Clear Sky Science · en
Selective clearance of monoclonal antibodies via the mannose receptor is dependent on glycan pairing
Why sugar patterns on medicines matter
Many of today’s blockbuster drugs are monoclonal antibodies—engineered proteins that can stay in the bloodstream for weeks and precisely target diseases such as cancer and autoimmune disorders. But not all antibodies behave the same once injected. This study asks a deceptively simple question with big consequences: how does the fine “sugar pattern” attached to an antibody decide how quickly the body removes it, and could this also shape side effects like immune reactions?
Tiny sugars as traffic signals
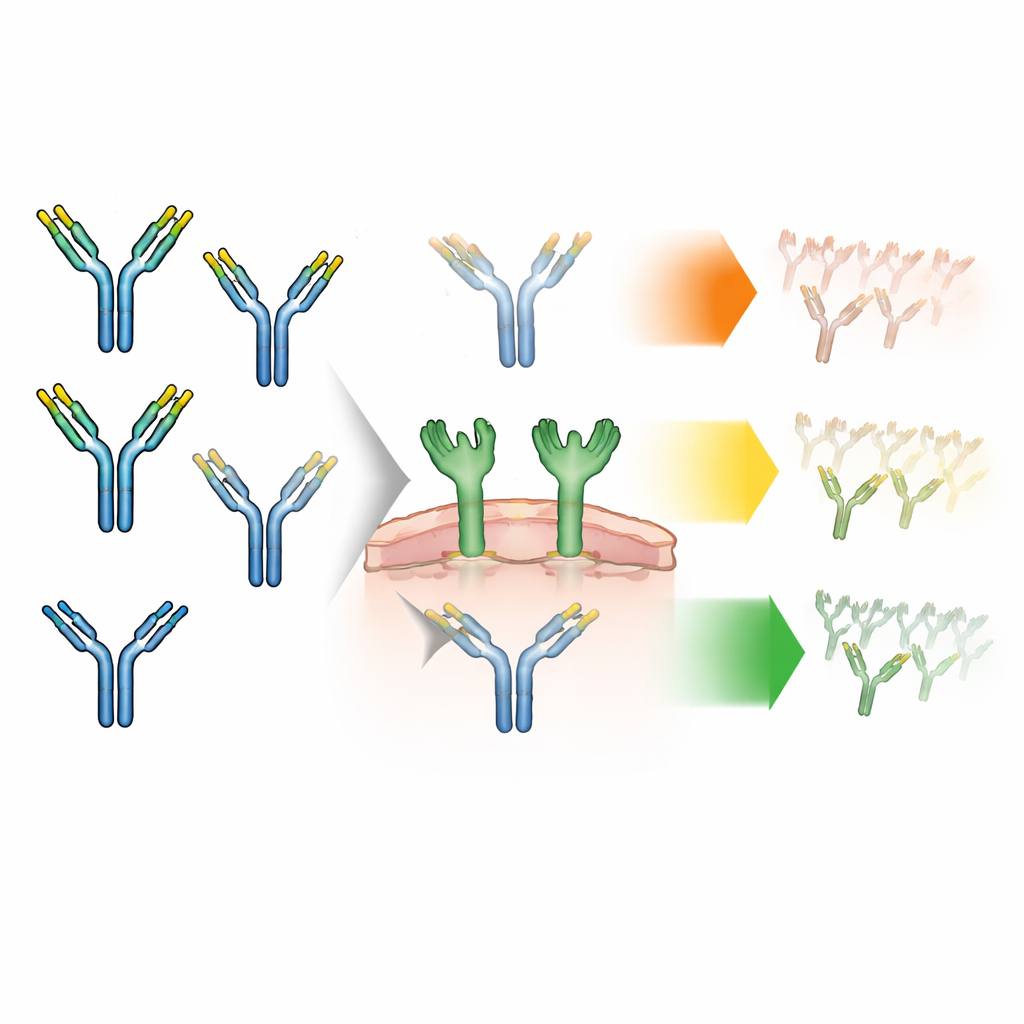
Antibodies naturally carry small chains of sugars—called glycans—on their backbone. These glycans come in different flavors. Most are “complex” shapes, but a minority are “high-mannose,” a simpler type that previous work had linked to faster removal from the blood. Each antibody has two sugar sites that act as a pair, and these can match (symmetrical pairs) or differ (asymmetrical pairs). Until now, drug makers mostly counted how many of each sugar type was present overall, without asking how the two sugars on the same antibody were paired. The authors suspected that this pairing pattern could change how strongly antibodies interact with a special sugar-sensing protein on immune cells called the mannose receptor—and thus how fast the body clears the drug.
Sorting antibodies by their sugar pairs
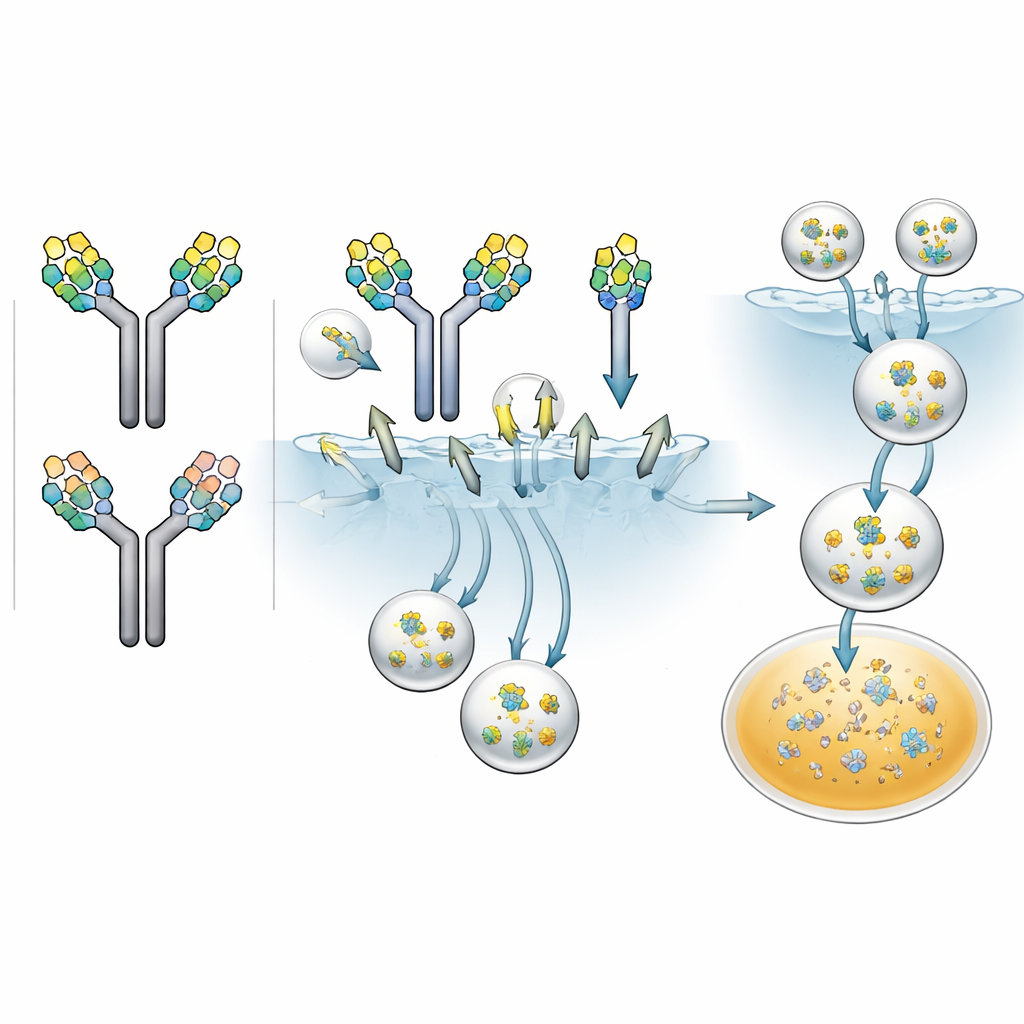
To probe this, the team engineered antibodies enriched in high-mannose sugars and then physically separated them into three main groups: antibodies with two complex sugars, with one complex and one high-mannose sugar (asymmetrical), or with two high-mannose sugars (symmetrical). They carefully verified that these groups were almost identical in every other way: same size, shape, charge, and stability, and similar binding to the recycling receptor that normally protects antibodies from degradation. That meant any differences they later observed in cell experiments or in animals could be traced back to sugar pairing alone, not to hidden damage or impurities.
Watching cells pull antibodies inside
Next, the researchers asked how quickly each sugar pattern allowed antibodies to be swallowed by cells that carry the mannose receptor. They labeled the different antibody fractions with a fluorescent tag and incubated them with human cells engineered to display this receptor. Over nearly three days, they tracked the glow inside the cells as a measure of uptake. All antibodies were taken up to some extent, but the rates differed: those with two high-mannose sugars were internalized the fastest, those with one high-mannose sugar were intermediate, and those with only complex sugars were slowest. When the team added mannan—a natural sugar that competes for the mannose receptor—the uptake of the high-mannose antibodies dropped sharply, confirming that this receptor was responsible for much of the rapid internalization.
Following sugar-marked antibodies in live animals
The crucial test was whether these cellular differences would translate into real-world drug behavior. The scientists formulated a drug product rich in both symmetrical and asymmetrical high-mannose pairs, along with complex-only pairs, and injected it into rats. Using sensitive mass spectrometry methods, they could track not just the total antibody level over two weeks, but also how each sugar pairing declined over time. The pattern was striking: antibodies with two high-mannose sugars disappeared the fastest, with a half-life of about 2.4 days; those with one high-mannose sugar lasted longer, about 7.2 days; and those with only complex sugars persisted for roughly 17.4 days. Exposure over time—the area under the concentration curve—fell to 38% for the double high-mannose pair and 73% for the mixed pair compared with the complex-only pair. In essence, each extra high-mannose sugar acted like an additional “clear me” flag for the mannose receptor.
What this means for safer, longer-lasting drugs
For patients, these results clarify why two antibody drugs that look similar on paper can behave differently in the body. The study shows that not just the presence but the pairing of high-mannose sugars strongly steers how quickly antibodies are removed, likely through mannose-receptor-driven uptake and breakdown inside immune cells. Because the same pathway can also feed antigens into the machinery that triggers immune responses, these sugar patterns may influence the risk of anti-drug antibodies and allergic reactions as well. The authors argue that manufacturers should monitor and control glycan pairing—not just overall sugar composition—when designing and testing antibody therapies. Doing so could help ensure more predictable dosing, longer-lasting benefits, and a lower chance of unwanted immune responses.
Citation: Baumeister, J., Meudt, M., Wei, W. et al. Selective clearance of monoclonal antibodies via the mannose receptor is dependent on glycan pairing. Sci Rep 16, 8489 (2026). https://doi.org/10.1038/s41598-026-40268-1
Keywords: monoclonal antibodies, glycosylation, mannose receptor, pharmacokinetics, immunogenicity