Clear Sky Science · en
Kinetics of dissolution and computational modeling of calcium oxalate monohydrate crystals in the presence of aqueous coffee bioactive extract compounds
Why coffee and kidney stones matter
Kidney stones are notoriously painful, and most of them are made from crystals of a salt called calcium oxalate. These crystals can form and dissolve inside our kidneys and urinary tract. This study asks an everyday question with a high-tech twist: what happens to these kidney stone crystals when they meet the natural compounds found in coffee, especially caffeine and related plant molecules? By blending lab experiments with computer simulations, the researchers show how coffee ingredients can cling to crystal surfaces and quietly change their behavior.
Kidney stones and stubborn crystals
Kidney stones are common worldwide, affecting up to one in five people in some regions. The most frequent and most stable form is a crystal called calcium oxalate monohydrate. Because it does not dissolve easily in body fluids, once it forms, it tends to persist and can reappear even after treatment. Earlier work had shown that certain drugs and natural substances can slow crystal growth or dissolution by sticking to the crystal surface. This study explores whether compounds in coffee can act in a similar way, potentially offering a natural route to influence how these stones behave.
What is inside a cup of coffee
The team began by dissecting the chemistry of a coffee extract using advanced tools that separate and identify thousands of molecules. Liquid chromatography and mass spectrometry revealed that the extract is rich in chlorogenic acids and related quinic acid derivatives, along with caffeine as a major component. Nuclear magnetic resonance, a technique that reads molecular “fingerprints,” confirmed caffeine as a dominant ingredient. Together, these analyses built a clear picture of the main bioactive players likely to interact with kidney stone crystals. 
Watching crystals dissolve in real time
To probe how coffee compounds affect crystal behavior, the researchers grew synthetic calcium oxalate monohydrate crystals and placed them in carefully controlled solutions that mimic the chemistry of urine. Using an automated setup that keeps the surrounding solution at a constant composition, they tracked how fast the crystals dissolved over time. As they added small, increasing amounts of coffee extract, the dissolution rate dropped in a concentration‑dependent way, even at very low levels. Analysis of how the rate changed with solution conditions pointed to a “surface‑film controlled” mechanism: coffee molecules adsorb onto reactive spots on the crystal, forming a thin layer that blocks the normal pathways by which the solid dissolves into the liquid.
Probing the surface: from microscopes to math
Several complementary techniques zoomed in on what was happening at the crystal surface. Infrared spectroscopy showed that the internal crystal structure of calcium oxalate stayed the same, with only subtle shifts consistent with molecules sitting on the surface rather than burrowing inside. X‑ray diffraction patterns likewise matched the original crystal phase, though with slight changes in peak intensity suggesting altered surface order. Electron microscopy revealed that crystals exposed to coffee extract became rougher and less sharply faceted, indicating that adsorbed molecules had modified their outer faces. Elemental analysis confirmed small shifts in surface calcium, carbon, and oxygen ratios, again pointing to a change in surface composition without creating a new material.
Simulating caffeine at the atomic level
To understand these interactions in finer detail, the team turned to quantum‑level computer calculations based on density functional theory. They modeled a caffeine molecule approaching a calcium oxalate unit in water and found that caffeine can form stable hydrogen bonds and weak attractive forces known as van der Waals interactions with the crystal. The calculated adsorption energy was modest but clearly favorable, indicating a spontaneous, physical sticking process rather than strong chemical bonding. Electronic structure analysis showed that electron density is shared across the caffeine–crystal pair, stabilizing the complex while leaving the underlying lattice intact. All of this supports the idea that caffeine and related compounds form a protective molecular coating on the crystal surface. 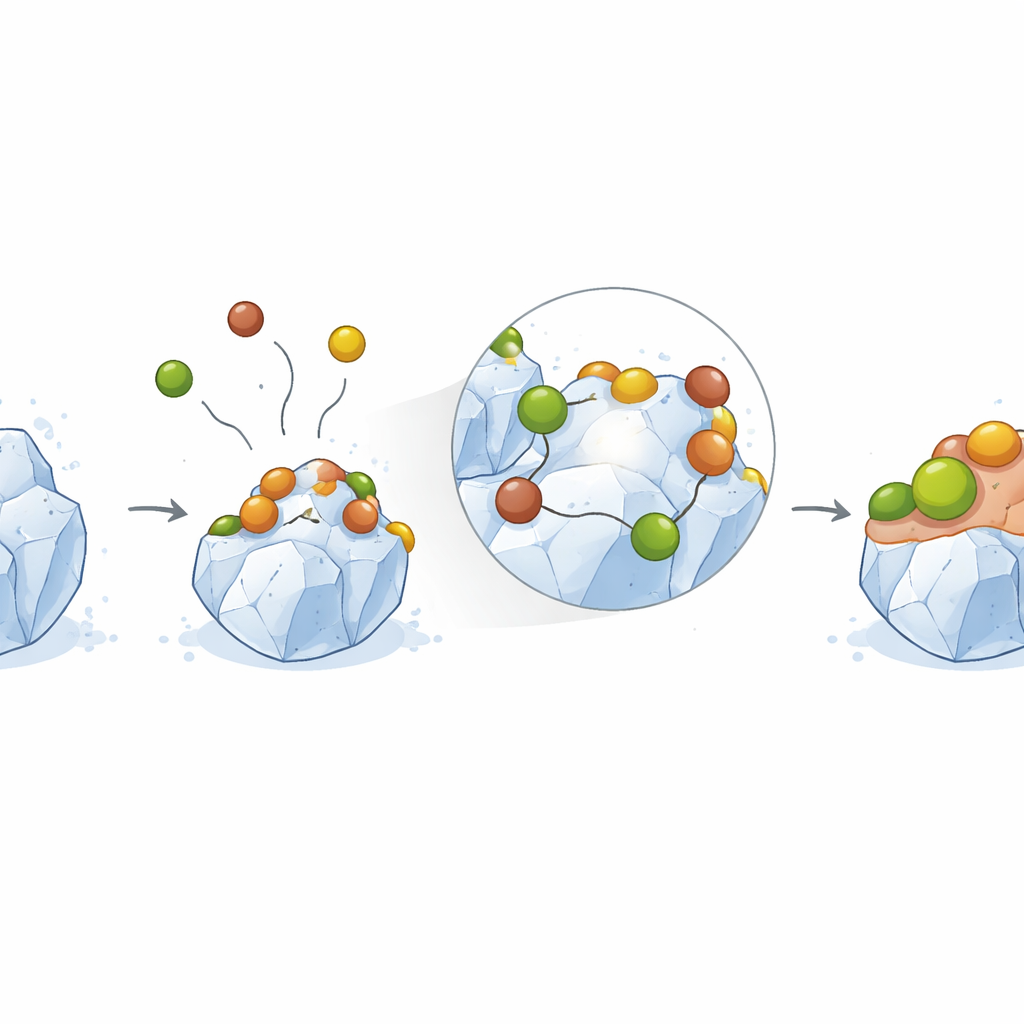
What this means for everyday life
Put simply, this research suggests that some of the natural chemicals in coffee, especially caffeine and quinic acid derivatives, can attach themselves to the surfaces of calcium oxalate kidney stone crystals and slow their dissolution by forming a thin, physical film. They do not rebuild the crystal or change its internal structure, but they do subtly reshape and stabilize its outer skin through gentle molecular attractions. While these experiments were done in simplified model solutions rather than real urine, they highlight coffee metabolites as promising natural modulators of kidney stone behavior and provide a detailed roadmap for how such small molecules can influence crystal surfaces inside the body.
Citation: Khattab, E.T., Yehia, N.S., Sakr, M.A.S. et al. Kinetics of dissolution and computational modeling of calcium oxalate monohydrate crystals in the presence of aqueous coffee bioactive extract compounds. Sci Rep 16, 9681 (2026). https://doi.org/10.1038/s41598-026-40198-y
Keywords: kidney stones, calcium oxalate, caffeine, coffee extract, crystal dissolution