Clear Sky Science · en
Caveolin-1 modulates Notch transcriptional activity during in vitro respiratory multiciliated cell maturation
Why the tiny hairs in your airways matter
Every breath you take sweeps dust, microbes, and pollutants into your airways. A thin cellular lining acts as a self-cleaning conveyor belt, using beating hair-like structures to push mucus and trapped particles out of the lungs. This study looks at how that conveyor belt is built and maintained, focusing on a little-known membrane protein called caveolin-1 and how it works together with a major cell–fate switch inside the cell. Understanding this partnership could eventually help scientists design better treatments for chronic lung diseases where the airway lining is damaged or imbalanced.
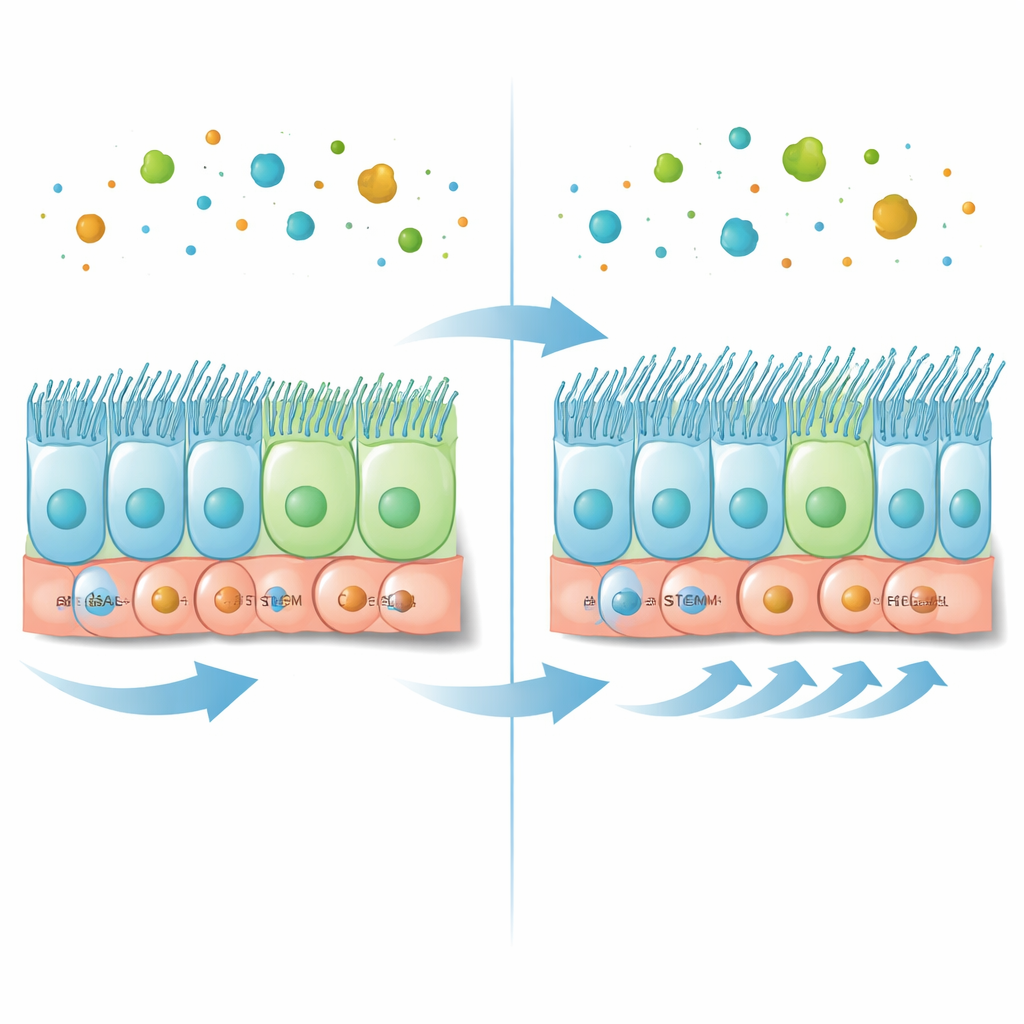
Building blocks of the airway’s cleaning machine
The inner surface of the larger airways is made of a single sheet of different cell types. At the base sit basal stem cells, a reserve pool that can both renew itself and give rise to specialized cells. Some of their descendants become secretory cells that produce mucus, while others become multiciliated cells studded with many moving hairs that drive mucus toward the mouth. The right ratio of these cells is essential for clear airways and healthy breathing. In earlier work, researchers noticed that a protein called caveolin-1 is enriched in basal stem cells and in multiciliated cells, but its precise role in this delicate balance was unclear.
Tracking a hidden organizer in the airway lining
In this study, scientists examined mouse airway tissue and lab-grown airway cultures to map where caveolin-1 appears during the maturation of the epithelium. Using high‑resolution microscopy and gene expression analyses, they found that caveolin-1 is most abundant in basal stem cells and in certain intermediate cells poised to become either mucus producers or multiciliated cells. As the tissue matures in culture, caveolin-1 levels drop overall, in parallel with a rise in markers of multiciliated cells. This pattern suggested that caveolin-1 might act as a brake or fine‑tuner during the transition from stem cells to fully ciliated cells.
What happens when the brake is lifted
To test this idea, the team reduced or removed caveolin-1 in stem cells and followed how the airway lining developed in vitro. The basic organization and barrier function of the epithelium remained intact, and stem cells still divided normally. However, tissues lacking caveolin-1 consistently produced more multiciliated cells, and these cells matured faster. Gene‑expression profiling early in the differentiation process revealed a strong early activation of cilia‑related programs when caveolin-1 was absent. Later on, microscopic imaging showed that cilia were longer and beat more frequently, indicating that not only the number but also the performance of multiciliated cells was enhanced.
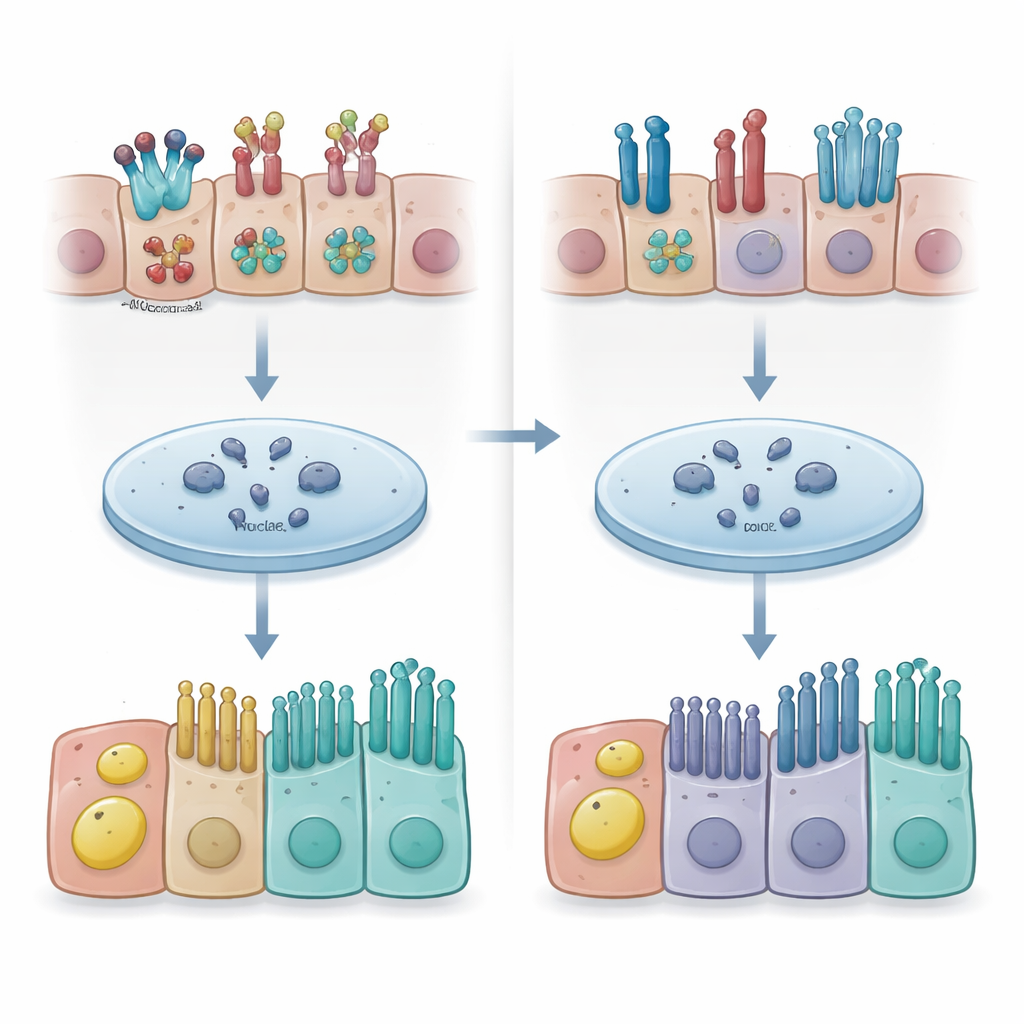
A conversation between the membrane and the cell’s decision switch
The researchers then asked how caveolin-1 could influence such broad changes in cell fate. They turned to Notch signaling, a pathway that acts as a cellular switchboard: when active, it steers basal stem cells toward secretory fates; when suppressed, it favors multiciliated cells. Notch receptors sit in the cell membrane and, when engaged, release an intracellular fragment that travels to the nucleus to control gene activity. The team found that in cells lacking caveolin-1, early Notch signal output was reduced even though receptor and ligand genes were expressed at normal levels. Chromatin‑binding studies showed that the active Notch fragment bound to far fewer sites on DNA when caveolin-1 was depleted. Further biochemical analyses suggested that caveolin-1 helps organize how Notch1 and Notch2 receptors are processed and how effectively their active fragments reach and engage the genome.
Why this matters for lungs in health and disease
Together, these findings point to caveolin-1 as a key coordinator of how airway stem cells decide their fate. Rather than directly changing how often the stem cells divide, caveolin-1 tunes the strength of Notch signaling at the beginning of differentiation, which in turn sets how many cells become multiciliated and how quickly their cilia mature. When caveolin-1 is missing or reduced, Notch’s influence weakens, tipping the balance toward more, and more active, multiciliated cells. In practical terms, this work reveals a molecular lever that could someday be targeted to restore healthy airway linings in conditions such as asthma, chronic obstructive pulmonary disease, or cystic fibrosis, where the cleaning machinery of the lung is often compromised.
Citation: Olivera-Gómez, M., Cumplido-Laso, G., Benitez, D.A. et al. Caveolin-1 modulates Notch transcriptional activity during in vitro respiratory multiciliated cell maturation. Sci Rep 16, 9165 (2026). https://doi.org/10.1038/s41598-026-40201-6
Keywords: airway epithelium, multiciliated cells, caveolin-1, Notch signaling, lung regeneration