Clear Sky Science · en
Multimodal characterization of flow-induced thrombus initiation and growth in extracorporeal membrane oxygenation
Why blood clots in life-support machines matter
Extracorporeal membrane oxygenation, or ECMO, is a form of heart–lung bypass that can keep critically ill patients alive while their organs recover. But running blood through pumps and plastic tubes exposes it to harsh mechanical forces that our bodies were never designed to handle. These forces can trigger dangerous blood clots inside the circuit, which in turn raise the risk of stroke, organ damage, or machine failure. This study set out to uncover how, exactly, the flow of blood through an ECMO pump shapes the birth and growth of these clots, with the long-term goal of making ECMO both safer and more effective.
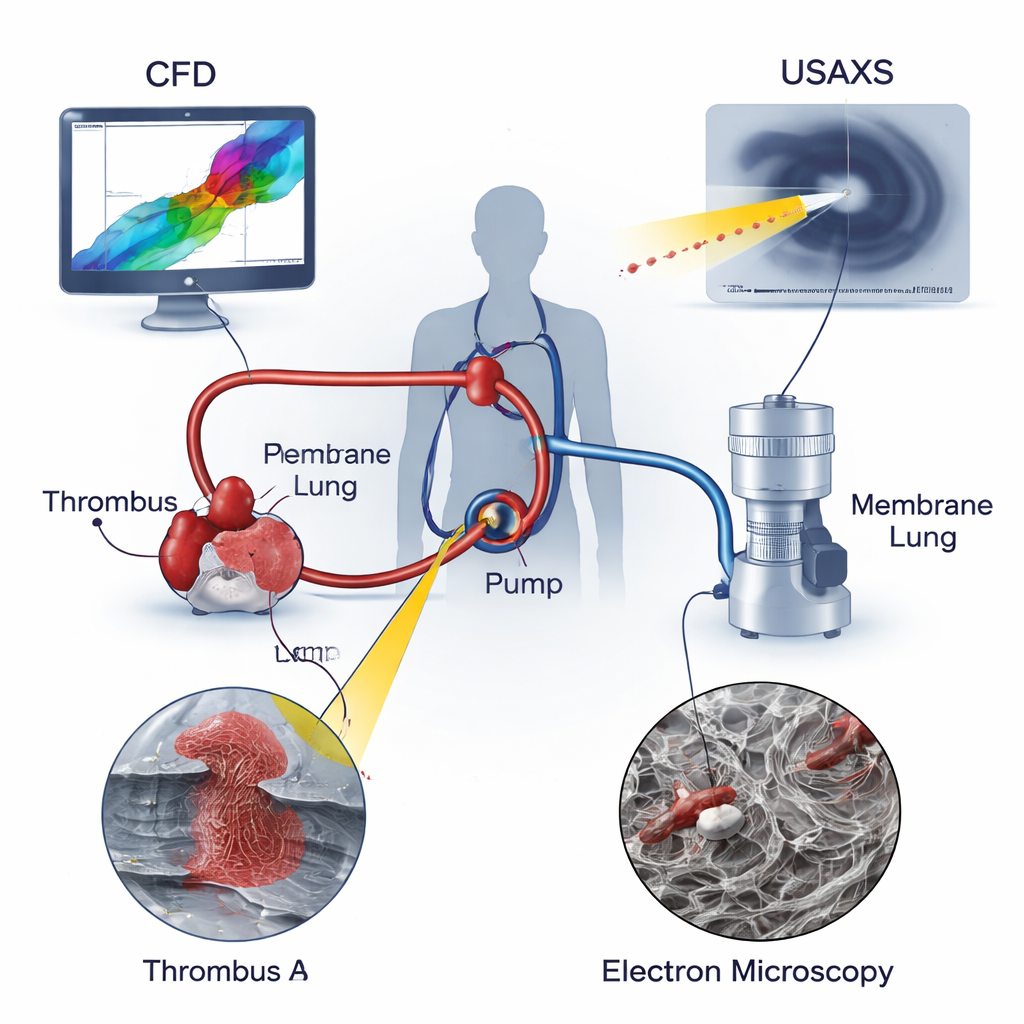
How ECMO keeps patients alive
In ECMO, blood is drawn from a large vein or artery, pushed by a centrifugal pump through a “membrane lung” that adds oxygen and removes carbon dioxide, and then returned to the patient. Unlike blood flowing smoothly through natural vessels, flow in an ECMO circuit includes extreme conditions: very fast streams, sharp changes in direction, and nearly stagnant pockets where blood lingers. These environments are known to damage blood cells and to stimulate clotting. Classic medical models focus on slow or blocked flow in blood vessels, but they do not fully account for the intense shearing and stretching that blood experiences inside a spinning pump. The authors argue that to truly understand clot risk in ECMO, one must study both the mechanical forces in the device and the microscopic structure of the clots that emerge.
Looking at clots from three angles
The researchers combined three powerful tools to analyze two real clots taken from ECMO circuits used in children: one clot at the pump inlet (Thrombus A) and another in tubing just downstream of the pump (Thrombus B). Computational fluid dynamics (CFD) simulated how blood moved through the pump, revealing regions of recirculating and swirling flow and pinpointing where shear and stretching forces peaked. Ultra-small-angle X-ray scattering (USAXS) probed deep into each clot, measuring how tightly packed and how directionally aligned the fibrin scaffold—the protein mesh that holds clots together—was throughout the sample. Scanning electron microscopy (SEM) provided highly magnified images of the clot surfaces, showing the shapes of red blood cells, white blood cells, platelets, and the surrounding fibrin fibers. By overlaying these three views, the team could link local flow conditions to the internal architecture of each clot.
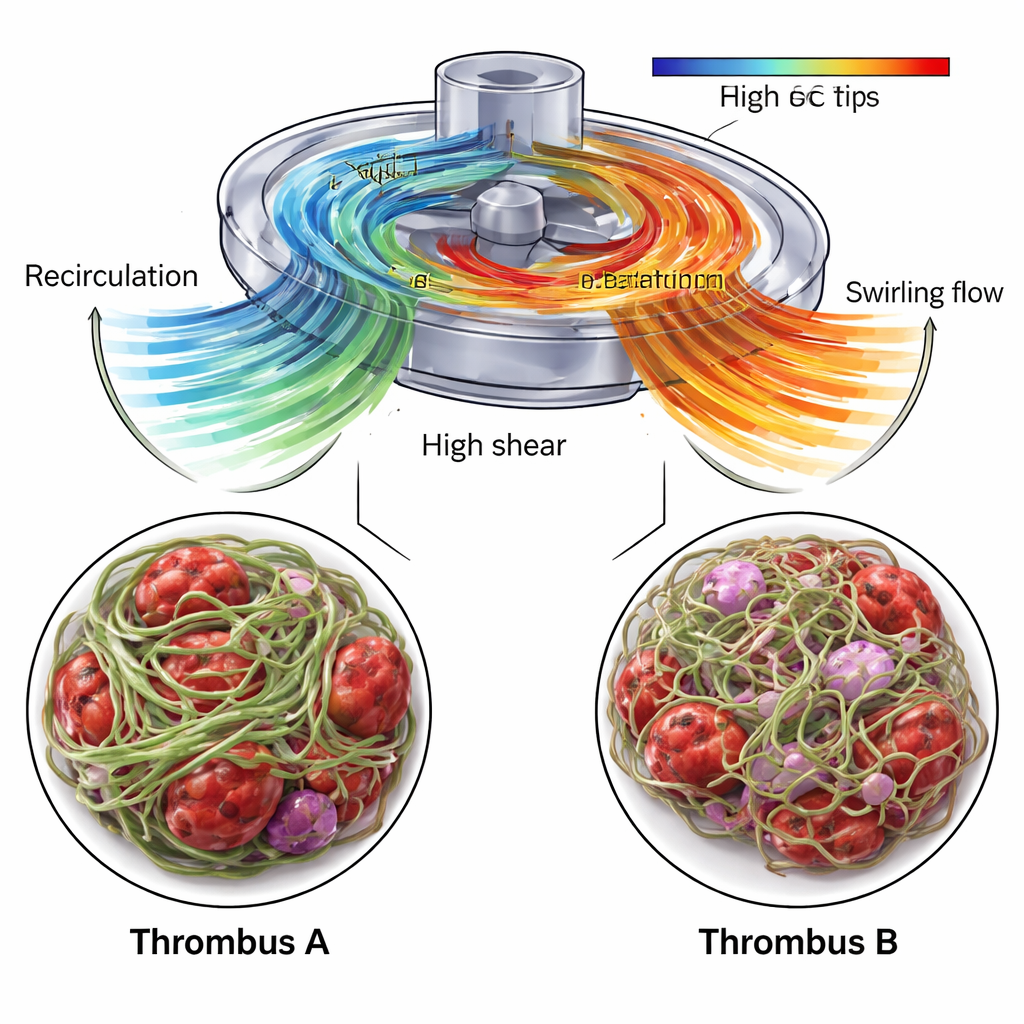
A recirculation zone builds a stiff, aligned clot
CFD showed that the area near the pump inlet, where Thrombus A formed, contained a recirculation zone: blood was pushed backward and upward along the housing before rejoining the main stream. This region allowed blood components to linger while also experiencing sharp velocity differences at the boundary between recirculating and incoming flow. Inside Thrombus A, USAXS indicated a high fibrin content—at least 70 percent—and a strong overall alignment of fibers in a preferred direction, suggesting a dense, stiff scaffold. SEM images confirmed a tightly woven network of fibrin, interspersed with abnormally shaped red blood cells and fragments of platelets. The authors propose that the combination of long residence times and strong local shear favored the growth of a compact, highly organized clot able to withstand the mechanical stresses of the pump.
Swirling outflow shapes a looser, twisted clot
In contrast, Thrombus B, taken from tubing after the pump, grew in a region dominated by swirling outflow. CFD revealed rotating, spiral-like flow structures emerging from the pump outlet, and the USAXS data showed a fibrin network that was still dominant but less dense and less strongly aligned overall. The main direction of fibrin orientation shifted gradually across the clot, from one tilt angle to another, echoing the twisting flow pattern. SEM images showed fibrin threads of varying thickness and numerous trapped red and white blood cells, including evidence of cell damage and inflammation. Importantly, the simulations also identified small but significant portions of the pump volume where stretching forces were high enough to unfold von Willebrand factor, a key blood protein that becomes sticky under stress and can rapidly recruit platelets. These zones were concentrated near the impeller blades and outlet, making them likely sites for the initial activation events that led to Thrombus B.
Toward safer life-support machines
By merging detailed simulations of blood flow with X-ray and electron microscope measurements of real ECMO clots, this work shows that the internal “grain” and density of a clot mirror the mechanical environment in which it formed. Recirculating flow near the pump inlet was linked to a dense, highly aligned fibrin scaffold, while swirling outflow was associated with a more twisted, open network that still trapped many blood and immune cells. These insights highlight specific regions in ECMO pumps and tubing where design changes or targeted adjustments to anticoagulation might most effectively reduce clot risk. In the long run, such multiscale mapping—from pump geometry and flow patterns down to protein arrangement—could guide safer device designs and help clinicians better balance the twin dangers of bleeding and thrombosis during life-saving ECMO support.
Citation: Nilsson, F., Sochor, B., Henriksson, S. et al. Multimodal characterization of flow-induced thrombus initiation and growth in extracorporeal membrane oxygenation. Sci Rep 16, 7166 (2026). https://doi.org/10.1038/s41598-026-40177-3
Keywords: ECMO, blood clotting, shear stress, centrifugal blood pump, fibrin structure