Clear Sky Science · en
Bi-functional Caulis polygoni multiflori inhibits Staphylococcus aureus and potentiates the activity of erythromycin in vitro
Why a climbing herb matters for stubborn infections
Drug-resistant “superbug” infections, especially those caused by Staphylococcus aureus on the skin and in wounds, are increasingly hard to treat. Many patients need powerful last‑line antibiotics that can be costly and toxic. This study explores whether a traditional Chinese medicinal vine, Caulis Polygoni Multiflori (CPM), can help in two ways at once: by directly harming these bacteria and by making a common antibiotic, erythromycin, work better—even against tough methicillin‑resistant strains (MRSA). 
The growing problem of hard-to-kill germs
Staphylococcus aureus is a major cause of infections ranging from everyday boils to life‑threatening pneumonia and bloodstream infections. Its methicillin‑resistant form, MRSA, shrugs off many standard antibiotics and often hides inside protective communities called biofilms. In a biofilm, bacteria stick to surfaces such as wound tissue or medical devices and wrap themselves in a slimy matrix that blocks drugs and immune cells. This forces doctors to rely on last‑resort antibiotics like vancomycin and linezolid, which can have serious side effects and are not always effective. As some MRSA strains even begin to resist these drugs, researchers are looking for new strategies that either hit the bacteria in novel ways or weaken their defenses.
Turning to a traditional remedy
CPM, known as Shouwuteng, is the woody stem of a climbing plant long used in Chinese medicine for various health purposes. Unlike a single‑molecule antibiotic, CPM is a mixture of many natural compounds that may attack bacteria on multiple fronts. In this study, researchers prepared a simple water extract—much like a strong herbal decoction—and tested it against eight strains of Staphylococcus aureus, including four MRSA isolates from hospital patients. They measured how much CPM was needed to stop bacterial growth and how much was required to kill the bacteria outright. Across all strains, CPM consistently slowed and then eliminated free‑floating (planktonic) bacteria, and its effectiveness did not depend on whether the strain was resistant to common antibiotics.
Blocking the first foothold and early communities
Infections often begin when bacteria latch onto wound components such as fibrin, the mesh that helps clots form. The team found that CPM strongly reduced the ability of Staphylococcus aureus to bind to fibrin in a dose‑dependent fashion: the more CPM present, the fewer bacteria could attach. CPM also altered how the bacteria clumped together. At lower levels, it slightly discouraged aggregation, but at higher levels it drove bacteria into large, disordered clumps that settled out of the liquid. Microscopy showed that treated cells became smaller and their surfaces rougher. When the researchers looked at biofilm formation—the step where attached bacteria build a structured, protective layer—CPM again showed strong, dose‑dependent inhibition. It interfered with early attachment and, at higher concentrations, curtailed the growth of the sticky matrix and the maturation of biofilm “microcolonies.” However, once a mature biofilm was already in place, CPM could not break it down, likely because its bulky natural compounds had trouble penetrating deeply into the biofilm fortress. 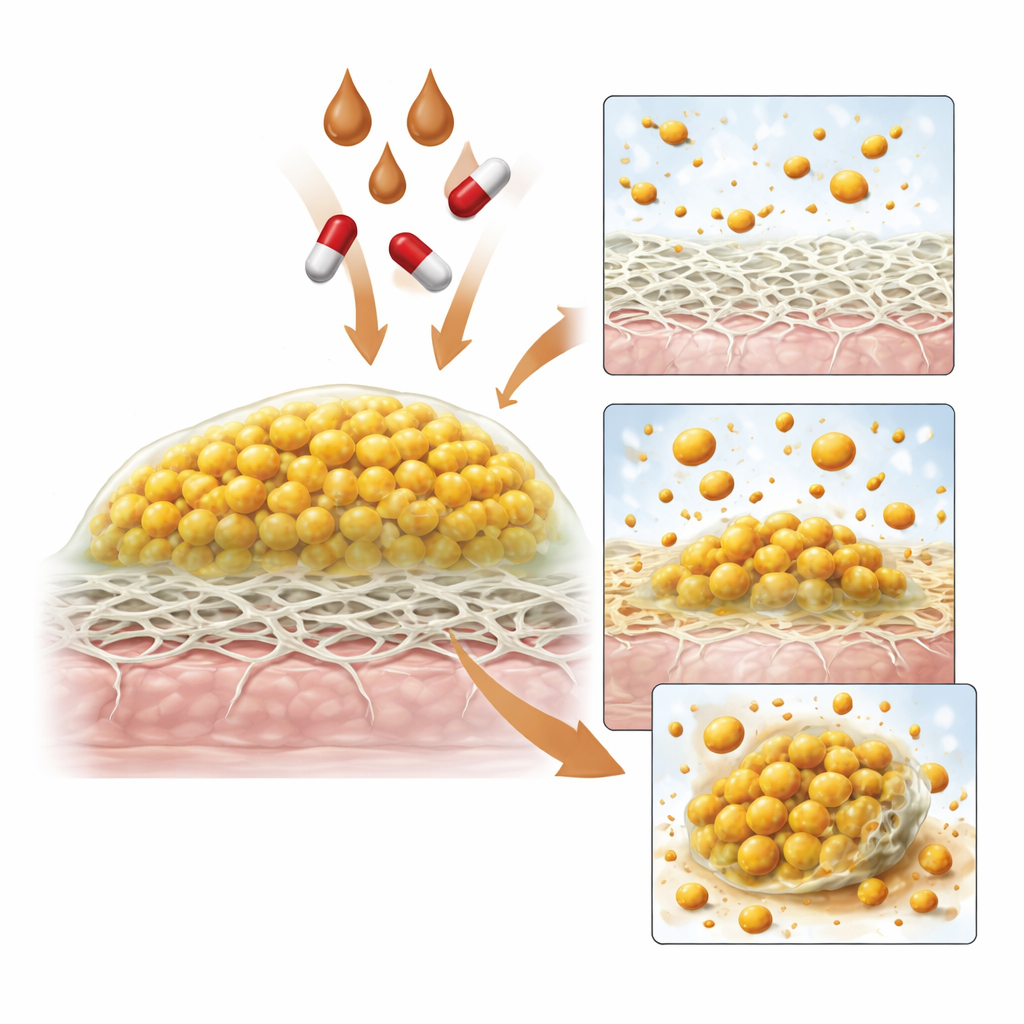
Helping an old antibiotic hit harder
Because doctors often combine drugs to outsmart resistant bacteria, the researchers asked whether CPM could work alongside standard antibiotics. In simple plate tests, CPM sometimes enlarged the zones where certain antibiotics, especially erythromycin and penicillin, stopped bacterial growth, hinting at synergy in some strains. To examine this more carefully, they used a broth “checkerboard” method, mixing many different concentration pairs of CPM and erythromycin. In liquid culture, CPM consistently boosted erythromycin’s performance against all eight strains. Together, the two agents achieved bacterial inhibition and killing at much lower doses than either alone, a pattern classified as synergistic or at least additive. This suggests that CPM may weaken bacterial defenses or virulence factors in ways that open the door for erythromycin to be effective again, even against MRSA.
What this could mean for patients
For a lay reader, the key message is that a traditional herbal extract, CPM, showed two valuable properties in the lab: it directly killed Staphylococcus aureus and it disarmed the bacteria by preventing them from firmly attaching and building protective biofilms. At the same time, it made an existing antibiotic, erythromycin, much more powerful in liquid culture, including against resistant MRSA strains. The extract did not dissolve established biofilms, and the work was done only in vitro with a limited number of strains, so it is not ready as a stand‑alone cure. Still, CPM emerges as a promising candidate for future topical treatments—such as creams or dressings for skin and wound infections—used alongside conventional antibiotics to both attack the germs and undercut their defenses.
Citation: Li, Z., Wang, W., Xu, W. et al. Bi-functional Caulis polygoni multiflori inhibits Staphylococcus aureus and potentiates the activity of erythromycin in vitro. Sci Rep 16, 9168 (2026). https://doi.org/10.1038/s41598-026-40228-9
Keywords: MRSA, biofilms, erythromycin synergy, traditional Chinese medicine, Caulis Polygoni Multiflori