Clear Sky Science · en
A modular platform for automated organoid culture and longitudinal imaging
Mini Organs on a Chip
Imagine being able to watch tiny, lab‑grown versions of human organs develop in real time, like a slow‑motion movie of how our tissues grow, get sick, or respond to medicines. That is the promise of organoids—3D cell clusters that mimic real organs. This paper describes a new benchtop platform that can automatically keep brain organoids alive, fed, and carefully watched for days at a time, without needing a bulky incubator. It opens the door to more reliable disease models, smarter drug testing, and eventually more personalized medicine.
Why Growing Mini Organs Is So Hard
Organoids have become powerful tools for studying the brain, gut, kidney, and more, because they capture many of the structures and cell types found in real organs. But keeping them healthy is surprisingly tricky. Standard methods rely on people manually changing the liquid that feeds the cells and placing flasks on shaking platforms inside warm, humid incubators. This setup is labor‑intensive and can be inconsistent from lab to lab. Worse, the cramped, wet environment inside an incubator makes it hard to place cameras and other electronics nearby, so researchers usually cannot continuously watch how the tissue changes over time. Existing microfluidic systems improve control of the feeding liquid, but most still live inside those same incubators, limiting long‑term imaging.
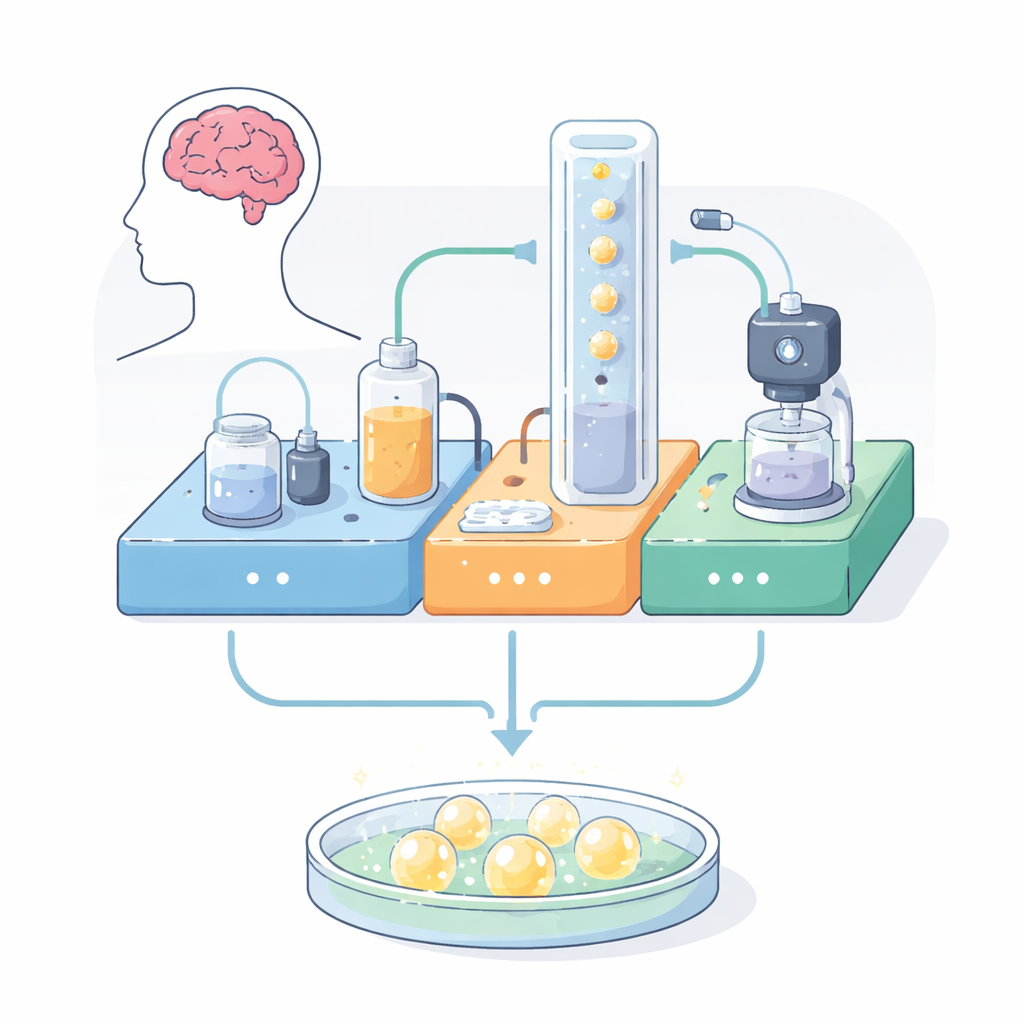
A Self‑Contained Life Support System
The authors built a modular platform that brings together three functions normally scattered across different pieces of equipment: automated feeding, live imaging, and environmental control. Everything is mounted on a compact, perforated metal board using 3D‑printed holders and off‑the‑shelf parts. One module pumps warm nutrient liquid through a closed loop, recirculating it past the organoids while filtering out impurities. A second module constantly adjusts temperature and acidity, using a heater and controlled carbon dioxide bubbles to keep conditions close to those in the body. A third module holds a small digital microscope near the culture chamber, capturing bright‑field and fluorescent images and even detecting common green and red glowing tags inside cells. All three modules talk to each other through simple electronics, allowing long, unattended runs.
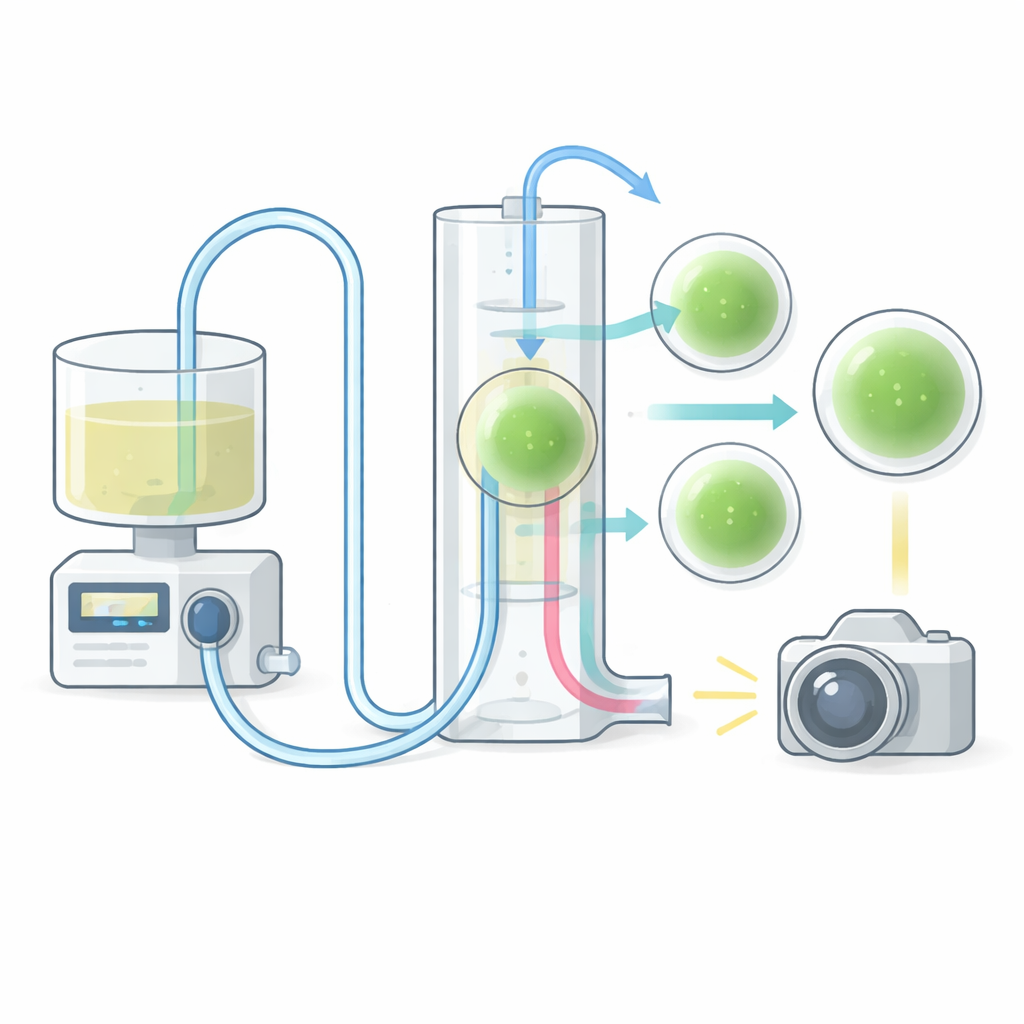
The Vertical Chip That Shows Everything
At the heart of the system is a custom culture chamber: a clear silicone well bonded to a glass microscope slide. Unlike traditional flat plates, this chip is mounted vertically. That unusual orientation lets researchers watch how liquid actually flows around and past each organoid, instead of just seeing a top‑down snapshot. The well includes inlet and outlet channels placed high enough to keep the samples from washing out, and the opening is large enough to load organoids with a standard lab pipette while still allowing gas exchange. The chip can be fabricated in less than a day using 3D‑printed molds and inexpensive materials, and it can be configured as a single large well or as multiwell versions that hold several organoids either in series or in parallel. This flexibility makes it easy to scale experiments or compare different flow patterns.
Do the Mini Brains Stay Healthy?
To see whether the platform actually supports living tissue, the team grew mouse brain organoids and split them between standard incubator conditions and the new device. After six days, they stained the organoids with a dye that marks live cell membranes and checked their structure with antibodies against a neuron‑specific protein. Organoids on the platform were just as viable and structurally organized as those in the incubator, across all chip designs. They also measured key nutrients and salts—such as glucose, sodium, potassium, calcium, and chloride—in the culture liquid. Levels stayed stable and statistically indistinguishable from incubator controls, showing that the automated flow did not stress the tissue. Time‑lapse images revealed steady growth in organoid size, and the multiwell chips reduced unwanted fusion of neighboring organoids, a common problem in traditional cultures.
Watching Nutrients Move in Real Time
The platform is not only a life support system; it is also a window into how molecules move through 3D tissue. In one experiment, the researchers pulsed a fluorescent dye through the chip and tracked its uptake into different regions of an organoid over several minutes. The resulting brightness patterns matched computer simulations of fluid flow and diffusion, confirming that the device can capture dynamic transport processes with fine detail. This ability to align real movies of dye or drug penetration with predictive models could help optimize how therapies are delivered and how pattern‑forming signals are applied during development‑like experiments.
What This Means Going Forward
In plain terms, this work delivers a compact, affordable “mini‑incubator on a board” that can grow brain organoids automatically while filming them and measuring their environment. It overcomes the usual trade‑off between keeping tissues in a realistic, well‑controlled state and being able to watch what is happening inside them. While the current study focuses on mouse brain tissue over about a week, the same approach could be extended to human organoids and longer experiments, providing a more faithful and informative test bed for studying brain development, neurological disease, and new drugs.
Citation: Torres-Montoya, S., Hernandez, S., Seiler, S.T. et al. A modular platform for automated organoid culture and longitudinal imaging. Sci Rep 16, 9717 (2026). https://doi.org/10.1038/s41598-026-40231-0
Keywords: organoids, brain models, microfluidics, live cell imaging, automated cell culture