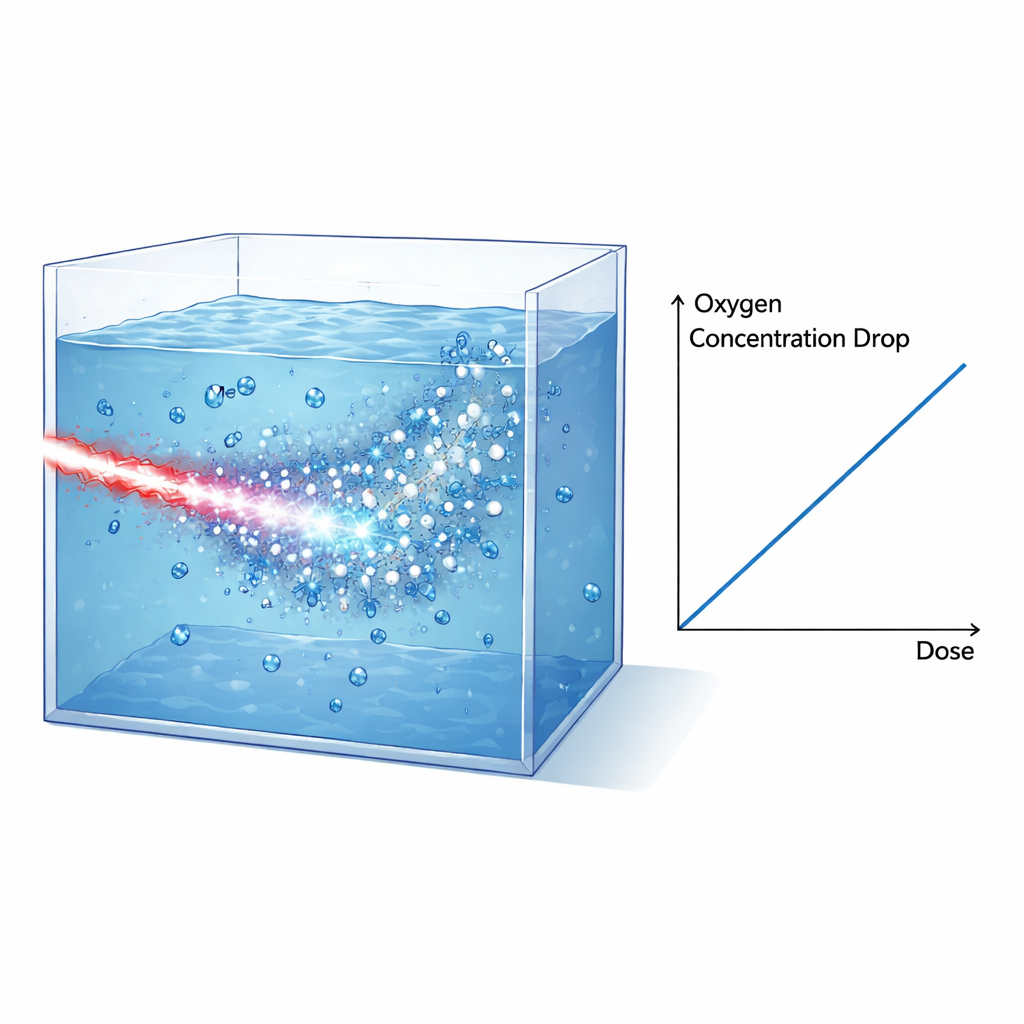
Linking experiment and theory
The key finding is that the decrease in dissolved oxygen is proportional to the delivered dose for both water and culture medium, with very similar slopes. After correcting for a small additional dose from X-rays, the measured oxygen consumption in water is about 0.32 micromoles per liter per gray. This value matches remarkably well with predictions from detailed Monte Carlo track-structure simulations (TRAX-CHEM), which follow the paths of electrons in water and model the rapid chemistry that unfolds within microseconds after irradiation. Crucially, in this laser-driven setup, almost the entire dose is delivered within about one picosecond – much shorter than the characteristic times for the ensuing chemical reactions. That means the experiment closely reproduces the ideal conditions assumed in these simulations, providing a stringent test of the underlying models.
Building a better testbed for FLASH science
Beyond validating theory, the authors use their results to design an improved experimental layout. By simplifying the geometry, eliminating magnets, and symmetrically placing water and dose-measuring films around the beam, future setups could directly measure the dose inside the tank without complex reconstruction, while further suppressing unwanted proton and X-ray contributions. Simulations show that modifying the target – for example, adding thin plastic or gold layers – allows the dose per shot to be tuned between roughly 40 and 80 gray, offering a flexible platform to explore a wide range of conditions relevant for FLASH.
What this means for future cancer treatments
For non-specialists, the core message is that this laser-based source creates extremely intense, ultra-short electron pulses that mimic – and in some ways surpass – the conditions envisioned for FLASH radiotherapy. The study shows that these pulses rapidly consume dissolved oxygen in water and biological media in a way that agrees with advanced theoretical predictions. Since oxygen depletion and related radical chemistry are leading candidates to explain why FLASH may spare healthy tissue, having a controllable, well-understood source like this is a major step forward. It provides a powerful testbed for refining models and, ultimately, for guiding the design of future clinical machines that could treat cancer more effectively while reducing side effects.
Citation: Gyrdymov, M., Bukharskii, N., Fabian, V. et al. Ultra-intense pulsed source of ionizing radiation based on direct laser acceleration of electrons for studying the FLASH effect. Sci Rep 16, 7164 (2026). https://doi.org/10.1038/s41598-026-40281-4
Keywords: FLASH radiotherapy, laser-driven electrons, ultra-high dose rate, oxygen depletion, water radiolysis